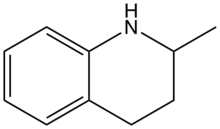
2-Methyltetrahydroquinoline
2-Methyltetrahydroquinoline is one of the methyl-substituted derivatives of tetrahydroquinoline. A colorless oil, it is a chiral compound owing to the presence of the methyl substituent. It is produced by the hydrogenation of quinaldine.[1] It is of interest in medicinal chemistry.[2]
 | |
| Names | |
|---|---|
| Other names
Tetrahydroquinaldine, 1,2,3,4-Tetrahydroquinaldine | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL |
|
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H13N | |
| Molar mass | 147.221 g·mol−1 |
| Appearance | colorless oil |
| Boiling point | 125 °C (257 °F; 398 K) (17 Torr) |
| Hazards | |
| GHS pictograms |  |
| GHS Signal word | Warning |
| H315, H319 | |
| P264, P280, P302+352, P305+351+338, P321, P332+313, P337+313, P362 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
References
- Chakraborty, Sumit; Brennessel, William W.; Jones, William D. (2014). "A Molecular Iron Catalyst for the Acceptorless Dehydrogenation and Hydrogenation of N-Heterocycles". Journal of the American Chemical Society. 136 (24): 8564–8567. doi:10.1021/ja504523b. PMID 24877556.
- Sridharan, Vellaisamy; Suryavanshi, Padmakar A.; Menéndez, J. Carlos (2011). "Advances in the Chemistry of Tetrahydroquinolines". Chemical Reviews. 111 (11): 7157–7259. doi:10.1021/cr100307m. PMID 21830756.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.