Danheiser annulation
The Danheiser annulation or Danheiser TMS-cyclopentene annulation is an organic reaction of an α,β-unsaturated ketone and a trialkylsilylallene (e.g., trimethylsilyl- or triisopropylsilyl-) in the presence of a Lewis Acid to give a trialkylsilylcyclopentene in a regiocontrolled annulation.[1][2][3][4][5][6]
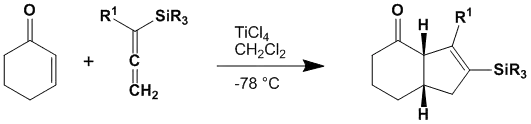
References
- R. L. Danheiser; D. J. Carini; A. Basak (1981). "TMS-Cyclopentene Annulation: A Regiocontrolled Approach to the Synthesis of Five-Membered Rings". J. Am. Chem. Soc. 103 (6): 1604. doi:10.1021/ja00396a071.
- R. L. Danheiser; D. J. Carini; D. M. Fink; A. Basak (1983). "Scope and Stereochemical Course of the (Trimethylsilyl)cyclopentene Annulation". Tetrahedron. 39 (6): 935. doi:10.1016/S0040-4020(01)88592-6.
- R. L. Danheiser; D. M. Fink (1985). "The Reaction of Allenysilanes with α,β-Unsaturated Acylsilanes: New Annulation Approaches to Five and Six-Membered Carbocyclic Compounds". Tetrahedron Letters. 26 (21): 2513. doi:10.1016/S0040-4039(00)98824-5.
- R. L. Danheiser; D. M. Fink; Y. -M. Tsai (1988). "A General [3+2] Annulation: cis-4-Exo-isopropenyl-1,9-dimethyl-8-(trimethylsilyl)bicyclo-[4.3.0]non-8-en-2-one". Organic Syntheses. 66: 8. doi:10.15227/orgsyn.066.0008.
- Alfred Hassner; C. Stumer (2002). Organic Syntheses Based on Name Reactions. Elsevier. ISBN 008043259X.
- Jie Jack Li (2003). Name Reactions: A Collection of Detailed Reaction Mechanisms. Springer. p. 102. ISBN 3540402039.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.