Dioctyl terephthalate
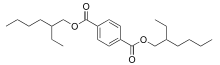
Dioctyl terephthalate (bis(2-ethylhexyl) benzene-1,4-dicarboxylate or Di(2-ethylhexyl) terephthalate), commonly abbreviated DOTP or DEHT, is an organic compound with the formula C6H4(CO2C8H17)2. It is a non-phthalate plasticizer, being the diester of terephthalic acid and the branched-chain 2-ethylhexanol. This colorless viscous liquid used for softening PVC plastics is known for chemical similarity to general purpose phthalates such as DEHP and DINP, but without any negative regulatory pressure. It possesses very good plasticizing properties and may be used as a direct replacement for DEHP and DINP in many applications.
 | |
| Names | |
|---|---|
| IUPAC name
Bis(2-ethylhexyl) benzene-1,4-dicarboxylate | |
| Other names
Dioctyl Terephthalate; Bis(2-ethylhexyl) terephthalate; Di(ethylhexyl) terephthalate; 1,4-Benzenedicarboxylic acid bis(2-ethylhexyl) ester | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | DOTP, DEHT |
| ChemSpider | |
| ECHA InfoCard | 100.026.524 |
| EC Number |
|
| MeSH | C053316 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C24H38O4 | |
| Molar mass | 390.564 g·mol−1 |
| Appearance | Clear viscous liquid |
| Density | 0.984 g/mL |
| Boiling point | 400 °C (752 °F; 673 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Production
One method of manufacture entails the transesterification of dimethyl terephthalate with 2-ethylhexanol:
- C6H4(CO)2(OCH3)2 + 2 C8H17OH → C6H4(CO2 C8H17)2 + 2CH3OH
A second method of manufacture is a direct esterification of terephthalic acid with 2-ethylhexanol:
- C6H4(CO2H)2 + 2 C8H17OH → C6H4(CO2 C8H17)2 + 2H2O
Use
DOTP is a general purpose plasticizer that is considered safer than ortho-phthalate plasticizers due to its reduced toxicity profile. The terephthalates exhibit none of the peroxisome proliferation of liver enzymes that some ortho-phthalates have shown in several studies.[1] It has uses in applications like extrusion, calendering, injection molding, rotational molding, dip molding, slush molding and coating.[2]
Alternative plasticizers
There are several alternative plasticizers offering similar technical properties to DOTP. These alternatives include phthalates such as DINP, DOP, DPHP, DIDP as well as non-phthalates such as DINCH and citrate esters.
Possible Health Effects
According to a panel appointed by the French Agency for the Safety of Health Products (ANSM), there is very little clinical assessment data regarding the toxicity, migration from Medical Devices, and population exposure of alternative plasticizers (e.g. DOTP/DEHT) and their metabolites.[3] The actual French government report concludes, "The presently available information indicates that DEHTP is not expected to pose any health or environmental risks. DEHTP is not considered as toxic for reproduction and no alert was found on potential endocrine disruption properties of the substance."[4]
Researchers from Korea state that: "DEHT, a phthalate ester stoichiometrically eqivaluent to DEHP, has been shown to have potential reproductive and developmental toxicity."[5] The NOAELs quoted are actually quite high, indicating the potential for reproductive toxicity is low.
References
- OECD SIDS Initial Assessment Report for SIAM 17 (2003)
- "Archived copy". Archived from the original on 2012-03-22. Retrieved 2011-04-11.CS1 maint: archived copy as title (link)
- Bui, Thuy T.; Giovanoulis, Georgios; Cousins, Anna Palm; Magnér, Jörgen; Cousins, Ian T.; de Wit, Cynthia A. (2016-01-15). "Human exposure, hazard and risk of alternative plasticizers to phthalate esters". Science of the Total Environment. 541: 451–467. doi:10.1016/j.scitotenv.2015.09.036. PMID 26410720.
- French RMOA - DEHTP
- Kwon, Bareum; Ji, Kyunghee (2016). "Estrogenic and Androgenic Potential of Phthalates and Their Alternatives". Korean Journal of Environmental Health Sciences. 42 (3): 169–188. doi:10.5668/JEHS.2016.42.3.169.