Fluo-3
Fluo-3 is a fluorescence indicator of intracellular calcium (Ca2+). It is used to measure Ca2+ inside living cells in flow cytometry and confocal laser scanning microscopy using visible light excitation (compatible with argon laser sources operating at 488 nm). Fluo-3 is an essentially nonfluorescent compound, but upon binding of Ca2+ its fluorescence increases sharply with an emission maximum at 525 nm suitable for conventionally used detectors designed for fluorescein isothiocyanate (FITC) measurements. This large change in fluorescence coupled with a good yield of photons provides very high contrast which allowed the detection of microscopic Ca2+ release events inside cells called "Calcium sparks".[1] Whereas the salts of fluo-3 are unable to penetrate cells, loading can be achieved using its acetoxymethyl (AM) ester derivative. Once inside the cell, unspecific esterases cleave the ester effectively trapping fluo-3.[2]
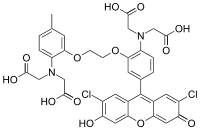 | |
| Names | |
|---|---|
| IUPAC name
{[2-(2-{2-[Bis(carboxymethyl)amino]-5-(2,7-dichloro-6-hydroxy-3-oxo-3H-xanthen-9-yl)phenoxy}ethoxy)-4-methylphenyl](carboxymethyl)amino}acetic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C36H30Cl2N2O13 | |
| Molar mass | 769.54 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
As calcium is a key second messenger within cells, the specific properties of fluo-3 enable researchers to investigate the time-resolved dynamics of intracellular signal transduction in a diverse range of cells.[3][4]
References
- Cheng, H.; Lederer, W.J.; Cannell, M.B. (1993). "Calcium Sparks - Elementary Events Underlying Excitation-Contraction Coupling in Heart-Muscle". Science. 262 (5134): 740–744. doi:10.1126/science.8235594. PMID 8235594.
- Haugland, RP. Handbook of Fluorescent Probes and Research Products. Molecular Probes, 2010
- Gamsjäger, T. Flow Cytometry of Intracellular Calcium in Platelets. Grin, 2012
- Lambert, DG. Calcium Signaling Protocols. Humana Press, 2006