Lymphocyte cytosolic protein 2
Lymphocyte cytosolic protein 2 (SH2 domain containing leukocyte protein of 76kDa), also known as LCP2 or SLP-76, is a gene that encodes a signal-transducing adaptor protein.[5]
Structure
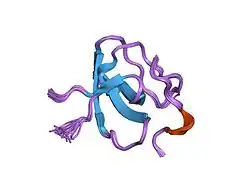
No full structure for SLP-76 has been solved. The PDB file 1H3H depicts the SH3 domain of GRAP2 in complex with an RSTK-containing peptide representing residues 226–235 of SLP-76.
Function
SLP-76 was originally identified as a substrate of the ZAP-70 protein tyrosine kinase following T cell receptor (TCR) ligation in the leukemic T cell line Jurkat. The SLP-76 locus has been localized to human chromosome 5q33 and the gene structure has been partially characterized in mice. The human and murine cDNAs both encode 533 amino acid proteins that are 72% identical and composed of three modular domains. The NH2-terminus contains an acidic region that includes a PEST domain and several tyrosine residues that are phosphorylated following TCR ligation. SLP-76 also contains a central proline-rich domain and a COOH-terminal SH2 domain. A number of additional proteins have been identified that associate with SLP-76 both constitutively and inducibly following receptor ligation, supporting the notion that SLP-76 functions as an adaptor or scaffold protein. Studies using SLP-76-deficient T cell lines or mice have provided strong evidence that SLP-76 plays a positive role in promoting T cell development and activation as well as mast cell and platelet function.[5] SLP-76 might serve as an integration point for signals by activating NK cell receptors.[6] In NK cells, SLP-76 can be phosphorylated by SYK or ZAP70 following ligation of activating receptors.[7]
Interactions
Lymphocyte cytosolic protein 2 has been shown to interact with:
See also
References
- GRCh38: Ensembl release 89: ENSG00000043462 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000002699 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Entrez Gene: LCP2 lymphocyte cytosolic protein 2 (SH2 domain containing leukocyte protein of 76kDa)".
- Kim, Hun Sik; Long, Eric O. (2012-07-10). "Complementary Phosphorylation Sites in the Adaptor Protein SLP-76 Promote Synergistic Activation of Natural Killer Cells". Science Signaling. 5 (232): ra49. doi:10.1126/scisignal.2002754. ISSN 1937-9145. PMC 3842037. PMID 22786724.
- Fasbender, Frank; Claus, Maren; Wingert, Sabine; Sandusky, Mina; Watzl, Carsten (2017-07-07). "Differential Requirements for Src-Family Kinases in SYK or ZAP70-Mediated SLP-76 Phosphorylation in Lymphocytes". Frontiers in Immunology. 8: 789. doi:10.3389/fimmu.2017.00789. ISSN 1664-3224. PMC 5500614. PMID 28736554.
- Park RK, Izadi KD, Deo YM, Durden DL (September 1999). "Role of Src in the modulation of multiple adaptor proteins in FcalphaRI oxidant signaling". Blood. 94 (6): 2112–2120. doi:10.1182/blood.V94.6.2112. PMID 10477741.
- Erdreich-Epstein A, Liu M, Kant AM, Izadi KD, Nolta JA, Durden DL (April 1999). "Cbl functions downstream of Src kinases in Fc gamma RI signaling in primary human macrophages". J. Leukoc. Biol. 65 (4): 523–534. doi:10.1002/jlb.65.4.523. PMID 10204582. S2CID 18340540.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (October 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–1178. Bibcode:2005Natur.437.1173R. doi:10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Ellis JH, Ashman C, Burden MN, Kilpatrick KE, Morse MA, Hamblin PA (June 2000). "GRID: a novel Grb-2-related adapter protein that interacts with the activated T cell costimulatory receptor CD28". J. Immunol. 164 (11): 5805–5814. doi:10.4049/jimmunol.164.11.5805. PMID 10820259.
- Liu SK, Fang N, Koretzky GA, McGlade CJ (January 1999). "The hematopoietic-specific adaptor protein gads functions in T-cell signaling via interactions with the SLP-76 and LAT adaptors". Curr. Biol. 9 (2): 67–75. doi:10.1016/S0960-9822(99)80017-7. PMID 10021361. S2CID 14131281.
- Asada H, Ishii N, Sasaki Y, Endo K, Kasai H, Tanaka N, Takeshita T, Tsuchiya S, Konno T, Sugamura K (May 1999). "Grf40, A novel Grb2 family member, is involved in T cell signaling through interaction with SLP-76 and LAT". J. Exp. Med. 189 (9): 1383–1390. doi:10.1084/jem.189.9.1383. PMC 2193052. PMID 10224278.
- Lewitzky M, Kardinal C, Gehring NH, Schmidt EK, Konkol B, Eulitz M, Birchmeier W, Schaeper U, Feller SM (March 2001). "The C-terminal SH3 domain of the adapter protein Grb2 binds with high affinity to sequences in Gab1 and SLP-76 which lack the SH3-typical P-x-x-P core motif". Oncogene. 20 (9): 1052–1062. doi:10.1038/sj.onc.1204202. PMID 11314042.
- Robinson A, Gibbins J, Rodríguez-Liñares B, Finan PM, Wilson L, Kellie S, Findell P, Watson SP (July 1996). "Characterization of Grb2-binding proteins in human platelets activated by Fc gamma RIIA cross-linking". Blood. 88 (2): 522–530. doi:10.1182/blood.V88.2.522.bloodjournal882522. PMID 8695800.
- Hendricks-Taylor LR, Motto DG, Zhang J, Siraganian RP, Koretzky GA (January 1997). "SLP-76 is a substrate of the high affinity IgE receptor-stimulated protein tyrosine kinases in rat basophilic leukemia cells". J. Biol. Chem. 272 (2): 1363–1367. doi:10.1074/jbc.272.2.1363. PMID 8995445.
- Shim EK, Moon CS, Lee GY, Ha YJ, Chae SK, Lee JR (September 2004). "Association of the Src homology 2 domain-containing leukocyte phosphoprotein of 76 kD (SLP-76) with the p85 subunit of phosphoinositide 3-kinase". FEBS Lett. 575 (1–3): 35–40. doi:10.1016/j.febslet.2004.07.090. PMID 15388330. S2CID 24678709.
- Bunnell SC, Diehn M, Yaffe MB, Findell PR, Cantley LC, Berg LJ (January 2000). "Biochemical interactions integrating Itk with the T cell receptor-initiated signaling cascade". J. Biol. Chem. 275 (3): 2219–2230. doi:10.1074/jbc.275.3.2219. PMID 10636929.
- Gross BS, Lee JR, Clements JL, Turner M, Tybulewicz VL, Findell PR, Koretzky GA, Watson SP (February 1999). "Tyrosine phosphorylation of SLP-76 is downstream of Syk following stimulation of the collagen receptor in platelets". J. Biol. Chem. 274 (9): 5963–5971. doi:10.1074/jbc.274.9.5963. PMID 10026222.
- Wunderlich L, Faragó A, Downward J, Buday L (April 1999). "Association of Nck with tyrosine-phosphorylated SLP-76 in activated T lymphocytes". Eur. J. Immunol. 29 (4): 1068–1075. doi:10.1002/(SICI)1521-4141(199904)29:04<1068::AID-IMMU1068>3.0.CO;2-P. PMID 10229072.
- Yablonski D, Kadlecek T, Weiss A (July 2001). "Identification of a phospholipase C-gamma1 (PLC-gamma1) SH3 domain-binding site in SLP-76 required for T-cell receptor-mediated activation of PLC-gamma1 and NFAT". Mol. Cell. Biol. 21 (13): 4208–4218. doi:10.1128/MCB.21.13.4208-4218.2001. PMC 87082. PMID 11390650.
- Binstadt BA, Billadeau DD, Jevremović D, Williams BL, Fang N, Yi T, Koretzky GA, Abraham RT, Leibson PJ (October 1998). "SLP-76 is a direct substrate of SHP-1 recruited to killer cell inhibitory receptors". J. Biol. Chem. 273 (42): 27518–27523. doi:10.1074/jbc.273.42.27518. PMID 9765283.
- Mizuno K, Katagiri T, Hasegawa K, Ogimoto M, Yakura H (August 1996). "Hematopoietic cell phosphatase, SHP-1, is constitutively associated with the SH2 domain-containing leukocyte protein, SLP-76, in B cells". J. Exp. Med. 184 (2): 457–463. doi:10.1084/jem.184.2.457. PMC 2192711. PMID 8760799.
- Lindholm CK, Henriksson ML, Hallberg B, Welsh M (July 2002). "Shb links SLP-76 and Vav with the CD3 complex in Jurkat T cells". Eur. J. Biochem. 269 (13): 3279–3288. doi:10.1046/j.1432-1033.2002.03008.x. PMID 12084069.
- Raab M, da Silva AJ, Findell PR, Rudd CE (February 1997). "Regulation of Vav-SLP-76 binding by ZAP-70 and its relevance to TCR zeta/CD3 induction of interleukin-2". Immunity. 6 (2): 155–164. doi:10.1016/S1074-7613(00)80422-7. PMID 9047237.
- Onodera H, Motto DG, Koretzky GA, Rothstein DM (September 1996). "Differential regulation of activation-induced tyrosine phosphorylation and recruitment of SLP-76 to Vav by distinct isoforms of the CD45 protein-tyrosine phosphatase". J. Biol. Chem. 271 (36): 22225–22230. doi:10.1074/jbc.271.36.22225. PMID 8703037.
Further reading
- Jackman JK, Motto DG, Sun Q, Tanemoto M, Turck CW, Peltz GA, Koretzky GA, Findell PR (1995). "Molecular cloning of SLP-76, a 76-kDa tyrosine phosphoprotein associated with Grb2 in T cells". J. Biol. Chem. 270 (13): 7029–7032. doi:10.1074/jbc.270.13.7029. PMID 7706237.
- Sunden SL, Carr LL, Clements JL, Motto DG, Koretzky GA (1996). "Polymorphism in and localization of the gene LCP2 (SLP-76) to chromosome 5q33.1-qter". Genomics. 35 (1): 269–270. doi:10.1006/geno.1996.0354. PMID 8661136.
- Motto DG, Ross SE, Wu J, Hendricks-Taylor LR, Koretzky GA (1996). "Implication of the GRB2-associated phosphoprotein SLP-76 in T cell receptor-mediated interleukin 2 production". J. Exp. Med. 183 (4): 1937–1943. doi:10.1084/jem.183.4.1937. PMC 2192521. PMID 8666952.
- Bubeck Wardenburg J, Fu C, Jackman JK, Flotow H, Wilkinson SE, Williams DH, Johnson R, Kong G, Chan AC, Findell PR (1996). "Phosphorylation of SLP-76 by the ZAP-70 protein-tyrosine kinase is required for T-cell receptor function". J. Biol. Chem. 271 (33): 19641–19644. doi:10.1074/jbc.271.33.19641. PMID 8702662.
- Onodera H, Motto DG, Koretzky GA, Rothstein DM (1996). "Differential regulation of activation-induced tyrosine phosphorylation and recruitment of SLP-76 to Vav by distinct isoforms of the CD45 protein-tyrosine phosphatase". J. Biol. Chem. 271 (36): 22225–22230. doi:10.1074/jbc.271.36.22225. PMID 8703037.
- Mizuno K, Katagiri T, Hasegawa K, Ogimoto M, Yakura H (1996). "Hematopoietic cell phosphatase, SHP-1, is constitutively associated with the SH2 domain-containing leukocyte protein, SLP-76, in B cells". J. Exp. Med. 184 (2): 457–463. doi:10.1084/jem.184.2.457. PMC 2192711. PMID 8760799.
- Raab M, da Silva AJ, Findell PR, Rudd CE (1997). "Regulation of Vav-SLP-76 binding by ZAP-70 and its relevance to TCR zeta/CD3 induction of interleukin-2". Immunity. 6 (2): 155–164. doi:10.1016/S1074-7613(00)80422-7. PMID 9047237.
- Musci MA, Hendricks-Taylor LR, Motto DG, Paskind M, Kamens J, Turck CW, Koretzky GA (1997). "Molecular cloning of SLAP-130, an SLP-76-associated substrate of the T cell antigen receptor-stimulated protein tyrosine kinases". J. Biol. Chem. 272 (18): 11674–11677. doi:10.1074/jbc.272.18.11674. PMID 9115214.
- da Silva AJ, Li Z, de Vera C, Canto E, Findell P, Rudd CE (1997). "Cloning of a novel T-cell protein FYB that binds FYN and SH2-domain-containing leukocyte protein 76 and modulates interleukin 2 production". Proc. Natl. Acad. Sci. U.S.A. 94 (14): 7493–7498. Bibcode:1997PNAS...94.7493D. doi:10.1073/pnas.94.14.7493. PMC 23849. PMID 9207119.
- Fu C, Chan AC (1997). "Identification of two tyrosine phosphoproteins, pp70 and pp68, which interact with phospholipase Cgamma, Grb2, and Vav after B cell antigen receptor activation". J. Biol. Chem. 272 (43): 27362–27368. doi:10.1074/jbc.272.43.27362. PMID 9341187.
- Zhang W, Sloan-Lancaster J, Kitchen J, Trible RP, Samelson LE (1998). "LAT: the ZAP-70 tyrosine kinase substrate that links T cell receptor to cellular activation". Cell. 92 (1): 83–92. doi:10.1016/S0092-8674(00)80901-0. PMID 9489702. S2CID 1806525.
- Fu C, Turck CW, Kurosaki T, Chan AC (1998). "BLNK: a central linker protein in B cell activation". Immunity. 9 (1): 93–103. doi:10.1016/S1074-7613(00)80591-9. PMID 9697839.
- Chu J, Liu Y, Koretzky GA, Durden DL (1998). "SLP-76-Cbl-Grb2-Shc interactions in FcgammaRI signaling". Blood. 92 (5): 1697–1706. doi:10.1182/blood.V92.5.1697. PMID 9716598.
- Binstadt BA, Billadeau DD, Jevremović D, Williams BL, Fang N, Yi T, Koretzky GA, Abraham RT, Leibson PJ (1998). "SLP-76 is a direct substrate of SHP-1 recruited to killer cell inhibitory receptors". J. Biol. Chem. 273 (42): 27518–27523. doi:10.1074/jbc.273.42.27518. PMID 9765283.
- Liu SK, Fang N, Koretzky GA, McGlade CJ (1999). "The hematopoietic-specific adaptor protein gads functions in T-cell signaling via interactions with the SLP-76 and LAT adaptors". Curr. Biol. 9 (2): 67–75. doi:10.1016/S0960-9822(99)80017-7. PMID 10021361. S2CID 14131281.
- Gross BS, Lee JR, Clements JL, Turner M, Tybulewicz VL, Findell PR, Koretzky GA, Watson SP (1999). "Tyrosine phosphorylation of SLP-76 is downstream of Syk following stimulation of the collagen receptor in platelets". J. Biol. Chem. 274 (9): 5963–5971. doi:10.1074/jbc.274.9.5963. PMID 10026222.
- Erdreich-Epstein A, Liu M, Kant AM, Izadi KD, Nolta JA, Durden DL (1999). "Cbl functions downstream of Src kinases in Fc gamma RI signaling in primary human macrophages". J. Leukoc. Biol. 65 (4): 523–534. doi:10.1002/jlb.65.4.523. PMID 10204582. S2CID 18340540.
- Asada H, Ishii N, Sasaki Y, Endo K, Kasai H, Tanaka N, Takeshita T, Tsuchiya S, Konno T, Sugamura K (1999). "Grf40, A novel Grb2 family member, is involved in T cell signaling through interaction with SLP-76 and LAT". J. Exp. Med. 189 (9): 1383–1390. doi:10.1084/jem.189.9.1383. PMC 2193052. PMID 10224278.
- Wunderlich L, Faragó A, Downward J, Buday L (1999). "Association of Nck with tyrosine-phosphorylated SLP-76 in activated T lymphocytes". Eur. J. Immunol. 29 (4): 1068–1075. doi:10.1002/(SICI)1521-4141(199904)29:04<1068::AID-IMMU1068>3.0.CO;2-P. PMID 10229072.
- Gross BS, Melford SK, Watson SP (1999). "Evidence that phospholipase C-gamma2 interacts with SLP-76, Syk, Lyn, LAT and the Fc receptor gamma-chain after stimulation of the collagen receptor glycoprotein VI in human platelets". Eur. J. Biochem. 263 (3): 612–623. doi:10.1046/j.1432-1327.1999.00560.x. PMID 10469124.
External links
- SLP-76+signal+Transducing+adaptor+proteins at the US National Library of Medicine Medical Subject Headings (MeSH)