Millon's reagent
Millon's reagent is an analytical reagent used to detect the presence of soluble proteins. A few drops of the reagent are added to the test solution, which is then heated gently. A reddish-brown coloration or precipitate indicates the presence of tyrosine residue which occur in nearly all proteins.[1] The test was developed by the French chemist Auguste Nicolas Eugene Millon (1812–1867).
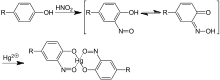
The reagent is made by dissolving metallic mercury in nitric acid and diluting with water. In the test, the phenol group in the side chain of tyrosine gets nitrated, and that product then complexes with Hg(I) or Hg(II) ions to give red coloration or precipitate. Millon's test is not specific for proteins; it also gives a positive test for other compounds containing the phenol functional group. Therefore, the biuret test or the ninhydrin reaction are used along with it to confirm the presence of proteins.
References