Nitrogen difluoride
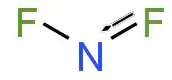
Nitrogen difluoride, also known as difluoroamino is a reactive radical molecule with formula NF2. This small molecule is in equilibrium with its dimer dinitrogen tetrafluoride.[2]
- N2F4 ⇌ 2 NF2•
 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| NF 2 | |
| Related compounds | |
Related nitrogen fluorides |
Nitrogen trifluoride dinitrogen tetrafluoride nitrogen monofluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
As the temperature increases the proportion of NF2 increases.[3]
The molecule is unusual in that it has an odd number of electrons, yet is stable enough to study experimentally.[4]
Properties
The energy needed to break the N-N bond in N2F4 is 20.8 kcal/mol, with an entropy change of 38.6 eu.[5] For comparison, the dissociation energy of the N-N bond is 14.6 kcal/mol in N2O4, 10.2 kcal/mol in N2O2, and 60 kcal/mol in N2H4. The enthalpy of formation of N2F4 (ΔHf) is 34.421 kJ/mol.[6]
At room temperature N2F4 is mostly associated with only 0.7% in the form of NF2 at 5mm Hg pressure. When the temperature rises to 225 °C, it mostly dissociates with 99% in the form of NF2.[5]
In NF2, the N–F bond length is 1.3494 Å and the angle subtended at FNF is 103.33°.[7]
In the infrared spectrum the N-F bond in NF2 has a symmetrical stretching frequency of 1075 cm−1. This compares to 1115 in NF, 1021 in NF3 and 998 in N2F4.[5]
The microwave spectrum shows numerous lines due to spin transitions, with or without nuclear spin transitions. The lines form set of two triplets for anti-symmetric singlet, or two triplets of triplets for symmetric triplet. Lines appear around 14-15, 24, 25, 26, 27, 28-29, 33, 60, 61, 62, 65 GHz. The rotational constants for the NF2 molecule are A = 70496 MHz, B = 11872.2 MHz, and C = 10136.5 MHz. The inertial defect Δ = 0.1204 mu⋅Å2. The centrifugal distortion constants are τaaaa = −7.75, τbbbb = −0.081, τaabb = 0.30, and τabab = −0.13.[7]
The dipole moment is 0.13 D (4.5×10−31 C⋅m).[7]
The ground electronic state of the molecule is 2B1.[7]
The gas is often contaminated with NO or N2O.[5]
Use
Nitrogen difluoride is formed during the function of a xenon monofluoride excimer laser. Nitrogen trifluoride is the halide carrier gas, which releases fluoride ions when impacted by electrons:[1]
- NF3 + e− → NF2 + F−
The free fluoride ion goes on to react with xenon cations.[1]
Nitrogen difluoride can be consumed further to yield nitrogen monofluoride.
- NF2 + e− → NF + F−[1]
References
- Trainor, Daniel W. (February 1989). "Electron dissociative attachment to nitrogen difluoride radicals". The Journal of Physical Chemistry. 93 (3): 1134–1136. doi:10.1021/j100340a022.
- Jäger, Susanne; von Jouanne, Jörn; Keller-Rudek, Hannelore; Koschel, Dieter; Kuhn, Peter; Merlet, Peter; Rupecht, Sigrid; Vanecek, Hans; Wagner, Joachim (1986). Koschel, Dieter; Kuhn, Peter; Merlet, Peter; Ruprecht, Sigrid; Wagner, Joachim (eds.). F Fluorine: Compounds with Oxygen and Nitrogen. Gmelin Handbook of Inorganic Chemistry. 4. Berlin: Springer. p. 162. doi:10.1007/978-3-662-06339-2. ISBN 978-3-662-06341-5. Retrieved 29 August 2015.
- Johnson, Frederic A.; Colburn, Charles B. (July 1961). "The Tetrafluorohydrazine-Difluoroamino Radical Equilibrium". Journal of the American Chemical Society. 83 (14): 3043–3047. doi:10.1021/ja01475a018.
- Brown, R. D.; Burden, F. R.; Hart, B. T.; Williams, G. R. (1973). "The electronic structure of the NF2 radical". Theoretica Chimica Acta. 28 (4): 339–353. doi:10.1007/BF00529015. S2CID 100649705.
- Bohn, Robert K.; Bauer, Simon Harvey (February 1967). "An electron diffraction study of the structures of NF2 and N2F4". Inorganic Chemistry. 6 (2): 304–309. doi:10.1021/ic50048a024. molecule dimensions and angles
- "Nitrogen difluoride NF2(g)".
- Brown, R.D.; Burden, F.R.; Godfrey, P.D.; Gillard, I.R. (August 1974). "Microwave spectrum of NF2". Journal of Molecular Spectroscopy. 52 (2): 301–321. Bibcode:1974JMoSp..52..301B. doi:10.1016/0022-2852(74)90121-0.
Extra reading
- Goodfriend, P.L.; Woods, H.P. (January 1964). "The absorption spectrum of NF2". Journal of Molecular Spectroscopy. 13 (1–4): 63–66. Bibcode:1964JMoSp..13...63G. doi:10.1016/0022-2852(64)90055-4.
- Jacox, Marilyn E.; Milligan, Dolphus E.; Guillory, William A.; Smith, Jerry J. (August 1974). "Matrix-isolation study of the vacuum-ultraviolet photolysis of NF3". Journal of Molecular Spectroscopy. 52 (2): 322–327. Bibcode:1974JMoSp..52..322J. doi:10.1016/0022-2852(74)90122-2.
- Heidner, R. F.; Helvajian, Henry; Koffend, J. Brooke (August 1987). "Tunable UV laser photolysis of NF2: Quantum yield for NF(a1Δ) production". The Journal of Chemical Physics. 87 (3): 1520–1524. Bibcode:1987JChPh..87.1520H. doi:10.1063/1.453262.
- Papakondylis, Aristotle; Mavridis, Aristides (December 1993). "Electronic and geometrical structure of the NF2 radical" (PDF). Chemical Physics Letters. 216 (1–2): 167–172. Bibcode:1993CPL...216..167P. doi:10.1016/0009-2614(93)E1254-E.
- Cai, Z.-L.; Sha, G.-H.; Zhang, C.-H.; Huang, M.-B. (March 1991). "Ab initio study of low-lying electronic states of the NF2 radical". Chemical Physics Letters. 178 (2–3): 273–278. Bibcode:1991CPL...178..273C. doi:10.1016/0009-2614(91)87068-M.