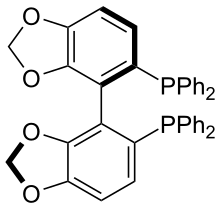
SEGPHOS
SEGPHOS is a chiral ligand developed by Takasago that is used in asymmetric synthesis.[1] It was developed after BINAP and was investigated since it has a narrower dihedral angle between the aromatic faces. This was predicted and then confirmed to increase the enantioselectivity and activity of metal complexes of SEGPHOS. After its commercialization, SEGPHOS and its substituted derivatives have been found to constitute a privileged ligand class for a variety of transition metal catalysts and chemical transformations beyond its original application in ruthenium-catalyzed enantioselective hydrogenation.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
4,4′-Bi-1,3-benzodioxole-5,5′-diylbis(diphenylphosphane) | |
| Other names
SEGPHOS 5,5′-Bis(diphenylphosphino)-4,4′-bi-1,3-benzodioxole | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
PubChem CID |
|
| |
| |
| Properties | |
| C38H28O4P2 | |
| Molar mass | 610.57 g/mol |
| Appearance | colorless solid |
| organic solvents | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
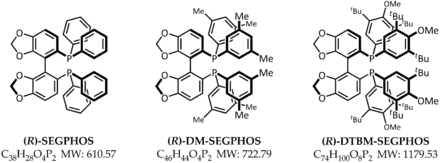
In addition to the parent ligand bearing phenyl groups on the phosphorus atoms, the bulkier derivatives DM-SEGPHOS and DTBM-SEGPHOS are also commercially available.[2] In DM-SEGPHOS and DTBM-SEGPHOS, the phenyl groups of SEGPHOS are replaced by 3,5-dimethylphenyl and 3,5-di-tert-butyl-4-methoxyphenyl groups, respectively.
According to Takasago, as BINAP is likened to a butterfly, SEGPHOS is like a seagull that "flies higher and faster" than a butterfly.[3] Thus the ligand was name SE(A)G(ULL) + PHOS.
References
- Shimizu, H., Nagasaki, I., Matsumura, K., Sayo, N., Saito, T. (2007). "Developments in Asymmetric Hydrogenation from an Industrial Perspective". Acc. Chem. Res. 40 (12): 1385–1393. doi:10.1021/ar700101x. PMID 17685581.CS1 maint: multiple names: authors list (link)
- "SEGPHOS". www.strem.com. Retrieved 2016-08-10.
- "Takasago Received The Molecular Chirality Awards 2002". Takasago International Corporation. Retrieved 2019-08-14.