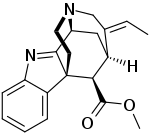
Strictamine
Strictamine is an alkaloid isolated from Alstonia scholaris.[1]
 | |
| Names | |
|---|---|
| IUPAC name
methyl (10S,12R,13E,18S)-13-Ethylidene-8,15-diazapentacyclo[10.5.1.01,9.02,7.010,15]octadeca-2,4,6,8-tetraene-18-carboxylate | |
| Other names
Desacetyldesformoakuammiline; Vincamidine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C20H22N2O2 | |
| Molar mass | 322.408 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Because of its unusual chemical structure, it has attracted research interest and several laboratory syntheses have been reported.[2][3][4][5]
References
- Bhattacharya, S. K.; Bose, R.; Dutta, S. C.; Ray, A. B.; Guha, S. R. (1979). "Neuropharmacological studies on strictamine isolated from Alstonia scholaris". Indian Journal of Experimental Biology. 17 (6): 598–600. PMID 500142.
- Smith, Myles W.; Zhou, Zhiyao; Gao, Alison X.; Shimbayashi, Takuya; Snyder, Scott A. (2017). "A 7-Step Formal Asymmetric Total Synthesis of Strictamine via an Asymmetric Propargylation and Metal-Mediated Cyclization". Organic Letters. 19 (5): 1004–1007. doi:10.1021/acs.orglett.6b03839. PMID 28253628.
- Li, Wenfei; Chen, Zhitao; Yu, Di; Peng, Xin; Wen, Guohua; Wang, Siqi; Xue, Fei; Liu, Xiao-Yu; Qin, Yong (2019). "Asymmetric Total Syntheses of the Akuammiline Alkaloids (−)-Strictamine and (−)-Rhazinoline". Angewandte Chemie International Edition. 58 (18): 6059–6063. doi:10.1002/anie.201901074. PMID 30775833.
- Moreno, Jesus; Picazo, Elias; Morrill, Lucas A.; Smith, Joel M.; Garg, Neil K. (2016). "Enantioselective Total Syntheses of Akuammiline Alkaloids (+)-Strictamine, (−)-2(S)-Cathafoline, and (−)-Aspidophylline A". Journal of the American Chemical Society. 138 (4): 1162–1165. doi:10.1021/jacs.5b12880. PMC 5154302. PMID 26783944.
- Xiao, Tao; Chen, Zhi-Tao; Deng, Lin-Feng; Zhang, Dan; Liu, Xiao-Yu; Song, Hao; Qin, Yong (2017). "Formal total synthesis of the akuammiline alkaloid (+)-strictamine". Chemical Communications. 53 (94): 12665–12667. doi:10.1039/C7CC08153G. PMID 29167841.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.