Allostatic load
Allostatic load is "the wear and tear on the body" which accumulates as an individual is exposed to repeated or chronic stress. The term was coined by Bruce McEwen and Stellar in 1993. It represents the physiological consequences of chronic exposure to fluctuating or heightened neural or neuroendocrine response which results from repeated or prolonged chronic stress.
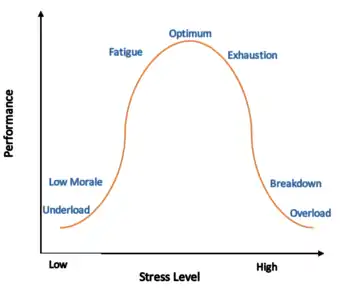
Regulatory model
The term allostatic load is "the wear and tear on the body" which accumulates as an individual is exposed to repeated or chronic stress.[1] It was coined by McEwen and Stellar in 1993.[2]
The term is part of the regulatory model of allostasis, where the predictive regulation or stabilisation of internal sensations in response to stimuli is ascribed to the brain.[3] Allostasis involves the regulation of homeostasis in the body to decrease physiological consequences on the body.[4][5] Predictive regulation refers to the brain's ability to anticipate needs and prepare to fulfill them before they arise.[3]
Part of efficient regulation is the reduction of uncertainty. Humans naturally do not like feeling as if surprise is inevitable. Because of this, we constantly strive to reduce the uncertainty of future outcomes, and allostasis helps us do this by anticipating needs and planning how to satisfy them ahead of time.[6] But it takes a considerable amount of the brain's energy to do this, and if it fails to resolve the uncertainty, the situation may become chronic and result in the accumulation of allostatic load.[6]
The concept of allostatic load provides that "the neuroendocrine, cardiovascular, neuroenergetic, and emotional responses become persistently activated so that blood flow turbulences in the coronary and cerebral arteries, high blood pressure, atherogenesis, cognitive dysfunction and depressed mood accelerate disease progression."[6] All long-standing effects of continuously activated stress responses are referred to as allostatic load. Allostatic load can result in permanently altered brain architecture and systemic pathophysiology.[6]
Allostatic load minimizes an organism's ability to cope with and reduce uncertainty in the future.[6]
Types
McEwen and Wingfield propose two types of allostatic load with different etiologies and distinct consequences:-
Type 1 allostatic load occurs when energy demand exceeds supply, resulting in activation of the emergency life history stage. This serves to direct the animal away from normal life history stages into a survival mode that decreases allostatic load and regains positive energy balance. The normal life cycle can be resumed when the perturbation has passed. Typical situations ending up in type 1 allostasis are starvation, hibernation and critical illness. Of note, the life-threatening consequences of critical illness may be both cause and consequences of allostatic load.[7][8][9]
Type 2 allostatic load results from sufficient or even excess energy consumption being accompanied by social conflict or other types of social dysfunction. The latter is the case in human society and certain situations affecting animals in captivity. In all cases, secretion of glucocorticosteroids and activity of other mediators of allostasis such as the autonomic nervous system, CNS neurotransmitters, and inflammatory cytokines wax and wane with allostatic load. If allostatic load is chronically high, then pathologies may develop. Type 2 allostatic overload does not trigger an escape response, and can only be counteracted through learning and changes in the social structure[10].
Whereas both types of allostatic load are associated with increased release of cortisol and catecholamines, they differentially affect thyroid homeostasis: Concentrations of the thyroid hormone triiodothyronine are decreased in type 1 allostasis, but elevated in type 2 allostasis.[9] This may result from an interaction of type 2 allostatic load with the set point of thyroid function.[11]
Measurement
Allostatic load is generally measured through a composite index of indicators of cumulative strain on several organs and tissues, primarily biomarkers associated with the neuroendocrine, cardiovascular, immune and metabolic systems.[12]
Indices of allostatic load are diverse across studies and are frequently assessed differently, using different biomarkers and different methods of assembling an allostatic load index. Allostatic load is not unique to humans and may be used to evaluate the physiological effects of chronic or frequent stress in non-human primates as well.[12]
In the endocrine system, the increase or repeated levels of stress results in the increased levels of the hormone Corticotropin-Releasing Factor (CRH), which is associated with activation of HPA axis.[5] The Hypothalamic–pituitary–adrenal axis is the central stress response system responsible for modulating inflammatory responses throughout the body. Prolonged stress levels can lead to decreased levels of cortisol in the morning and increased levels in the afternoon, leading to greater daily output of cortisol which in the long term increases blood sugar levels.
In the nervous system, structural and functional abnormalities are a result of chronic prolonged stress. The increase of stress levels causes a shortening of dendrites in a neuron. Therefore, the shortening of dendrites causes the decrease in attention.[5] Chronic stress also causes greater response to fear of the unlearned in the nervous system, and fear conditioning.
In the immune system, the increase in levels of chronic stress results in the elevation of inflammation. The increase in inflammation levels is caused by the ongoing activation of the sympathetic nervous system.[5] The impairment of cell-mediated acquired immunity is also a factor resulting in the immune system due to chronic stress.[5]
Relationship to allostasis and homeostasis
The largest contribution to the allostatic load is the effect of stress on the brain. Allostasis is the system which helps to achieve homeostasis.[13] Homeostasis is the regulation of physiological processes, whereby systems in the body respond to the state of the body and to the external environment.[13] The relationship between allostasis and allostatic load is the concept of anticipation. Anticipation can drive the output of mediators. Examples of mediators include hormones and cortisol. Excess amounts of such mediators will result in an increase in allostatic load, contributing to anxiety and anticipation.[13]
Allostasis and allostatic load are related to the amount of health-promoting and health-damaging behaviours like for example cigarette smoking, consumption of alcohol, poor diet and physical inactivity.[13]
Three physiological processes cause an increase in allostatic load:
- Frequent stress: the magnitude and frequency of response to stress is what determines the level of allostatic load which affects the body.
- Failed shut-down: the inability of the body to shut off while stress accelerates and levels in the body exceed normal levels, for example, elevated blood pressure.
- Inadequate response: the failure of the body systems to respond to challenge, for example, excess levels of inflammation due to inadequate endogenous glucocorticoid responses.
The importance of homeostasis is to regulate the stress levels encountered on the body to reduce allostatic load.
Dysfunctional allostasis causes allostatic load to increase which may, over time, lead to disease, sometimes with decompensation of the allostatically controlled problem. Allostatic load effects can be measured in the body. When tabulated in the form of allostatic load indices using sophisticated analytical methods, it gives an indication of cumulative lifetime effects of all types of stress on the body.[14]
Implications of allostatic load on health
Increased allostatic load constitutes a significant health hazard. Several studies documented a strong association of allostatic load to the incidence of coronary heart disease,[15] to surrogate markers of cardiovascular health[16][17][18][19] and to hard endpoints, including cause-specific and all-cause mortality.[20][21] Mediators connecting allostatic load to morbidity and mortality include the function of the autonomic nervous system,[22] cytokines and stress hormones, e.g. catecholamines,[23][24] cortisol[25][26][27][28] and thyroid hormones.[29]
Reducing risk
To reduce and manage high allostatic load, an individual should pay attention to structural and behavioural factors. Structural factors include the social environment, and access to health services. Behavioural factors include diet, physical health and tobacco smoking, which can lead to chronic disease.[4] Actions such as tobacco smoking are brought about from the stress levels that an individual experiences. Therefore, controlling stress levels from the beginning, for example by not leading to tobacco smoking, will reduce the chance of chronic disease development and high allostatic load.
Low socio-economic status affects allostatic load and therefore, focusing on the causes of low SES will reduce allostatic load levels. Reducing societal polarisation, material deprivation, and psychological demands on health helps to manage allostatic load.[30] Support from the community and the social environment can manage high allostatic load.[30] In addition, healthy lifestyle that encompasses a broad array of lifestyle change including healthy eating and regular physical exercise may reduce allostatic load.[31] Empowering financial help from the government allows people to gain control and improve their psychological health.[30] Improving inequalities in health decreases the stress levels and improves health by reducing high allostatic load on the body.[30]
Interventions can include encouraging sleep quality and quantity, social support, self-esteem and wellbeing, improving diet, avoiding alcohol or drug consumption and participating in physical activity.[32] Providing cleaner and safer environments and the incentive towards a higher education will reduce the chance of stress and improve mental health significantly, therefore, reducing the onset of high allostatic load.[32]
Allostatic load differs by sex and age, and the social status of an individual. Protective factors could, at various times of an individual's life span, be implemented to reduce stress and, in the long run, eliminate the onset of allostatic load.[4] Protective factors include parental bonding, education, social support, healthy workplaces, a sense of meaning towards life and choices being made,[32] and positive feelings in general.[4]
References
- Ogden J (2004). Health Psychology: A textbook (3rd ed.). Open University Press - McGraw-Hill Education. pp. 259. ISBN 978-0335214716.
- McEwen BS, Stellar E (September 1993). "Stress and the individual. Mechanisms leading to disease". Archives of Internal Medicine. 153 (18): 2093–101. doi:10.1001/archinte.153.18.2093. PMID 8379800.
- Sterling P (April 2012). "Allostasis: a model of predictive regulation". Physiology & Behavior. 106 (1): 5–15. doi:10.1016/j.physbeh.2011.06.004. PMID 21684297. S2CID 27164469.
- Schenk HM, Jeronimus BF, van der Krieke L, Bos EH, de Jonge P, Rosmalen JG (2017). "Associations of Positive Affect and Negative Affect With Allostatic Load: A Lifelines Cohort Study". Psychosomatic Medicine. 80 (2): 160–166. doi:10.1097/PSY.0000000000000546. PMID 29215457. S2CID 20121114.
- Danese A, McEwen BS (April 2012). "Adverse childhood experiences, allostasis, allostatic load, and age-related disease". Physiology & Behavior. 106 (1): 29–39. doi:10.1016/j.physbeh.2011.08.019. PMID 21888923. S2CID 3840754.
- Peters A, McEwen BS, Friston K (September 2017). "Uncertainty and stress: Why it causes diseases and how it is mastered by the brain" (PDF). Progress in Neurobiology. 156: 164–188. doi:10.1016/j.pneurobio.2017.05.004. PMID 28576664. S2CID 286501.

- Brame AL, Singer M (October 2010). "Stressing the obvious? An allostatic look at critical illness". Critical Care Medicine. 38 (10 Suppl): S600-7. doi:10.1097/CCM.0b013e3181f23e92. PMID 21164403. S2CID 39889109.
- Cuesta JM, Singer M (December 2012). "The stress response and critical illness: a review". Critical Care Medicine. 40 (12): 3283–9. doi:10.1097/CCM.0b013e31826567eb. PMID 22975887. S2CID 7064946.
- Chatzitomaris A, Hoermann R, Midgley JE, Hering S, Urban A, Dietrich B, et al. (2017). "Thyroid Allostasis-Adaptive Responses of Thyrotropic Feedback Control to Conditions of Strain, Stress, and Developmental Programming". Frontiers in Endocrinology. 8: 163. doi:10.3389/fendo.2017.00163. PMC 5517413. PMID 28775711.
- McEwen BS, Wingfield JC (January 2003). "The concept of allostasis in biology and biomedicine". Hormones and Behavior. 43 (1): 2–15. doi:10.1016/S0018-506X(02)00024-7. PMID 12614627. S2CID 11329342.
- Dietrich JW, Hoermann R, Midgley JE, Bergen F, Müller P (26 October 2020). "The Two Faces of Janus: Why Thyrotropin as a Cardiovascular Risk Factor May Be an Ambiguous Target". Frontiers in Endocrinology. 11: 542710. doi:10.3389/fendo.2020.542710. PMC 7649136. PMID 33193077.
- Edes AN, Crews DE (January 2017). "Allostatic load and biological anthropology". American Journal of Physical Anthropology. 162 Suppl 63: 44–70. doi:10.1002/ajpa.23146. PMID 28105719.
- McEwen BS (May 1998). "Stress, adaptation, and disease. Allostasis and allostatic load". Annals of the New York Academy of Sciences. 840 (1): 33–44. Bibcode:1998NYASA.840...33M. doi:10.1111/j.1749-6632.1998.tb09546.x. PMID 9629234. S2CID 20043016.
- McEwen BS (February 2000). "Allostasis and allostatic load: implications for neuropsychopharmacology". Neuropsychopharmacology. 22 (2): 108–24. doi:10.1016/S0893-133X(99)00129-3. PMID 10649824.
- Gillespie, SL; Anderson, CM; Zhao, S; Tan, Y; Kline, D; Brock, G; Odei, J; O'Brien, E; Sims, M; Lazarus, SA; Hood, DB; Williams, KP; Joseph, JJ (November 2019). "Allostatic load in the association of depressive symptoms with incident coronary heart disease: The Jackson Heart Study". Psychoneuroendocrinology. 109: 104369. doi:10.1016/j.psyneuen.2019.06.020. PMC 7232849. PMID 31307010.
- Veronesi, G; Cavicchiolo, M; Ferrario, MM (December 2019). "Allostatic load as a mediator of the association between psychosocial risk factors and cardiovascular diseases. Recent evidence and indications for prevention". Giornale Italiano di Medicina del Lavoro ed Ergonomia. 41 (4): 333–336. PMID 32126604.
- Viljoen, M; Claassen, N (June 2017). "Allostatic load and heart rate variability as health risk indicators". African Health Sciences. 17 (2): 428–435. doi:10.4314/ahs.v17i2.17. PMC 5637028. PMID 29062338.
- Magnusson Hanson, LL; Rod, NH; Vahtera, J; Virtanen, M; Ferrie, J; Shipley, M; Kivimäki, M; Westerlund, H (August 2020). "Job insecurity and risk of coronary heart disease: Mediation analyses of health behaviors, sleep problems, physiological and psychological factors". Psychoneuroendocrinology. 118: 104706. doi:10.1016/j.psyneuen.2020.104706. PMID 32460194. S2CID 218580825.
- Milad, E; Bogg, T (1 November 2020). "Personality Traits, Coping, Health-related Behaviors, and Cumulative Physiological Health in a National Sample: 10 Year Prospective Effects of Conscientiousness via Perceptions of Activity on Allostatic Load". Annals of Behavioral Medicine. 54 (11): 880–892. doi:10.1093/abm/kaaa024. PMC 7646150. PMID 32359064.
- Borrell, LN; Rodríguez-Álvarez, E; Dallo, FJ (2020). "Racial/ethnic inequities in the associations of allostatic load with all-cause and cardiovascular-specific mortality risk in U.S. adults". PLOS ONE. 15 (2): e0228336. Bibcode:2020PLoSO..1528336B. doi:10.1371/journal.pone.0228336. PMC 7018050. PMID 32053626.
- Robertson, T; Beveridge, G; Bromley, C (2017). "Allostatic load as a predictor of all-cause and cause-specific mortality in the general population: Evidence from the Scottish Health Survey". PLOS ONE. 12 (8): e0183297. Bibcode:2017PLoSO..1283297R. doi:10.1371/journal.pone.0183297. PMC 5559080. PMID 28813505.
- Goldstein, DS (August 2020). "The extended autonomic system, dyshomeostasis, and COVID-19". Clinical Autonomic Research. 30 (4): 299–315. doi:10.1007/s10286-020-00714-0. PMC 7374073. PMID 32700055.
- Brindley, DN; Rolland, Y (November 1989). "Possible connections between stress, diabetes, obesity, hypertension and altered lipoprotein metabolism that may result in atherosclerosis". Clinical Science. 77 (5): 453–61. doi:10.1042/cs0770453. PMID 2684477.
- Rigney, T (September 2010). "Allostatic load and delirium in the hospitalized older adult". Nursing Research. 59 (5): 322–30. doi:10.1097/NNR.0b013e3181ec156b. PMID 20671582. S2CID 205891002.
- Skinner, ML; Shirtcliff, EA; Haggerty, KP; Coe, CL; Catalano, RF (November 2011). "Allostasis model facilitates understanding race differences in the diurnal cortisol rhythm". Development and Psychopathology. 23 (4): 1167–86. doi:10.1017/S095457941100054X. PMC 3583352. PMID 22018088.
- Adam, EK; Heissel, JA; Zeiders, KH; Richeson, JA; Ross, EC; Ehrlich, KB; Levy, DJ; Kemeny, M; Brodish, AB; Malanchuk, O; Peck, SC; Fuller-Rowell, TE; Eccles, JS (December 2015). "Developmental histories of perceived racial discrimination and diurnal cortisol profiles in adulthood: A 20-year prospective study". Psychoneuroendocrinology. 62: 279–91. doi:10.1016/j.psyneuen.2015.08.018. PMC 4739843. PMID 26352481.
- Price, JL; Frazier, IR; Lewis, B; Walker, R; Javors, MA; Nixon, SJ; Adinoff, B (February 2019). "Differences in pituitary-adrenal reactivity in Black and White men with and without alcohol use disorder". Psychoneuroendocrinology. 100: 180–189. doi:10.1016/j.psyneuen.2018.10.004. PMC 6333532. PMID 30347319.
- Casavant, SG; Cong, X; Fitch, RH; Moore, J; Rosenkrantz, T; Starkweather, A (March 2019). "Allostatic Load and Biomarkers of Stress in the Preterm Infant: An Integrative Review". Biological Research for Nursing. 21 (2): 210–223. doi:10.1177/1099800418824415. PMID 30654634. S2CID 58652915.
- Aweimer, A; El-Battrawy, I; Akin, I; Borggrefe, M; Mügge, A; Patsalis, PC; Urban, A; Kummer, M; Vasileva, S; Stachon, A; Hering, S; Dietrich, JW (12 November 2020). "Abnormal thyroid function is common in takotsubo syndrome and depends on two distinct mechanisms: results of a multicentre observational study". Journal of Internal Medicine. doi:10.1111/joim.13189. PMID 33179374.
- Kristenson M, Eriksen HR, Sluiter JK, Starke D, Ursin H (April 2004). "Psychobiological mechanisms of socioeconomic differences in health". Social Science & Medicine. 58 (8): 1511–22. doi:10.1016/s0277-9536(03)00353-8. PMID 14759694.
- Tan M, Mamun A, Kitzman H, Dodgen L (2019-04-18). "Longitudinal Changes in Allostatic Load during a Randomized Church-based, Lifestyle Intervention in African American Women". Ethnicity & Disease. 29 (2): 297–308. doi:10.18865/ed.29.2.297. PMC 6478040. PMID 31057315.
- Juster RP, McEwen BS, Lupien SJ (September 2010). "Allostatic load biomarkers of chronic stress and impact on health and cognition". Neuroscience and Biobehavioral Reviews. 35 (1): 2–16. doi:10.1016/j.neubiorev.2009.10.002. PMID 19822172. S2CID 3562563.
