Chemical defense
Chemical defense is a life history strategy employed by many organisms to avoid consumption by producing toxic or repellent metabolites.[1] The production of defensive chemicals occurs in plants, fungi, and bacteria, as well as invertebrate and vertebrate animals.[2][3] The class of chemicals produced by organisms that are considered defensive may be considered in a strict sense to only apply to those aiding an organism in escaping herbivory or predation.[1] However, the distinction between types of chemical interaction is subjective and defensive chemicals may also be considered to protect against reduced fitness by pests, parasites, and competitors.[4][5][6] Many chemicals used for defensive purposes are secondary metabolites derived from primary metabolites which serve a physiological purpose in the organism.[1] Secondary metabolites produced by plants are consumed and sequestered by a variety of arthropods and, in turn, toxins found in some amphibians, snakes, and even birds can be traced back to arthropod prey.[7][8] There are a variety of special cases for considering mammalian antipredatory adaptations as chemical defenses as well.[9]

Prokaryotes and fungi
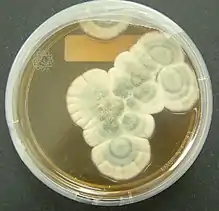
Bacteria of the genera Chromobacterium, Janthinobacterium, and Pseudoalteromonas produce a toxic secondary metabolite, violacein, to deter protozoan predation. Violacein is released when bacteria are consumed, killing the protozoan. Another bacteria, Pseudomonas aeruginosa, aggregates into quorum sensing biofilms which may aid the coordinated release of toxins to protect against predation by protozoans. Flagellates were allowed to grow and were present in a biofilm of P. aeruginosa grown for three days, but no flagellates were detected after seven days. This suggests that concentrated and coordinated release of extracellular toxins by biofilms has a greater effect than unicellular excretions.[10] Bacterial growth is inhibited not only by bacterial toxins, but also by secondary metabolites produced by fungi as well.[3][6] The most well-known of these, first discovered and published by Alexander Fleming in 1929, described the antibacterial properties of a "mould juice" isolated from Penicillium notatum. He named the substance penicillin, and it became the world's first broad-spectrum antibiotic.[3][11] Many fungi are either pathogenic saprophytic, or live within plants without harming them as endophytes, and many of these have been documented to produce chemicals with antagonistic effects against a variety of organisms, including fungi, bacteria, and protozoa.[3] Studies of coprophilous fungi have found antifungal agents which reduce the fitness of competing fungi.[6] In addition, sclerotia of Aspergillus flavus contained a number of previously unknown aflavinines which were much more effective at reducing predation by the fungivorous beetle, Carpophilus hemipterus, than aflatoxins which A. flavus also produced and it has been hypothesized that ergot alkaloids, mycotoxins produced by Claviceps purpurea, may have evolved to discourage herbivory of the host plant.[6]
Plants
A wealth of literature exists on the defensive chemistry of secondary metabolites produced by terrestrial plants and their antagonistic effects on pests and pathogens, likely owing to the fact that human society depends upon large-scale agricultural production to sustain global commerce. Since the 1950s, over 200,000 secondary metabolites have been documented in plants.[12] These compounds serve a variety of physiological and allelochemical purposes, and provide a sufficient stock for the evolution of defensive chemicals. Examples of common secondary metabolites used as chemical defenses by plants include alkaloids, phenols, and terpenes.[13] Defensive chemicals used to avoid consumption may be broadly characterized as either toxins or substances reducing the digestive capacity of herbivores. Although toxins are defined in a broad sense as any substance produced by an organism that reduces the fitness of another, in a more specific sense toxins are substances which directly affect and diminish the functioning of certain metabolic pathways.[14][15] Toxins are minor constituents (<2% dry weight), active in small concentrations, and more present in flowers and young leaves. On the other hand, indigestible compounds make up to 60% dry weight of tissue and are predominately found in mature, woody species.[15] Many alkaloids, pyrethrins, and phenols are toxins. Tannins are major inhibitors of digestion and are polyphenolic compounds with large molecular weights. Lignin and cellulose are important structural elements in plants and are also usually highly indigestible. Tannins are also toxic against pathogenic fungi at natural concentrations in a variety of woody tissues.[1] Not only useful as deterrents to pathogens or consumers, some of the chemicals produced by plants are effective in inhibiting competitors as well. Two separate shrub communities in the California chaparral were found to produce phenolic compounds and volatile terpenes which accumulated in soil and prevented various herbs from growing near the shrubs. Other plants were only observed to grow when fire removed shrubs, but herbs subsequently died off after shrubs returned.[5] Although the focus has been on broad-scale patterns in terrestrial plants, Paul and Fenical in 1986 demonstrated a variety of secondary metabolites in marine algae which prevented feeding or induced mortality in bacteria, fungi, echinoderms, fishes, and gastropods.[16] In nature, pests are a severe problem to plant communities as well, leading to the co-evolution of plant chemical defenses and herbivore metabolic strategies to detoxify their plant food.[17] A variety of invertebrates consume plants, but insects have received a majority of the attention. Insects are pervasive agricultural pests and sometimes occur in such high densities that they can strip fields of crops.[18]
Animals
Invertebrates
Many insects are distasteful to predators and excrete irritants or secrete poisonous compounds that cause illness or death when ingested. Secondary metabolites obtained from plant food may also be sequestered by insects and used in the production of their own toxins.[17][19] One of the more well-known examples of this is the monarch butterfly, which sequesters poison obtained from the milkweed plant. Among the most successful insect orders employing this strategy are beetles (Coleoptera), grasshoppers (Orthoptera), and moths and butterflies (Lepidoptera).[20][21] Insects also biosynthesize unique toxins, and while sequestration of toxins from food sources is claimed to be the energetically favorable strategy, this has been contested.[17][22] Passion-vine associated butterflies in the tribe Heliconiini (sub-family Heliconiinae) either sequester or synthesize de novo defensive chemicals, but moths in the genus Zygaena (family Zygaenidae) have evolved the ability to either synthesize or sequester their defensive chemicals through convergence.[17] Some coleopterans sequester secondary metabolites to be used as defensive chemicals but most biosynthesize their own de novo. Anatomical structures have developed to store these substances, and some are circulated in the hemolyph and released associated with a behavior called reflex bleeding.[19]
Vertebrates

Vertebrates can also biosynthesize defensive chemicals or sequester them from plants or prey.[8][22] Sequestered compounds have been observed in frogs, natricine snakes, and two genera of birds, Pitohui and Ifrita.[8] It is suspected that some well-known compounds such as tetrodotoxin produced by newts and pufferfish[23] are derived from invertebrate prey. Bufadienolides, defensive chemicals produced by toads, have been found in glands of natricine snakes used for defense.[8]
Amphibians
Frogs acquire the toxins needed for chemical defense by either producing them through glands on their skin or through their diet.[24] The source of toxins in their diet are primarily arthropods, ranging from beetles to millipedes.[25] When the required dietary components are absent, such as in captivity, the frog is no longer able to produce the toxins, making them nonpoisonous.[26] The profile of toxins may even change with the season, as is the case for the Climbing Mantella, whose diet and feeding behavior differ between wet and dry seasons[27]
The evolutionary advantage of producing such toxins is the deterrence of predators. There is evidence to suggest that the ability to produce toxins evolved along with aposematic coloration, acting as a visual cue to predators to remember which species are not palatable.[24]
While the toxins produced by frogs are frequently referred to as poisonous, the doses of toxins are low enough that they are more noxious than poisonous.[25] However, components of the toxins, namely the alkaloids, are very active in ion channels.[25] Therefore, they disrupt the victim's nervous system, making them much more effective. Within the frogs themselves, the toxins are accumulated and delivered through small, specialized transport proteins.[28]
.jpg.webp)
Besides providing defense from predators, the toxins that poison frogs secrete interest medical researchers. Poison dart frogs, of the Dendrobatidae family, secrete batrachotoxin.[26] This toxin has the potential to act as a muscle relaxant, heart stimulant, or anesthetic.[26] Multiple species of frogs secrete epibatidine, whose study has yielded several important results.[29] It was discovered that the frogs resist poisoning themselves through a single amino acid replacement that desensitizes the targeted receptors to the toxin, but still maintains the function of the receptor.[29] This finding gives insight to the roles of proteins, the nervous system, and the mechanics of chemical defense, all of which promote future biomedical research and innovation.
Mammals
Some mammals can emit foul smelling liquids from anal glands, such as the pangolin[30] and some members of families Mephitidae and Mustelidae including skunks, weasels, and polecats.[31] Monotremes have venomous spurs used to avoid predation[32] and slow lorises (Primates: Nycticebus) produce venom which appears to be effective at deterring both predators and parasites.[33] It has also been demonstrated that physical contact with a slow loris (without being bitten) can cause a reaction in humans – acting as a contact poison.[34]
See also
References
- Berenbaum MR (January 1995). "The chemistry of defense: theory and practice". Proceedings of the National Academy of Sciences of the United States of America. 92 (1): 2–8. Bibcode:1995PNAS...92....2B. doi:10.1073/pnas.92.1.2. PMC 42807. PMID 7816816.
- Clucas B (2010). "Defensive Chemicals". In Breed MD, Moore J (eds.). Encyclopedia of Animal Behavior. Oxford: Academic Press. pp. 481–486. doi:10.1016/B978-0-08-045337-8.00293-X. ISBN 9780080453378.
- Keller NP, Turner G, Bennett JW (December 2005). "Fungal secondary metabolism - from biochemistry to genomics". Nature Reviews. Microbiology. 3 (12): 937–47. doi:10.1038/nrmicro1286. PMID 16322742. S2CID 23537608.
- Walters D (2011). Plant defense: warding off attack by pathogens, herbivores and parasitic plants. John Wiley & Sons.
- Whittaker RH, Feeny PP (February 1971). "Allelochemics: chemical interactions between species". Science. New York, N.Y. 171 (3973): 757–70. Bibcode:1971Sci...171..757W. doi:10.1126/science.171.3973.757. JSTOR 1730763. PMID 5541160.
- Gloer JB (1995). "The chemistry of fungal antagonism and defense". Canadian Journal of Botany. 73 (S1): 1265–1274. doi:10.1139/b95-387.
- Lasley EN (1999). "Having Their Toxins and Eating Them Too: Study of the natural sources of many animals' chemical defenses is providing new insights into nature's medicine chest". BioScience. 49 (12): 945–950. doi:10.1525/bisi.1999.49.12.945.
- Savitzky AH, Mori A, Hutchinson DA, Saporito RA, Burghardt GM, Lillywhite HB, Meinwald J (September 2012). "Sequestered defensive toxins in tetrapod vertebrates: principles, patterns, and prospects for future studies". Chemoecology. 22 (3): 141–158. doi:10.1007/s00049-012-0112-z. PMC 3418492. PMID 22904605.
- Hettyey A, Üveges B, Móricz ÁM, Drahos L, Capon RJ, Van Buskirk J, et al. (December 2019). "Predator-induced changes in the chemical defence of a vertebrate". The Journal of Animal Ecology. 88 (12): 1925–1935. doi:10.1111/1365-2656.13083. PMID 31408536.
- Matz C, Kjelleberg S (July 2005). "Off the hook--how bacteria survive protozoan grazing". Trends in Microbiology. 13 (7): 302–7. doi:10.1016/j.tim.2005.05.009. PMID 15935676.
- Fleming A (1929). "On the antibacterial action of cultures of a penicillium, with special reference to their use in the isolation of B. influenzae". British Journal of Experimental Pathology. 10 (3): 226–236. PMC 2048009.
- Hartmann T (2007). "From waste products to ecochemicals: fifty years research of plant secondary metabolism". Phytochemistry. 68 (22–24): 2831–46. doi:10.1016/j.phytochem.2007.09.017. PMID 17980895.
- Levin DA (1976). "The chemical defenses of plants to pathogens and herbivores". Annual Review of Ecology and Systematics. 7 (1): 121–159. doi:10.1146/annurev.es.07.110176.001005.
- Wittstock U, Gershenzon J (August 2002). "Constitutive plant toxins and their role in defense against herbivores and pathogens". Current Opinion in Plant Biology. 5 (4): 300–7. doi:10.1016/S1369-5266(02)00264-9. PMID 12179963.
- Cates RG, Rhoades DF (1977). "Patterns in the production of antiherbivore chemical defenses in plant communities". Biochemical Systematics and Ecology. 5 (3): 185–193. doi:10.1016/0305-1978(77)90003-5.
- Paul VJ, Fenical W (1986). "Chemical defense in tropical green algae, order Caulerpales". Marine Ecology Progress Series. 34: 157–169. Bibcode:1986MEPS...34..157P. doi:10.3354/meps034157.
- Fürstenberg-Hägg J, Zagrobelny M, Jørgensen K, Vogel H, Møller BL, Bak S (2014). "Chemical defense balanced by sequestration and de novo biosynthesis in a lepidopteran specialist". PLOS ONE. 9 (10): e108745. Bibcode:2014PLoSO...9j8745F. doi:10.1371/journal.pone.0108745. PMC 4191964. PMID 25299618.
- Lomer CJ, Bateman RP, Johnson DL, Langewald J, Thomas M (2001). "Biological control of locusts and grasshoppers". Annual Review of Entomology. 46 (1): 667–702. doi:10.1146/annurev.ento.46.1.667. PMID 11112183.
- Dettner K (1987). "Chemosystematics and evolution of beetle chemical defenses". Annual Review of Entomology. 32 (1): 17–48. doi:10.1146/annurev.en.32.010187.000313.
- Schmidt JO (2008). "Venoms and toxins in insects.". Encyclopedia of entomology. Netherlands: Springer. pp. 4076–4089. doi:10.1007/978-1-4020-6359-6_3957. ISBN 978-1-4020-6359-6.
- Trigo JR (2000). "The chemistry of antipredator defense by secondary compounds in neotropical Lepidoptera: facts, perspectives and caveats". Journal of the Brazilian Chemical Society. 11 (6): 551–561. doi:10.1590/S0103-50532000000600002.
- Mebs D (January 2001). "Toxicity in animals. Trends in evolution?". Toxicon. 39 (1): 87–96. doi:10.1016/S0041-0101(00)00155-0. PMID 10936625.
- Fuhrman FA (December 1986). "Tetrodotoxin, tarichatoxin, and chiriquitoxin: historical perspectives". Annals of the New York Academy of Sciences. 479 (1 Tetrodotoxin): 1–14. Bibcode:1986NYASA.479....1F. doi:10.1111/j.1749-6632.1986.tb15556.x. PMID 3468842. S2CID 44741246.
- Summers K, Clough ME (May 2001). "The evolution of coloration and toxicity in the poison frog family (Dendrobatidae)". Proceedings of the National Academy of Sciences of the United States of America. 98 (11): 6227–32. Bibcode:2001PNAS...98.6227S. doi:10.1073/pnas.101134898. PMC 33450. PMID 11353830.
- Daly J (1995). "The Chemistry of Poisons in Amphibian Skin". Chemical Ecology: The Chemistry of Biotic Interaction. Washington, D.C.: The National Academies Press. pp. 17–26. ISBN 9780309052818.
- "Poison frogs". Smithsonian's National Zoo. 2016-06-06. Retrieved 2020-12-01.
- Moskowitz NA, Roland AB, Fischer EK, Ranaivorazo N, Vidoudez C, Aguilar MT, et al. (2018-12-26). Chaves AV (ed.). "Seasonal changes in diet and chemical defense in the Climbing Mantella frog (Mantella laevigata)". PLOS ONE. 13 (12): e0207940. Bibcode:2018PLoSO..1307940M. doi:10.1371/journal.pone.0207940. PMC 6306172. PMID 30586404.
- Caty SN, Alvarez-Buylla A, Byrd GD, Vidoudez C, Roland AB, Tapia EE, et al. (June 2019). "Molecular physiology of chemical defenses in a poison frog". The Journal of Experimental Biology. 222 (Pt 12): jeb204149. doi:10.1242/jeb.204149. PMID 31138640. S2CID 169034903.
- Tarvin RD, Borghese CM, Sachs W, Santos JC, Lu Y, O'Connell LA, et al. (September 2017). "Interacting amino acid replacements allow poison frogs to evolve epibatidine resistance". Science. 357 (6357): 1261–1266. Bibcode:2017Sci...357.1261T. doi:10.1126/science.aan5061. PMC 5834227. PMID 28935799.
- "Pholidota (pangolins)".
- Andersen KK, Bernstein DT (1980). "Sulfur compounds in mustelids.". Natural Sulfur Compounds. Boston, MA.: Springer. pp. 399–406. doi:10.1007/978-1-4613-3045-5_35. ISBN 978-1-4613-3047-9.
- Hurum JH, Luo ZX, Kielan-Jaworowska Z (2006). "Were mammals originally venomous?" (PDF). Acta Palaeontologica Polonica. 51 (1): 1–11.
- Nekaris KA, Moore RS, Rode EJ, Fry BG (September 2013). "Mad, bad and dangerous to know: the biochemistry, ecology and evolution of slow loris venom". The Journal of Venomous Animals and Toxins Including Tropical Diseases. 19 (1): 21. doi:10.1186/1678-9199-19-21. PMC 3852360. PMID 24074353.
- Gardiner M, Weldon A, Poindexter SA, Gibson N, Nekaris KA (2018). "Survey of practitioners handling slow lorises (Primates: Nycticebus): an assessment of the harmful effects of slow loris bites". Journal of Venom Research. 9: 1–7. PMC 6055083. PMID 30090322.