Dimethylamine
Dimethylamine is an organic compound with the formula (CH3)2NH. This secondary amine is a colorless, flammable gas with an ammonia-like odor. Dimethylamine is commonly encountered commercially as a solution in water at concentrations up to around 40%. An estimated 270,000 tons were produced in 2005.[5]
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
N-Methylmethanamine | |
| Other names
(Dimethyl)amine | |
| Identifiers | |
3D model (JSmol) |
|
| 3DMet | |
| 605257 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.272 |
| EC Number |
|
| 849 | |
| KEGG | |
| MeSH | dimethylamine |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1032 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties[1][2] | |
| (CH3)2NH | |
| Molar mass | 45.085 g·mol−1 |
| Appearance | Colorless gas |
| Odor | Fishy, ammoniacal |
| Density | 649.6 kg m−3 (at 25 °C) |
| Melting point | −93.00 °C; −135.40 °F; 180.15 K |
| Boiling point | 7 to 9 °C; 44 to 48 °F; 280 to 282 K |
| 1.540 kg L−1 | |
| log P | −0.362 |
| Vapor pressure | 170.3 kPa (at 20 °C) |
Henry's law constant (kH) |
310 μmol Pa−1 kg−1 |
| Basicity (pKb) | 3.29 |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−21 to −17 kJ mol−1 |
| Hazards | |
| GHS pictograms |  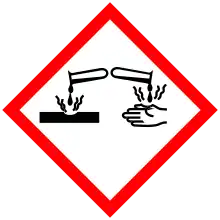  |
| GHS Signal word | Danger |
| H220, H302, H315, H318, H332, H335 | |
| P210, P261, P280, P305+351+338 | |
| NFPA 704 (fire diamond) | |
| Flash point | −6 °C (21 °F; 267 K) (liquid) |
| 401 °C (754 °F; 674 K) | |
| Explosive limits | 2.8–14.4% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
698 mg/kg (rat, oral) 316 mg/kg (mouse, oral) 240 mg/kg (rabbit, oral) 240 mg/kg (guinea pig, oral)[3] |
LC50 (median concentration) |
4700 ppm (rat, 4 hr) 4540 ppm (rat, 6 hr) 7650 ppm (mouse, 2 hr)[3] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 10 ppm (18 mg/m3)[4] |
REL (Recommended) |
TWA 10 ppm (18 mg/m3)[4] |
IDLH (Immediate danger) |
500 ppm[4] |
| Related compounds | |
Related amines |
|
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure and synthesis
The molecule consists of a nitrogen atom with two methyl substituents and one proton. Dimethylamine is a weak base and the pKa of the ammonium CH3-NH+
2-CH3 is 10.73, a value above methylamine (10.64) and trimethylamine (9.79).
Dimethylamine reacts with acids to form salts, such as dimethylamine hydrochloride, an odorless white solid with a melting point of 171.5 °C. Dimethylamine is produced by catalytic reaction of methanol and ammonia at elevated temperatures and high pressure:[6]
- 2 CH3OH + NH3 → (CH3)2NH + 2 H2O
Natural occurrence
Dimethylamine is found quite widely distributed in animals and plants, and is present in many foods at the level of a few mg/kg.[7]
Uses
Dimethylamine is a precursor to several industrially significant compounds.[5][8] It reacts with carbon disulfide to give dimethyl dithiocarbamate, a precursor to zinc bis(dimethyldithiocarbamate) and other chemicals used in the sulfur vulcanization of rubber. The solvents dimethylformamide and dimethylacetamide are derived from dimethylamine. It is raw material for the production of many agrichemicals and pharmaceuticals, such as dimefox and diphenhydramine, respectively. The chemical weapon tabun is derived from dimethylamine. The surfactant lauryl dimethylamine oxide is found in soaps and cleaning compounds. Unsymmetrical dimethylhydrazine, a rocket fuel, is prepared from dimethylamine.
Occurrence and reactions
It is an attractant for boll weevils.[9]
Dimethylamine undergoes nitrosation to give dimethylnitrosamine, a carcinogen.
Deprotonation of dimethylamine can be effected with organolithium compounds. The resulting LiNMe2, which adopts a cluster-like structure, serves as a source of "Me2N−". This lithium amide has been used to prepare volatile metal complexes such as tetrakis(dimethylamido)titanium and pentakis(dimethylamido)tantalum.
The Lewis base properties of dimethylamine are examined in the ECW model. Its relative donor strength toward a series of acids, versus other Lewis bases, can be illustrated by C-B plots.[10][11]
Safety
Dimethylamine is not very toxic with the following LD50 values: 736 mg/kg (mouse, i.p.); 316 mg/kg (mouse, p.o.); 698 mg/kg (rat, p.o.); 3900 mg/kg (rat, dermal); 240 mg/kg (guinea pig or rabbit, p.o.).[12]
See also
References
- "Dimethylamine". NIST Chemistry WebBook. USA: National Institute of Standards and Technology. Retrieved 21 October 2011.
- "Dimethylamine 38931 - ≥99.0%". Aldrich. Sigma-Aldrich Co. Retrieved 21 October 2011.
- "Dimethylamine". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- NIOSH Pocket Guide to Chemical Hazards. "#0219". National Institute for Occupational Safety and Health (NIOSH).
- Van Gysel, August B.; Musin, Willy (2000). "Methylamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a16_535.
- Corbin D.R.; Schwarz S.; Sonnichsen G.C. (1997). "Methylamines synthesis: A review". Catalysis Today. 37 (2): 71–102. doi:10.1016/S0920-5861(97)00003-5.
- Neurath, G. B.; et al. (1977). "Primary and secondary amines in the human environment". Food and Cosmetics Toxicology. 15 (4): 275–282. doi:10.1016/s0015-6264(77)80197-1.
- Ashford's Dictionary of Industrial Chemicals, 3rd edition, 2011, pages 3284-3286
- The Merck Index, 10th Ed. (1983), p.470, Rahway: Merck & Co.
- Laurence, C. and Gal, J-F. Lewis Basicity and Affinity Scales, Data and Measurement, (Wiley 2010) pp 50-51 IBSN 978-0-470-74957-9
- Cramer, R. E.; Bopp, T. T. (1977). "Graphical display of the enthalpies of adduct formation for Lewis acids and bases". Journal of Chemical Education. 54: 612–613. doi:10.1021/ed054p612. The plots shown in this paper used older parameters. Improved E&C parameters are listed in ECW model.
- Chemical Information Profile for Dimethylamine Borane, National Toxicology Program, NIEHS, NIH (2008), p.4: http://ntp.niehs.nih.gov/ntp/htdocs/Chem_Background/ExSumPdf/DimethylamineBorane508.pdf
External links
- International Chemical Safety Card 0260 (gas)
- International Chemical Safety Card 1485 (aqueous solution)
- NIOSH Pocket Guide to Chemical Hazards. "#0219". National Institute for Occupational Safety and Health (NIOSH).
- Properties from Air Liquide
- MSDS at airliquide.com
