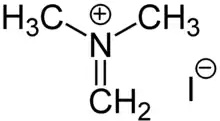
Eschenmoser's salt
Eschenmoser's salt, dimethyl(methylidene)ammonium iodide, is a strong dimethylaminomethylating agent, used to prepare derivatives of the type RCH
2N(CH
3)
2.[1][2] Enolates, enolsilylethers, and even more acidic ketones undergo efficient dimethylaminomethylation. Once prepared, such tertiary amines can be further methylated and then subjected to base-induced elimination to afford methylidenated ketones. The salt was first prepared by the group of Albert Eschenmoser[3] after whom the reagent is named.
 | |
| Names | |
|---|---|
| IUPAC name
Dimethyl(methylidene)ammonium iodide | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.046.968 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H8NI | |
| Molar mass | 185.01 g/mol |
| Appearance | colorless hygroscopic crystals |
| Melting point | 116 °C (241 °F; 389 K) |
| decomposes | |
| Hazards | |
| R-phrases (outdated) | R36/37/38 |
| S-phrases (outdated) | S26 S36 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Analogous salts, dimethyl(methylidene)ammonium chloride (Böhme's salt[1], after Horst Böhme) and trifluoroacetate, have similar properties and applications.[1]
See also
- Vilsmeier reagent, [(CH
3)
2NCHCl]Cl.
References
- E. F. Kleinman in "Dimethylmethyleneammonium Iodide and Chloride" in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289X.rd346
- Horst Böhme; Eberhard Mundlos; Otto-Erich Herboth (1957). "Über Darstellung und Eigenschaften α-Halogenierter Amine". Chemische Berichte. 90 (9): 2003–2008. doi:10.1002/cber.19570900942.
- Jakob Schreiber; Hans Maag; Naoto Hashimoto; Albert Eschenmoser (1971). "Dimethyl(methylene)ammonium Iodide". Angewandte Chemie International Edition in English. 10 (5): 330–331. doi:10.1002/anie.197103301.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.