Fat hydrogenation
Fat hydrogenation is the process of combining fat — typically, vegetable oils — with hydrogen, in order to make it more saturated.
The process is typically carried out at very high pressure, with the help of a nickel catalyst (that is removed from the final product).
Their partial hydrogenation reduces most, but not all, of these carbon-carbon double bonds. The degree of hydrogenation is controlled by restricting the amount of hydrogen, reaction temperature and time, and the catalyst.[1] The goal is to turn liquid oils into solid or semi-solid fats that can replace butter and shortening in spreads, candies, baked good, and other products.
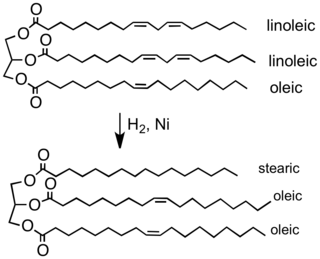 Partial hydrogenation of a typical plant oil to a typical component of margarine. Most of the C=C double bonds are removed in this process, which elevates the melting point of the product.
Partial hydrogenation of a typical plant oil to a typical component of margarine. Most of the C=C double bonds are removed in this process, which elevates the melting point of the product.
Hydrogenation converts liquid vegetable oils into solid or semi-solid fats, such as those present in margarine. Changing the degree of saturation of the fat changes some important physical properties, such as the melting range, which is why liquid oils become semi-solid. Solid or semi-solid fats are preferred for baking because the way the fat mixes with flour produces a more desirable texture in the baked product. Because partially hydrogenated vegetable oils are cheaper than animal fats, they are available in a wide range of consistencies, and have other desirable characteristics (such as increased oxidative stability and longer shelf life), they are the predominant fats used as shortening in most commercial baked goods.
Full hydrogenation results in a molecule containing the maximum amount of hydrogen (in other words, the conversion of an unsaturated fatty acid into a saturated one). Partial hydrogenation results in the addition of hydrogen atoms at some of the empty positions, with a corresponding reduction in the number of double bonds. Typical commercial hydrogenation is partial to obtain a malleable mixture of fats that is solid at room temperature, but melts during baking, or consumption.
The "trans fat problem"
A side effect of incomplete hydrogenation having implications for human health is the isomerization of some of the remaining unsaturated carbon bonds to their trans isomers. Trans fats (resulting from partial hydrogenation) have been implicated in circulatory diseases including heart disease.[2] The conversion from cis to trans bonds is chemically favored because the trans configuration has lower energy than the natural cis one. At equilibrium, the trans/cis isomer ratio is about 2:1. Many countries and regions have introduced mandatory labeling of trans fats on food products and appealed to the industry for voluntary reductions.[3][4][5] The food industry has moved away from partially hydrogenated fats (i.e. trans fats) and towards fully hydrogenated fats and interesterified fats in response to bad publicity about trans fats, labeling requirements, and removal of trans fats from the FDA list of foods Generally Recognized as Safe.[6][7][8]
Catalytic partial hydrogenation necessarily produces trans-fats, because of the reaction mechanism. In the first reaction step, one hydrogen is added, with the other, coordinatively unsaturated, carbon being attached to the catalyst. The second step is the addition of hydrogen to the remaining carbon, producing a saturated fatty acid. The first step is reversible, such that the hydrogen is readsorbed on the catalyst and the double bond is re-formed. The intermediate with only one hydrogen added contains no double bond and can freely rotate. Thus, the double bond can re-form as either cis or trans, of which trans is favored, regardless the starting material. Complete hydrogenation does not have this side effect, because it also hydrogenates any produced trans fats to give saturated fats.
Numerous studies have concluded that these trans fatty acids have negative health effects. As a result, many countries have enacted trans fat regulation that aims to eliminate or severely restrict their amount in industrialized food products.
History

Nobel laureate Paul Sabatier worked in the late 1890s to develop the chemistry of hydrogenation, which enabled the margarine, oil hydrogenation, and synthetic methanol industries.[9] Whereas Sabatier considered hydrogenation of only vapors, the German chemist Wilhelm Normann showed in 1901 that liquid oils could be hydrogenated, and patented the process in 1902.[10][11][12] During the years 1905–1910, Normann built a fat-hardening facility in the Herford company. At the same time, the invention was extended to a large-scale plant in Warrington, England, at Joseph Crosfield & Sons, Limited. It took only two years until the hardened fat could be successfully produced in the plant in Warrington, commencing production in the autumn of 1909. The initial year's production totalled nearly 3,000 tonnes.[13] In 1909, Procter & Gamble acquired the United States rights to the Normann patent;[14] in 1911, they began marketing the first hydrogenated shortening, Crisco (composed largely of partially hydrogenated cottonseed oil). Further success came from the marketing technique of giving away free cookbooks in which every recipe called for Crisco.

Normann's hydrogenation process made it possible to stabilize affordable whale oil or fish oil for human consumption, a practice kept secret to avoid consumer distaste.[13]
During Napoleon's reign in France in the early 19th century, a type of margarine was invented to feed the troops using tallow and buttermilk. It was not accepted in the United States. In the early 20th century, soybeans began to be imported into the United States as a source of protein; soybean oil was a by-product. What to do with that oil became an issue. At the same time, there was not enough butterfat available for consumers. The method of hydrogenating fat and turning a liquid fat into a solid one had been discovered, and now the ingredients (soybeans) and the need (shortage of butter) were there. Later, the means for storage, the refrigerator, was a factor in trans fat development. The fat industry found that hydrogenated fats provided some special features to margarines, which allowed margarine, unlike butter, to be taken out of a refrigerator and immediately spread on bread. By some minor changes to the chemical composition of hydrogenated fat, such hydrogenated fat was found to provide superior baking properties compared to lard. Margarine made from hydrogenated soybean oil began to replace butterfat. Hydrogenated fat such as Crisco and Spry, sold in England, began to replace butter and lard in baking bread, pies, cookies, and cakes in 1920.[15]
Production of hydrogenated fats increased steadily until the 1960s, as processed vegetable fats replaced animal fats in the United States and other Western countries. At first, the argument was a financial one due to lower costs; advocates also said that the unsaturated trans fats of margarine were healthier than the saturated fats of butter.[16]
See also
References
- Ian P. Freeman "Margarines and Shortenings" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a16_145
- Teresa Tarrago-Trani, Maria; Phillips, Katherine M.; Lemar, Linda E.; Holden, Joanne M. (2006). "New and Existing Oils and Fats Used in Products with Reduced Trans-Fatty Acid Content" (PDF). Journal of the American Dietetic Association. 106 (6): 867–880. doi:10.1016/j.jada.2006.03.010. PMID 16720128.
- "Deadly fats: why are we still eating them?". The Independent. 2008-06-10. Archived from the original on 2008-06-14. Retrieved 2008-06-16.
- "New York City passes trans fat ban". NBC News. 2006-12-05. Retrieved 2010-01-09.
- "F.D.A. Gives Food Industry 3 Years to Eliminate Trans Fats". The New York Times. 2015-06-16. Archived from the original on 2015-06-16. Retrieved 2015-06-16.
- "Ask the Experts: Hydrogenated Oils". Berkeley Wellness. Remedy Health Media. October 1, 2011. Retrieved 24 November 2018.
- Collette, Robert L. (July 7, 2015). "Why a Monograph for Fully Hydrogenated Oils and Fats is Needed". Quality Matters. United States Pharmacopeial Convention. Retrieved 24 November 2018.
- Haynes, Fiona (September 23, 2018). "Partially vs. Fully Hydrogenated Oils: The Truth About Trans Fats". The Spruce Eats. Dotdash. Retrieved 24 November 2018.
- Nobel Lectures, Chemistry, 1901–1921. Elsevier. 1966. Reprinted online: "Paul Sabatier, The Nobel Prize in Chemistry 1912". Nobel Foundation. Retrieved 7 January 2007.
- de 141029 Process for converting unsaturated fatty acids or their glycerides into saturated compounds
- gb 190301515 Process for converting unsaturated fatty acids or their glycerides into saturated compounds
- Patterson HB (1998). "Hydrogenation" (PDF). Sci Lecture Papers Series. Archived from the original (PDF) on 26 September 2007. Retrieved 7 January 2007.
- "Wilhelm Normann und die Geschichte der Fetthärtung von Martin Fiedler, 2001". 20 December 2011. Archived from the original on 1 October 2011. Retrieved 14 August 2007.
- Shurtleff W, Aoyagi A. "History of Soybeans and Soyfoods: 1100 B.C. to the 1980s". Archived from the original on 18 October 2005.
- Kummerow FA (2008). Cholesterol Won't Kill You – But Trans Fat Could. Trafford Publishing. ISBN 978-1-4251-3808-0. NOTE: Unreliable source.
- Ascherio A, Stampfer MJ, Willett WC. "Trans fatty acids and coronary heart disease". Archived from the original on 3 September 2006. Retrieved 14 September 2006.