Lyngbyatoxin-a
Lyngbyatoxin-a is a cyanotoxin produced by certain cyanobacteria species, most notably Moorea producens (formerly Lyngbya majuscula). It is produced as defense mechanism to ward off any would-be predators of the bacterium, being a potent blister agent as well as carcinogen. Low concentrations cause a common skin condition known as seaweed dermatitis.[1][2][3][4][5][6]
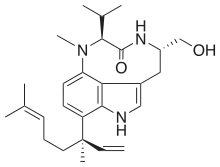 | |
| Names | |
|---|---|
| IUPAC name
(4S,7S)-11-((R)-3,7-dimethylocta-1,6-dien-3-yl)-4-(hydroxymethyl)-7-isopropyl-8-methyl-4,5,7,8-tetrahydro-1H-[1,4]diazonino[7,6,5-cd]indol-6(3H)-one | |
| Other names
Lyngbyatoxin-a | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H39N3O2 | |
| Molar mass | 437.628 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Biosynthesis

Lyngbyatoxin is a terpenoid indole alkaloid that belongs to the class of non-ribosomal peptides (NRP).[7] Lyngbyatoxin contains a nucleophilic indole ring that takes part in the activation of protein kinases. Figure 1, shows the biosynthesis of Lyngbyatoxin reported by Neilan et al. and Gerwick et al.The non-ribosomal peptide synthase (NRPS) LtxA protein condenses L-methyl-valine and L-tryptophan to form the linear dipeptide N-methyl-L-valyl-L-tryptophan. The latter is released via a NADPH-dependent reductive cleavage to form the aldehyde which is subsequently reduced to the corresponding alcohol. Then LtxB which is a P450-dependent monooxygenase serves as a catalyst in the oxidation and subsequent cyclization of N-methyl-L-valyl-L-tryptophan.Finally, LtxC which is a reverse prenyltransferase performs the transfer of a geranyl pyrophosphate (GPP) to carbon-7 of the indole ring which is accompanied by the lost of pyrophosphate.
References
- Cardellina, JH; Marner, FJ; Moore, RE (Apr 1979). "Seaweed dermatitis: structure of lyngbyatoxin A.". Science. 204 (4389): 193–5. doi:10.1126/science.107586. PMID 107586.
- Fujiki, H; Mori, M; Nakayasu, M; Terada, M; Sugimura, T; Moore, RE (Jun 1981). "Indole alkaloids: dihydroteleocidin B, teleocidin, and lyngbyatoxin A as members of a new class of tumor promoters". Proceedings of the National Academy of Sciences USA. 78 (6): 3872–6. doi:10.1073/pnas.78.6.3872. PMC 319675. PMID 6791164.
- Kozikowski, AP; Shum, PW; Basu, A; Lazo, JS (Aug 1991). "Synthesis of structural analogues of lyngbyatoxin A and their evaluation as activators of protein kinase C.". Journal of Medicinal Chemistry. 34 (8): 2420–30. doi:10.1021/jm00112a017. PMID 1875340.
- Osborne, NJ; Webb, PM; Shaw, GR (Nov 2001). "The toxins of Lyngbya majuscula and their human and ecological health effects". Environment International. 27 (5): 381–92. doi:10.1016/s0160-4120(01)00098-8. PMID 11757852.
- Ito, E; Satake, M; Yasumoto, T (May 2002). "Pathological effects of lyngbyatoxin A upon mice". Toxicon. 40 (5): 551–6. doi:10.1016/s0041-0101(01)00251-3. PMID 11821127.
- Edwards, DJ; Gerwick, WH (2004). "Lyngbyatoxin biosynthesis: sequence of biosynthetic gene cluster and identification of a novel aromatic prenyltransferase". Journal of the American Chemical Society. 126 (37): 11432–3. doi:10.1021/ja047876g. PMID 15366877.
- Ongley, SE; Bian, X; Zhang, Y; Chau, R; Gerwick, WH; Müller, R; Neilan, BA (2013). "High-Titer Heterologous Production in E. coli of Lyngbyatoxin, a Protein Kinase C Activator from an Uncultured Marine Cyanobacterium". ACS Chemical Biology. 8: 1888–1893. doi:10.1021/cb400189j. PMC 3880125. PMID 23751865.