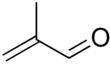
Methacrolein
Methacrolein, or methacrylaldehyde, is an unsaturated aldehyde. It is a clear, colorless, flammable liquid.
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Methylprop-2-enal | |||
| Other names
Methacrolein Methacrylaldehyde Isobutenal | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
| ECHA InfoCard | 100.001.046 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C4H6O | |||
| Molar mass | 70.09 g/mol | ||
| Density | 0.847 g/cm3 | ||
| Melting point | −81 °C (−114 °F; 192 K) | ||
| Boiling point | 69 °C (156 °F; 342 K) | ||
| Related compounds | |||
Related alkenals |
Citral | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Methacrolein is one of two major products resulting from the reaction of isoprene with OH in the atmosphere, the other product being methyl vinyl ketone (MVK, also known as butenone).[1] These compounds are important components of the atmospheric oxidation chemistry of biogenic chemicals, which can result in the formation of ozone and/or particulates. Methacrylaldehyde is also present in cigarette smoke.[2]It can be found in the essential oil of the plant Big Sagebrush (Artemisia tridentata) contains 5% methacrolein.[3]
Industrially, the primary use of methacrolein is in the manufacture of polymers and synthetic resins.
Exposure to methacrolein is highly irritating to the eyes, nose, throat and lungs.
See also
References
- Montzka, S. A.; Trainer, M.; Goldan, P. D.; Kuster, W. C.; Fehsenfeld, F. C. (1993). "Isoprene and its oxidation products, methyl vinyl ketone and methacrolein, in the rural troposphere". Journal of Geophysical Research: Atmospheres. 98 (D1): 1101–1111. Bibcode:1993JGR....98.1101M. doi:10.1029/92JD02382.
- Roy J. Shephard (1982). The risks of passive smoking. ISBN 9780709923343. Retrieved 2009-05-06.
- Shakhnoza, Azimova S. et al. (2012). Lipids, Lipophilic Components and Essential Oils from Plant Sources. Springer. p. 844. ISBN 9780857293237.CS1 maint: uses authors parameter (link)