Prato reaction
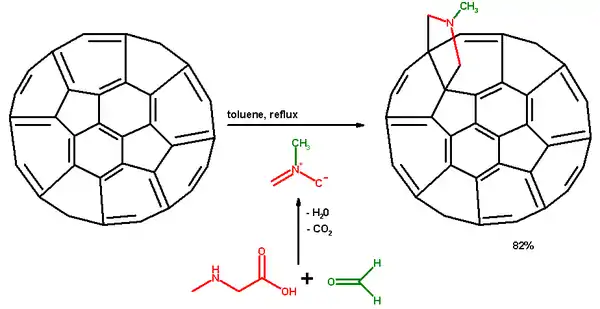
The Prato reaction is a particular example of the well-known 1,3-dipolar cycloaddition of azomethine ylides to olefins.[1] In fullerene chemistry this reaction refers to the functionalization of fullerenes and nanotubes. The amino acid sarcosine reacts with paraformaldehyde when heated at reflux in toluene to an ylide which reacts with a double bond in a 6,6 ring position in a fullerene via a 1,3-dipolar cycloaddition to yield a N-methylpyrrolidine derivative or pyrrolidinofullerene or pyrrolidino[[3,4:1,2]] [60]fullerene in 82% yield based on C60 conversion.[2]
Applications
In one application a liquid fullerene is obtained when the pyrrolidone substituent is a 2,4,6-tris(alkyloxy)phenyl group [3] although a small amount of solvent is still possibly present.
Origins
This reaction was derived from the work of Otohiko Tsuge [4] on Azomethine Ylide Chemistry developed in the late 1980s. Tsuge's work was applied to fullerenes by Maurizio Prato, thus gaining the name.
Metallofullerenes and Carbon Nanotubes
It is known that the Prato reaction is very useful to functionalize endohedral metallofullerenes. Prato reaction on M3N@C80 gives initially [6,6]-adduct (kinetic product), which convert upon heating to the [5,6]-adduct (thermodynamic product).[5] The rate of isomerization is highly dependent on the metal size inside the carbon cage.[6]
This method is also used in the functionalization of single wall nanotubes.[7] When the amino acid is modified with a glycine chain the resulting nanotubes are soluble in common solvents such chloroform and acetone. Another characteristic of the treated nanotubes is their larger aggregate dimensions compared to untreated nanotubes.
In an alternative method a nanotube addition is performed with the N-oxide of trimethylamine and LDA [8] at reflux in tetrahydrofuran with an efficiency of 1 functional group in 16 nanotube carbon atoms. When the amine also carries an aromatic group such as pyrene the reaction takes place even at room temperature because this group preorganizes itself to the nanotube surface prior to reaction by pi stacking.
Retro-Prato reaction
Just as in other fullerene reactions like the Bingel reaction or Diels-Alder reactions this reaction can be reversed. A thermal cycloelimination of a pyrrolidinofullerene with a strong dipolarophile such as maleic acid and a catalyst such as Wilkinson's catalyst or copper triflate in 1,2-dichlorobenzene at reflux 8 to 18 hours regenerates the pristine C60 fullerene.[9] The dipolarophile is required in a 30 fold excess and traps the ylide driving the reaction to completion. The N-methylpyrrolidine derivative reacts poorly (5% yield) and for a successful reaction the nitrogen ring also requires substitution in the α-position with methyl, phenyl or carboxylic ester groups.
Other methods have been investigated: by applying heat [10] or via a combination of ionic liquid and microwave chemistry.[11][12]
References
- Tsuge, Otohiko; Kanemasa, Shuji (1989). Recent Advances in Azomethine Ylide Chemistry. Advances in Heterocyclic Chemistry. 45. pp. 231–349. doi:10.1016/S0065-2725(08)60332-3. ISBN 9780120206452.
- Maggini, Michele; Scorrano, Gianfranco; Prato, Maurizio (1993). "Addition of azomethine ylides to C60: synthesis, characterization, and functionalization of fullerene pyrrolidines". J. Am. Chem. Soc. 115 (21): 9798–9799. doi:10.1021/ja00074a056.
- Michinobu T, Nakanishi T, Hill JP, Funahashi M, Ariga K (2006). "Room Temperature Liquid Fullerenes: An Uncommon Morphology of C60 Derivatives". J. Am. Chem. Soc. 128 (32): 10384–10385. doi:10.1021/ja063866z. PMID 16895401.
- Tsuge, Otohiko; Kanemasa, Shuji (1989). Recent Advances in Azomethine Ylide Chemistry. Advances in Heterocyclic Chemistry. 45. pp. 231–349. doi:10.1016/S0065-2725(08)60332-3. ISBN 9780120206452.
- Cardona, Claudia M.; Elliott, Bevan; Echegoyen, Luis (2006). "Unexpected Chemical and Electrochemical Properties of M3N@C80 (M = Sc, Y, Er)". J. Am. Chem. Soc. 128 (19): 6480–6485. doi:10.1021/ja061035n. PMID 16683813.
- Aroua, S.; Yamakoshi, Y. (2012). "Prato Reaction of M3N@Ih-C80 (M = Sc, Lu, Y, Gd) with Reversible Isomerization". J. Am. Chem. Soc. 134 (50): 20242–20245. doi:10.1021/ja309550z. PMID 23210903.
- Georgakilas V, Kordatos K, Prato M, Guldi DM, Holzinger M, Hirsch A (2002). "Organic Functionalization of Carbon Nanotubes". J. Am. Chem. Soc. 124 (5): 760–761. doi:10.1021/ja016954m. PMID 11817945.
- Ménard-Moyon C, Izard N, Doris E, Mioskowski C (2006). "Separation of Semiconducting from Metallic Carbon Nanotubes by Selective Functionalization with Azomethine Ylides". J. Am. Chem. Soc. 128 (20): 6552–6553. doi:10.1021/ja060802f. PMID 16704243.
- Martín N, Altable M, Filippone S, Martín-Domenech A, Echegoyen L, Cardona CM (2006). "Retro-Cycloaddition Reaction of Pyrrolidinofullerenes". Angewandte Chemie International Edition. 45 (1): 110–114. doi:10.1002/anie.200502556. PMID 16240308.
- Filippone, Salvatore; Barroso, Marta Izquierdo; Martín-Domenech, ÁNgel; Osuna, SíLvia; Solà, Miquel; Martín, Nazario (2008). "On the Mechanism of the Thermal Retrocycloaddition of Pyrrolidinofullerenes (Retro-Prato Reaction)". Chemistry: A European Journal. 14 (17): 5198–206. doi:10.1002/chem.200800096. PMID 18438770. S2CID 26077850.
- Guryanov, Ivan; Montellano López, Alejandro; Carraro, Mauro; Da Ros, Tatiana; Scorrano, Gianfranco; Maggini, Michele; Prato, Maurizio; Bonchio, Marcella (2009). "Metal-free, retro-cycloaddition of fulleropyrrolidines in ionic liquids under microwave irradiation". Chemical Communications (26): 3940–2. doi:10.1039/b906813a. PMID 19662259.
- Guryanov, Ivan; Toma, Francesca Maria; Montellano López, Alejandro; Carraro, Mauro; Da Ros, Tatiana; Angelini, Guido; D'aurizio, Eleonora; Fontana, Antonella; et al. (2009). "Microwave-Assisted Functionalization of Carbon Nanostructures in Ionic Liquids". Chemistry: A European Journal. 15 (46): 12837–45. doi:10.1002/chem.200901408. PMID 19847823.
External links
- Elizabeth Wilson (4 February 2002). "How to make the insoluble soluble: Attaching large organic groups is key to unclumping nanotubes". Chemical & Engineering News. 80 (5).