Prochlorococcus
Prochlorococcus is a genus of very small (0.6 µm) marine cyanobacteria with an unusual pigmentation (chlorophyll a2 and b2). These bacteria belong to the photosynthetic picoplankton and are probably the most abundant photosynthetic organism on Earth. Prochlorococcus microbes are among the major primary producers in the ocean, responsible for a large percentage of the photosynthetic production of oxygen.[1][2] Analysis of the genome sequences of 12 Prochlorococcus strains show that 1,100 genes are common to all strains, and the average genome size is about 2,000 genes.[1] In contrast, eukaryotic algae have over 10,000 genes.[3]
| Prochlorococcus | |
|---|---|
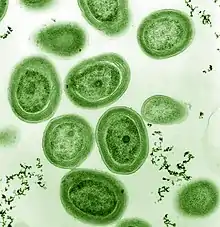 | |
| SEM of Prochlorococcus marinus (pseudo-colored) | |
| Scientific classification | |
| Domain: | Bacteria |
| Phylum: | Cyanobacteria |
| Class: | Cyanophyceae |
| Order: | Synechococcales |
| Family: | Prochloraceae |
| Genus: | Prochlorococcus Chisholm et al., 1992 |
| Species: | P. marinus |
| Binomial name | |
| Prochlorococcus marinus Chisholm et al., 1992 | |
Discovery
Although there had been several earlier records of very small chlorophyll-b-containing cyanobacteria in the ocean,[4][5] Prochlorococcus was discovered in 1986[6] by Sallie W. (Penny) Chisholm of the Massachusetts Institute of Technology, Robert J. Olson of the Woods Hole Oceanographic Institution, and other collaborators in the Sargasso Sea using flow cytometry. Chisholm was awarded the Crafoord Prize in 2019 for the discovery.[7] The first culture of Prochlorococcus was isolated in the Sargasso Sea in 1988 (strain SS120) and shortly another strain was obtained from the Mediterranean Sea (strain MED). The name Prochlorococcus[8] originated from the fact it was originally assumed that Prochlorococcus was related to Prochloron and other chlorophyll-b-containing bacteria, called prochlorophytes, but it is now known that prochlorophytes form several separate phylogenetic groups within the cyanobacteria subgroup of the bacteria domain.
The only species of the genus that has been described is Prochlorococcus marinus.
Morphology
Marine cyanobacteria are to date the smallest known photosynthetic organisms; Prochlorococcus is the smallest at just 0.5 to 0.7 micrometres in diameter.[9][2] The coccoid shaped cells are non-motile and free-living. Their small size and large surface-area-to-volume ratio, gives them an advantage in nutrient-poor water. Still, it is assumed that Prochlorococcus have a very small nutrient requirement.[10] Moreover, Prochlorococcus have adapted to use sulfolipids instead of phospholipids in their membranes to survive in phosphate deprived environments.[11] This adaptation allows them to avoid competition with heterotrophs that are dependent on phosphate for survival.[11] Typically, Prochlorococcus divide once a day in the subsurface layer or oligotrophic waters.[10]
Distribution
Prochlorococcus is abundant in the euphotic zone of the world's tropical oceans.[12] It is possibly the most plentiful genus on Earth: a single millilitre of surface seawater may contain 100,000 cells or more. Worldwide, the average yearly abundance is (2.8 to 3.0)×1027 individuals[13] (for comparison, that is approximately the number of atoms in a ton of gold). Prochlorococcus is ubiquitous between 40°N and 40°S and dominates in the oligotrophic (nutrient-poor) regions of the oceans.[10] Prochlorococcus is mostly found in a temperature range of 10-33 °C and some strains can grow at depths with low light (<1% surface light).[1] These strains are known as LL (Low Light) ecotypes, with strains that occupy shallower depths in the water column known as HL (High Light) ecotypes.[14] LL type Prochlorococcus have a higher ratio of chlorophyll b to chlorophyll a, which aids in their ability to absorb blue light.[15] Blue light is able to penetrate ocean waters deeper than the rest of the visible spectrum, and can reach depths of >200 m, depending on the turbidity of the water. This penetration depth of blue light, combined with the ability of LL type Prochlorococcus to utilise it for photosynthesis, allows populations of LL Prochlorococcus to survive at depths of up to 200 m.[16] Furthermore, Prochlorococcus are more plentiful in the presence of heterotrophs that have catalase abilities.[17] Prochlorococcus do not have mechanisms to degrade reactive oxygen species and rely on heterotrophs to protect them.[17] The bacterium accounts for an estimated 13-48% of the global photosynthetic production of oxygen, and forms part of the base of the ocean food chain.[18]
Pigments
Prochlorococcus is closely related to Synechococcus, another abundant photosynthetic cyanobacteria, which contains the light-harvesting antennae phycobilisomes. However, Prochlorochoccus has evolved to use a unique light-harvesting complex, consisting predominantly of divinyl derivatives of chlorophyll a (Chl a2) and b (Chl b2) and lacking monovinyl chlorophylls and phycobilisomes.[19] Prochlorococcus is the only known wild-type oxygenic phototroph that does not contain Chl a as a major photosynthetic pigment, and is the only known prokaryote with α-carotene.[20]
Prochlorococcus occupies two distinct niches, leading to the nomenclature of the low light (LL) and high light (HL) groups,[21] which vary in pigment ratios (LL has a high ratio of chlorophyll b2:a2 and HL has a low ratio of b2:a2), light requirements, nitrogen and phosphorus utilization, copper, and virus sensitivity. These "ecotypes" can be differentiated on the basis of the sequence of their ribosomal RNA gene. High-light adapted strains inhabit depths between 25 and 100 m, while low-light adapted strains inhabit waters between 80 and 200 m.[22]
Genome
Recently the genomes of several strains of Prochlorococcus have been sequenced.[23][24] Twelve complete genomes have been sequenced which reveal physiologically and genetically distinct lineages of Prochlorococcus marinus that are 97% similar in the 16S rRNA gene.[22]
The high-light ecotype has the smallest genome (1,657,990 basepairs, 1,716 genes) of any known oxygenic phototroph, but the genome of the low-light type is much larger (2,410,873 base pairs, 2,275 genes).[23]
Ecology
Despite Prochlorococcus being one of the smallest types of marine phytoplankton/bacteria in the world's oceans, its substantial number makes it responsible for a major part of the oceans' and world's photosynthesis and oxygen production.[2] The size of Prochlorococcus (0.5 to 0.7 μm)[10] and the adaptations of the various ecotypes allow the organism to grow abundantly in low nutrient waters such as the waters of the tropics and the subtropics (c. 40°N to 40°S);[25] however, they can be found in higher latitudes as high up as 60° north but at fairly minimal concentrations and the bacteria's distribution across the oceans suggest that the colder waters could be fatal to it. This wide range of latitude along with the bacteria's ability to survive up to depths of 100 to 150 metres, i.e. the average depth of the mixing layer of the surface ocean, allows it to grow to enormous numbers, up to 3 octillion individuals worldwide.[26] This enormous number makes the Prochlorococcus play an important role in the global carbon cycle and oxygen production. Along with Synechococcus (another genus of cyanobacteria that co-occurs with Prochlorococcus) these cyanobacteria are responsible for approximately 50% of marine carbon fixation, making it an important carbon sink via the biological carbon pump (i.e. the transfer of organic carbon from the surface ocean to the deep via several biological, physical and chemical processes).[27] The abundance, distribution and all other characteristics of the Prochlorococcus make it a key organism in oligotrophic waters serving as an important primary producer to the open ocean food webs.
References
- Munn, C. Marine Microbiology: ecology and applications Second Ed. Garland Science, 2011.
- "Life at the Edge of Sight — Scott Chimileski, Roberto Kolter | Harvard University Press". www.hup.harvard.edu. Retrieved 2018-01-26.
- Kettler GC, Martiny AC, Huang K, et al. (December 2007). "Patterns and Implications of Gene Gain and Loss in the Evolution of Prochlorococcus". PLOS Genetics. 3 (12): e231. doi:10.1371/journal.pgen.0030231. PMC 2151091. PMID 18159947.
- P. W. Johnson & J. M. Sieburth (1979). "Chroococcoid cyanobacteria in the sea: a ubiquitous and diverse phototrophic biomass". Limnology and Oceanography. 24 (5): 928–935. Bibcode:1979LimOc..24..928J. doi:10.4319/lo.1979.24.5.0928.
- W. W. C. Gieskes & G. W. Kraay (1983). "Unknown chlorophyll a derivatives in the North Sea and the tropical Atlantic Ocean revealed by HPLC analysis". Limnology and Oceanography. 28 (4): 757–766. Bibcode:1983LimOc..28..757G. doi:10.4319/lo.1983.28.4.0757.
- S. W. Chisholm; R. J. Olson; E. R. Zettler; J. Waterbury; R. Goericke; N. Welschmeyer (1988). "A novel free-living prochlorophyte occurs at high cell concentrations in the oceanic euphotic zone". Nature. 334 (6180): 340–343. Bibcode:1988Natur.334..340C. doi:10.1038/334340a0. S2CID 4373102.
- https://www.crafoordprize.se/press_release/the-crafoord-prize-in-biosciences-2019. Missing or empty
|title=(help) - Sallie W. Chisholm, S. L. Frankel, R. Goericke, R. J. Olson, B. Palenik, J. B. Waterbury, L. West-Johnsrud & E. R. Zettler (1992). "Prochlorococcus marinus nov. gen. nov. sp.: an oxyphototrophic marine prokaryote containing divinyl chlorophyll a and b". Archives of Microbiology. 157 (3): 297–300. doi:10.1007/BF00245165. S2CID 32682912.CS1 maint: multiple names: authors list (link)
- Biller, Steven J.; Berube, Paul M.; Lindell, Debbie; Chisholm, Sallie W. (1 December 2014). "Prochlorococcus: the structure and function of collective diversity" (PDF). Nature Reviews Microbiology. 13 (1): 13–27. doi:10.1038/nrmicro3378. hdl:1721.1/97151. PMID 25435307. S2CID 18963108.
- Partensky F, Hess WR, Vaulot D (1999). "Prochlorococcus, a marine photosynthetic prokaryote of global significance". Microbiology and Molecular Biology Reviews. 63 (1): 106–127. doi:10.1128/MMBR.63.1.106-127.1999. PMC 98958. PMID 10066832.
- Van Mooy, B. A. S.; Rocap, G.; Fredricks, H. F.; Evans, C. T.; Devol, A. H. (26 May 2006). "Sulfolipids dramatically decrease phosphorus demand by picocyanobacteria in oligotrophic marine environments". Proceedings of the National Academy of Sciences. 103 (23): 8607–8612. Bibcode:2006PNAS..103.8607V. doi:10.1073/pnas.0600540103. PMC 1482627. PMID 16731626.
- Chisholm, S.W.; Frankel, S.; Goericke, R.; Olson, R.; Palenik, B.; Waterbury, J.; West-Johnsrud, L.; Zettler, E. (1992). "Prochlorococcus marinus nov. gen. nov. sp.: an oxyphototrophic marine prokaryote containing divinyl chlorophyll a and b.". Archives of Microbiology. 157 (3): 297–300. doi:10.1007/bf00245165. S2CID 32682912.
- Flombaum, P.; Gallegos, J. L.; Gordillo, R. A.; Rincon, J.; Zabala, L. L.; Jiao, N.; Karl, D. M.; Li, W. K. W.; Lomas, M. W.; Veneziano, D.; Vera, C. S.; Vrugt, J. A.; Martiny, A. C. (2013). "Present and future global distributions of the marine Cyanobacteria Prochlorococcus and Synechococcus". Proceedings of the National Academy of Sciences. 110 (24): 9824–9829. Bibcode:2013PNAS..110.9824F. doi:10.1073/pnas.1307701110. PMC 3683724. PMID 23703908.
- Coleman, M.; Sullivan, M.; Martiny, A.; Steglich, C.; Barry, K.; DeLong, E.; Chisholm, S. (2006). "Genomic islands and the ecology and evolution of Prochlorococcus". Science. 311 (5768): 1768–1770. Bibcode:2006Sci...311.1768C. doi:10.1126/science.1122050. PMID 16556843. S2CID 3196592.
- Ralf, G.; Repeta, D. (1992). "The pigments of Prochlorococcus marinus: The presence of divinylchlorophyll a and b in a marine prokaryote". Limnology and Oceanography. 37 (2): 425–433. Bibcode:1992LimOc..37..425R. doi:10.4319/lo.1992.37.2.0425.
- Zinser, E.; Johnson, Z.; Coe, A.; Karaca, E.; Veneziano, D.; Chisholm, S. (2007). "Influence of light and temperature on Prochlorococcus ecotype distributions in the Atlantic Ocean". Limnology and Oceanography. 52 (5): 2205–2220. Bibcode:2007LimOc..52.2205Z. doi:10.4319/lo.2007.52.5.2205.
- Morris, J. J.; Kirkegaard, R.; Szul, M. J.; Johnson, Z. I.; Zinser, E. R. (23 May 2008). "Facilitation of Robust Growth of Prochlorococcus Colonies and Dilute Liquid Cultures by "Helper" Heterotrophic Bacteria". Applied and Environmental Microbiology. 74 (14): 4530–4534. doi:10.1128/AEM.02479-07. PMC 2493173. PMID 18502916.
- Johnson, Zachary I.; Zinser, Erik R.; Coe, Allison; McNulty, Nathan P.; Woodward, E. Malcolm S.; Chisholm, Sallie W. (2006). "Niche Partitioning among Prochlorococcus Ecotypes along Ocean-Scale Environmental Gradients". Science. 311 (5768): 1737–1740. Bibcode:2006Sci...311.1737J. doi:10.1126/science.1118052. PMID 16556835. S2CID 3549275.
- Ting CS, Rocap G, King J, Chisholm S (2002). "Cyanobacterial photosynthesis in the oceans: the origins and significance of divergent light-harvesting strategies". Trends in Microbiology. 10 (3): 134–142. doi:10.1016/s0966-842x(02)02319-3. PMID 11864823.
- Goericke R, Repeta D (1992). "The pigments of Prochlorococcus marinus: the presence of divinyl chlorophyll a and b in a marine prokaryote". Limnology and Oceanography. 37 (2): 425–433. Bibcode:1992LimOc..37..425R. doi:10.4319/lo.1992.37.2.0425.
- N. J. West & D. J. Scanlan (1999). "Niche-partitioning of Prochlorococcus in a stratified water column in the eastern North Atlantic Ocean". Applied and Environmental Microbiology. 65 (6): 2585–2591. doi:10.1128/AEM.65.6.2585-2591.1999. PMC 91382. PMID 10347047.
- Martiny AC, Tai A, Veneziano D, Primeau F, Chisholm S (2009). "Taxonomic resolution, ecotypes and biogeography of Prochlorococcus". Environmental Microbiology. 11 (4): 823–832. doi:10.1111/j.1462-2920.2008.01803.x. PMID 19021692.
- G. Rocap; F. W. Larimer; J. Lamerdin; S. Malfatti; P. Chain; N. A. Ahlgren; A. Arellano; M. Coleman; L. Hauser; W. R. Hess; Z. I. Johnson; M. Land; D. Lindell; A. F. Post; W. Regala; M. Shah; S. L. Shaw; C. Steglich; M. B. Sullivan; C. S. Ting; A. Tolonen; E. A. Webb; E. R. Zinser; S. W. Chisholm (2003). "Genome divergence in two Prochlorococcus ecotypes reflects oceanic niche differentiation" (PDF). Nature. 424 (6952): 1042–1047. Bibcode:2003Natur.424.1042R. doi:10.1038/nature01947. PMID 12917642. S2CID 4344597. Archived from the original (– Scholar search) on December 11, 2004.
- A. Dufresne; M. Salanoubat; F. Partensky; F. Artiguenave; I. M. Axmann; V. Barbe; S. Duprat; M. Y. Galperin; E. V. Koonin; F. Le Gall; K. S. Makarova; M. Ostrowski; S. Oztas; C. Robert; I. B. Rogozin; D. J. Scanlan; N. Tandeau de Marsac; J. Weissenbach; P. Wincker; Y. I. Wolf; W. R. Hess (2003). "Genome sequence of the cyanobacterium Prochlorococcus marinus SS120, a nearly minimal oxyphototrophic genome". Proceedings of the National Academy of Sciences. 100 (17): 10020–10025. Bibcode:2003PNAS..10010020D. doi:10.1073/pnas.1733211100. PMC 187748. PMID 12917486.
- Partensky, Blanchot, Vaulot (1999). "DifferentiaI distribution and ecology of Prochlorococcus and Synechococcus in oceanic waters: a review". Bulletin de l'Institut Océanographique. Monaco. N°. spécial 19 (spécial 19).CS1 maint: multiple names: authors list (link)
- Zinser, Erik R.; Coe, Allison; Johnson, Zackary I.; Martiny, Adam C.; Fuller, Nicholas J.; Scanlan, David J.; Chisholm, Sallie W. (January 2006). "Prochlorococcus Ecotype Abundances in the North Atlantic Ocean As Revealed by an Improved Quantitative PCR Method†". Applied and Environmental Microbiology. 72 (1): 723–732. doi:10.1128/aem.72.1.723-732.2006. PMC 1352191. PMID 16391112.
- Fu, Fei-Xue; Warner, Mark E.; Zhang, Yaohong; Feng, Yuanyuan; Hutchins, David A. (16 May 2007). "Effects of Increased Temperature and CO2 on Photosynthesis, Growth, and Elemental Ratios in Marine Synechococcus and Prochlorococcus (Cyanobacteria)". Journal of Phycology. 43 (3): 485–496. doi:10.1111/j.1529-8817.2007.00355.x. S2CID 53353243.
Further reading
- L. Campbell, H. A. Nolla & D. Vaulot (1994). "The importance of Prochlorococcus to community structure in the central North Pacific Ocean". Limnology and Oceanography. 39 (4): 954–961. Bibcode:1994LimOc..39..954C. doi:10.4319/lo.1994.39.4.0954.
- Jagroop Pandhal, Phillip C. Wright & Catherine A. Biggs (2007). "A quantitative proteomic analysis of light adaptation in a globally significant marine cyanobacterium Prochlorococcus marinus MED4". Journal of Proteome Research. 6 (3): 996–1005. doi:10.1021/pr060460c. PMID 17298086.
- Steve Nadis (2003). "The cells that rule the seas: the ocean's tiniest inhabitants, notes biological researcher Sallie W. Chisholm, hold the key to understanding the biosphere — and what happens when humans disrupt it". Scientific American: 52f.
- Melissa Garren (2012). "The sea we've hardly seen". TEDx Monterey: 52f.
External links
- M. D. Guiry. "Prochlorococcus S.W. Chisholm, S.L. Frankel, R. Goericke, R.J. Olson, B. Palenik, J.B. Waterbury, L. West-Johnsrud & E.R. Zettler 1992: 299". AlgaeBase.
- The Most Important Microbe You've Never Heard Of: NPR Story on Prochlorococcus
- Anacyte Laboratories
- Genome Information for Prochlorococcus marinus: National Center for Biotechnology Information.
