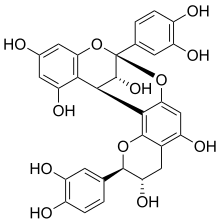
Procyanidin A1
Procyanidin A1 is an A type proanthocyanidin dimer.
 | |
| Names | |
|---|---|
| IUPAC name
(2R,3S,8S,14R,15R)-2,8-bis(3,4-dihydroxyphenyl)-2,3,4,14-tetrahydro-8,14-methanobenzo[7,8][1,3]dioxocino[4,5-h]chromene-3,5,11,13,15-pentaol | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C30H24O12 | |
| Molar mass | 576.510 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
It is an epicatechin-(2β→7,4β→8)-catechin dimer found in Rhododendron spiciferum,[1] in peanut skins[2] and in Ecdysanthera utilis.[3]
Procyanidin B1 can be converted into procyanidin A1 by radical oxidation using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals under neutral conditions.[4]
References
- Liu, YZ; Cao, YG; Ye, JQ; Wang, WG; Song, KJ; Wang, XL; Wang, CH; Li, RT; Deng, XM (2009). "Immunomodulatory effects of proanthocyanidin A-1 derived in vitro from Rhododendron spiciferum". Fitoterapia. 81 (2): 108–14. doi:10.1016/j.fitote.2009.08.005. PMID 19686816.
- Hongxiang Lou; Yamazaku Y.; Sasaku T.; Uchida M.; Tanaka H.; Oka S. (1999). "A-type proanthocyanidins from peanut skins". Phytochemistry. 51 (2): 297–308. doi:10.1016/S0031-9422(98)00736-5.
- Lin, Lie-Chwen; Kuo, Yuh-Chi; Chou, Cheng-Jen (2002). "Immunomodulatory Proanthocyanidins fromEcdysantherautilis". Journal of Natural Products. 65 (4): 505–8. doi:10.1021/np010414l. PMID 11975489.
- Conversion of procyanidin B-type (catechin dimer) to A-type: evidence for abstraction of C-2 hydrogen in catechin during radical oxidation. Kazunari Kondo, Masaaki Kurihara, Kiyoshi Fukuhara, Takashi Tanaka, Takashi Suzuki, Naoki Miyata and Masatake Toyoda, Tetrahedron Letters, 22 January 2000, Volume 41, Issue 4, Pages 485–488, doi:10.1016/S0040-4039(99)02097-3
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.