Small Maf
Small Maf (musculoaponeurotic fibrosarcoma) proteins are basic region leucine zipper-type transcription factors that can bind to DNA and regulate gene regulation.[1][2] There are three small Maf (sMaf) proteins, namely MafF, MafG, and MafK, in vertebrates.[3] HUGO Gene Nomenclature Committee (HGNC)-approved gene names of MAFF, MAFG and MAFK are “v-maf avian musculoaponeurotic fibrosarcoma oncogene homolog F, G, and K”, respectively.
Through the leucine zipper structures, sMafs form homodimers by themselves[2] and heterodimers with other specific bZIP transcription factors, such as transcription factors of the CNC (cap 'n' collar)[4][5][6][7] and Bach families.[8] Because CNC and Bach proteins cannot bind to DNA by themselves, sMafs are indispensable partners of the CNC and Bach families of transcription factors. Through interactions with these transcription factors, sMafs actively participate in transcriptional activation or repression depending on the nature of the heterodimeric partners.
Subtypes
The following genes encode small Maf proteins
History and discovery
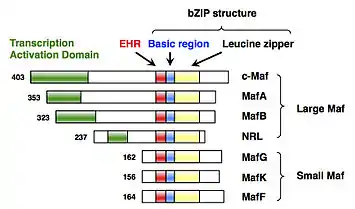
sMaf proteins were identified as members of the Maf family transcription factors.[2] The Maf family is divided into two subfamilies, as follows: the large Maf subfamily (c-Maf, MafA, MafB, and NRL); and the small Maf subfamily (MafF, MafG and MafK) (Fig. 1). The first member of the Maf family is c-Maf, which was cloned as a cellular counterpart of the v-Maf oncogene isolated from avian musculoaponeurotic fibrosarcoma.[9] The MafF, MafG, and MafK genes were later isolated.[1][2] Because MafF, MafG and MafK are well-conserved 18 kDa proteins that lack a transcriptional activation domain, they are classified into the small Maf subfamily, which is structurally and functionally distinct from the large Maf subfamily.[1]
Gene Structure and regulation
Three sMaf genes are widely expressed in various cell types and tissues under differential transcriptional regulation.[10][11] In mouse, each sMaf gene harbors multiple first exons, which partly contribute to their tissue-specific or stimulus-specific expression patterns.[11][12][13][14] Human MAFF is induced by proinflammatory cytokines.[15] Mouse Mafg gene is induced by oxidative stresses (e.g. reactive oxygen species and electrophilic compounds) or the presence of bile acids.[14][16] Mouse Mafk gene is under the regulation of GATA factors (GATA-1 and GATA-2 in hematopoietic tissues; and GATA-4 and GATA-6 in cardiac tissues).[17]
Protein Structure
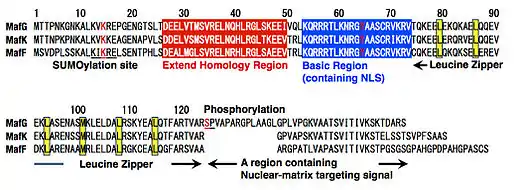
All members of the Maf family including sMafs have a bZIP structure that consists of the basic region for DNA binding and the leucine zipper structure for dimer formation (Fig. 2). The basic region of each Maf family protein contains a tyrosine residue, which is critical for the unique DNA-binding modes of these proteins (see below for details).[18] In addition, each Maf family protein possesses an extended homology region (EHR), which contributes to stable DNA binding.[19][20] The C-terminal region of sMaf includes a region required for its proper subnuclear localization.[21] Two modifications have been reported for MafG: SUMOylation through a SUMOylation motif at the N-terminal region;[22] phosphorylation through an ERK phosphorylation site in the C-terminal region.[23]
Function
sMaf proteins form homodimers by themselves and heterodimers with two other bZIP families of transcription factors, namely CNC (cap 'n' collar) proteins (p45 NF-E2 (NFE2), Nrf1 (NFE2L1), Nrf2 (NFE2L2), and Nrf3 (NFE2L3) – not to be confused with Nuclear Respiratory factors)[4][5][6][7] and Bach proteins (Bach1 and Bach2).[8] Because these proteins cannot bind DNA by themselves, sMaf proteins are indispensable partner molecules of the CNC and Bach transcription factors.
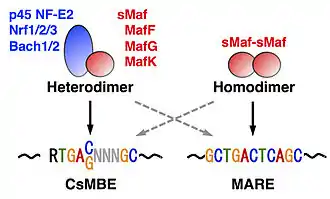
sMaf homodimers bind to a palindromic DNA sequence called the Maf recognition element (MARE: TGCTGACTCAGCA) and its related sequences.[2] Structural analyses have demonstrated that the basic region of a Maf factor recognizes the flanking GC sequences.[20] By contrast, CNC-sMaf or Bach-sMaf heterodimers preferentially bind to DNA sequences (RTGA(C/G)NNNGC: R=A or G) that are slightly different from MARE (Fig. 3).[24][25][26] The latter DNA sequences have been recognized as antioxidant/electrophile response elements[27][28] or NF-E2-binding motifs,[29][30] to which Nrf2-sMaf heterodimers and p45 NF-E2-sMaf heterodimers bind, respectively. It has been proposed that the latter sequences are classified as CNC-sMaf-binding elements (CsMBEs).[26]
It has also been reported that sMafs form heterodimers with other bZIP transcription factors, such as c-Jun and c-Fos.[31] However, the biological significance of these heterodimers remains unknown.
sMaf homodimer
Because sMafs lack any canonical transcriptional activation domains, the sMaf homodimer act as a negative regulator. Overexpression of MafG is known to inhibit proplatelet formation, which is thought to reflect a process of platelet production.[32] SUMOylation is required for MafG homodimer-mediated transcriptional repression.[22]
p45 NF-E2-sMaf heterodimer
The p45 NF-E2-sMaf heterodimers are critical for platelet production. Knockout mouse studies have shown that MafG knockout mice show mild thrombocytopenia,[33] whereas MafG and MafK double mutant mice show severe thrombocytopenia.[34] Similar results were also observed in p45 NF-E2 knockout mice.[35] The p45 NF-E2-sMaf heterodimer regulates genes responsible for platelet production and function.[36]
Nrf1-sMaf heterodimer
The Nrf1-sMaf heterodimers are critical for neuronal homeostasis. Knockout mouse studies have shown that Mafg knockout mice display mild ataxia.[33] Mafg and Mafk mutant mice (Mafg−/−::Mafk+/−) show more severe ataxia with progressive neuronal degeneration.[37] Similar results have also been observed in Nrf1 central nervous-specific knockout mice.[38][39] The Nrf1-sMaf heterodimers regulate genes responsible for proteasomal genes and metabolism genes.[40]
Nrf2-sMaf heterodimer
The Nrf2-sMaf heterodimers are critical for oxidative and electrophilic stress response. Nrf2 is known as a master regulator of antioxidant and xenobiotic metabolizing enzyme genes.[6] Induction of these cytoprotective genes is impaired in Nrf2 knockout mice.[6] While MafG, MafK and MafF triple knockout mice die in embryonic stage, cultured cells derived from the triple knockout embryo fail to induce Nrf2-dependent cytoprotective genes in response to stimuli.[41]
Bach1-sMaf heterodimer
The Bach1-sMaf heterodimer is critical for heme metabolism. Knockout mouse studies showed that heme oxygenase-1 gene expression is upregulated in Bach1 knockout mice.[42] Similar results were also observed in MafG and MafK double mutant mice (Mafg−/−::Mafk+/−).[37] These data show that the Bach1-sMaf heterodimer negatively regulates heme oxygenase-1 gene.
Bach2-sMaf heterodimer
The Bach2-sMaf heterodimers are critical for B cell differentiation.[43] Bach2 knockout mice studies have demonstrated that Bach2 is required for class switching and somatic hypermutation of immunoglobulin genes.[44] However, these phenotypes have not been examined in sMaf knockout mice.
sMaf function with compound or unknown partners
MafG and MafK double mutant mice (Mafg−/−::Mafk+/−) have cataracts.[45] However, the interaction of CNC partner(s) with sMafs in this context remains undetermined. MafG, MafK and MafF triple knockout mice die during embryogenesis, demonstrating that sMafs are indispensable for embryonic development.[46] Because Nrf1 and Nrf2 double mutant mice also die during embryogenesis,[47] the loss of function of both Nrf1-sMaf and Nrf2-sMaf may contribute to the lethality.
| Genotype | Phenotype | ||
|---|---|---|---|
| Maff | Mafg | Mafk | |
| −/− | No apparent phenotype under laboratory conditions [11] | ||
| −/− | Mild motor ataxia, mild thrombocytopenia [33] | ||
| −/− | No apparent phenotype under laboratory conditions [33] | ||
| −/− | +/− | Severe motor ataxia, progressive neuronal degeneration, severe thrombocytopenia, and cataract [37][45] | |
| −/− | −/− | More severe neuronal phenotypes, and perinatal lethal [34] | |
| −/− | +/− | −/− | No severe abnormality (Fertile) [46] |
| −/− | −/− | −/− | Growth retardation, fetal liver hypoplasia, and lethal around embryonic day, 13.5 [46] |
| +/− (heterozygote), −/− (homozygote), blank (wild-type) | |||
Disease association
sMafs have been suggested to be involved in various diseases as heterodimeric partners of CNC and Bach proteins. Because Nrf2-sMaf heterodimers regulate a battery of antioxidant and xenobiotic metabolizing enzymes,[6][41] impaired function of sMafs is expected to make cells vulnerable to various stresses and increase the risk of various diseases, such as cancers. SNPs associated with the cancer onset were reported in MAFF and MAFG genes.[48][49] In addition, Nrf2 is known to be critical for anti-inflammatory responses.[50][51] Thus, sMaf insufficiencies are expected to result in prolonged inflammation that can cause diseases, such as neurodegeneration and atherosclerosis.
Conversely, sMafs also appear to contribute to cancer malignancy. Certain cancers contain somatic mutations in NRF2(NFE2L2) or KEAP1 that cause constitutive activation of Nrf2 and promote cell proliferation.[52] It has also been reported that the Bach1-MafG heterodimer contributes to cancer malignancy by repressing tumor suppressor genes.[23] Thus, as partners of Nrf2 and Bach1, sMafs are expected to play critical roles in cancer cells.
References
- Fujiwara, KT (1993). "Two new members of the maf oncogene family, mafK and mafF, encode nuclear b-Zip proteins lacking putative trans-activator domain". Oncogene. 8 (9): 2371–80. PMID 8361754.
- Kataoka, K (1995). "Small Maf proteins heterodimerize with Fos and may act as competitive repressors of the NF-E2 transcription factor". Mol. Cell. Biol. 15 (4): 2180–90. doi:10.1128/mcb.15.4.2180. PMC 230446. PMID 7891713.
- "NCBI Gene".
- Igarashi, K (1994). "Regulation of transcription by dimerization of erythroid factor NF-E2 p45 with small Maf proteins". Nature. 367 (6463): 568–72. Bibcode:1994Natur.367..568I. doi:10.1038/367568a0. PMID 8107826. S2CID 4339431.
- Johnsen, O (1998). "Interaction of the CNC-bZIP factor TCF11/LCR-F1/Nrf1 with MafG: binding-site selection and regulation of transcription. Nucleic Acids Res". Nucleic Acids Res. 26 (2): 512–20. doi:10.1093/nar/26.2.512. PMC 147270. PMID 9421508.
- Itoh, K (1997). "An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements". Biochem. Biophys. Res. Commun. 236 (2): 313–22. doi:10.1006/bbrc.1997.6943. PMID 9240432.
- Kobayashi, A (1999). "Molecular cloning and functional characterization of a new Cap'n' collar family transcription factor Nrf3". J. Biol. Chem. 274 (10): 6443–52. doi:10.1074/jbc.274.10.6443. PMID 10037736.
- Oyake, T (1996). "Bach proteins belong to a novel family of BTB-basic leucine zipper transcription factors that interact with MafK and regulate transcription through the NF-E2 site". Mol. Cell. Biol. 16 (11): 6083–95. doi:10.1128/mcb.16.11.6083. PMC 231611. PMID 8887638.
- Nishizawa, M (1989). "v-maf, a viral oncogene that encodes a "leucine zipper" motif". Proc. Natl. Acad. Sci. USA. 86 (20): 7711–5. Bibcode:1989PNAS...86.7711N. doi:10.1073/pnas.86.20.7711. PMC 298140. PMID 2554284.
- Toki, T (1997). "Human small Maf proteins form heterodimers with CNC family transcription factors and recognize the NF-E2 motif". Oncogene. 14 (16): 1901–10. doi:10.1038/sj.onc.1201024. PMID 9150357.
- Onodera, K (1999). "Characterization of the murine mafF gene". J. Biol. Chem. 274 (30): 21162–9. doi:10.1074/jbc.274.30.21162. PMID 10409670.
- Motohashi, H (1996). "Mesodermal- vs. neuronal-specific expression of MafK is elicited by different promoters". Genes Cells. 1 (2): 223–38. doi:10.1046/j.1365-2443.1996.d01-230.x. PMID 9140066.
- Motohashi, H (1998). "A core region of the mafK gene IN promoter directs neurone-specific transcription in vivo". Genes Cells. 3 (10): 671–84. doi:10.1046/j.1365-2443.1998.00222.x. PMID 9893024.
- Katsuoka, F (2005). "Nrf2 transcriptionally activates the mafG gene through an antioxidant response element". J. Biol. Chem. 280 (6): 4483–90. doi:10.1074/jbc.M411451200. PMID 15574414.
- Massrieh, W (2006). "Regulation of the MAFF transcription factor by proinflammatory cytokines in myometrial cells". Biol. Reprod. 74 (4): 699–705. doi:10.1095/biolreprod.105.045450. PMID 16371591. S2CID 11823930.
- de Aguiar Vallim, TQ (2015). "MAFG is a transcriptional repressor of bile acid synthesis and metabolism". Cell Metab. 21 (2): 298–310. doi:10.1016/j.cmet.2015.01.007. PMC 4317590. PMID 25651182.
- Katsuoka, F (2000). "One enhancer mediates mafK transcriptional activation in both hematopoietic and cardiac muscle cells". EMBO J. 19 (12): 2980–91. doi:10.1093/emboj/19.12.2980. PMC 203348. PMID 10856242.
- Kimura, M (2007). "Molecular basis distinguishing the DNA binding profile of Nrf2-Maf heterodimer from that of Maf homodimer". J. Biol. Chem. 282 (46): 33681–90. doi:10.1074/jbc.M706863200. PMID 17875642.
- Kusunoki, H (2002). "Solution structure of the DNA-binding domain of MafG". Nat. Struct. Biol. 9 (4): 252–6. doi:10.1038/nsb771. PMID 11875518. S2CID 23687470.
- Kurokawa, H (2009). "Structural basis of alternative DNA recognition by Maf transcription factors". Mol. Cell. Biol. 29 (23): 6232–44. doi:10.1128/MCB.00708-09. PMC 2786689. PMID 19797082.
- Motohashi, H (2011). "Molecular determinants for small Maf protein control of platelet production". Mol. Cell. Biol. 31 (1): 151–62. doi:10.1128/MCB.00798-10. PMC 3019851. PMID 20974807.
- Motohashi, H (2006). "MafG sumoylation is required for active transcriptional repression". Mol. Cell. Biol. 26 (12): 4652–63. doi:10.1128/MCB.02193-05. PMC 1489127. PMID 16738329.
- Fang, M (2014). "The BRAF oncoprotein functions through the transcriptional repressor MAFG to mediate the CpG Island Methylator phenotype". Mol. Cell. 55 (6): 904–15. doi:10.1016/j.molcel.2014.08.010. PMC 4170521. PMID 25219500.
- Hirotsu, Y (2012). "Nrf2-MafG heterodimers contribute globally to antioxidant and metabolic networks". Nucleic Acids Res. 40 (20): 10228–39. doi:10.1093/nar/gks827. PMC 3488259. PMID 22965115.
- Warnatz, HJ (2011). "The BTB and CNC homology 1 (BACH1) target genes are involved in the oxidative stress response and in control of the cell cycle". J. Biol. Chem. 286 (26): 23521–32. doi:10.1074/jbc.M111.220178. PMC 3123115. PMID 21555518.
- Otsuki, A (2015). "Unique cistrome defined as CsMBE is strictly required for Nrf2-sMaf heterodimer function in cytoprotection". Free Radical Bio. 91: 45–57. doi:10.1016/j.freeradbiomed.2015.12.005. PMID 26677805.
- Friling, RS (1990). "Xenobiotic-inducible expression of murine glutathione S-transferase Ya subunit gene is controlled by an electrophile-responsive element". Proc. Natl. Acad. Sci. USA. 87 (16): 6258–62. Bibcode:1990PNAS...87.6258F. doi:10.1073/pnas.87.16.6258. PMC 54512. PMID 2166952.
- Rushmore, TH (1991). "The antioxidant responsive element. Activation by oxidative stress and identification of the DNA consensus sequence required for functional activity". J. Biol. Chem. 266 (18): 11632–9. PMID 1646813.
- Mignotte, V (1989). "Cis- and trans-acting elements involved in the regulation of the erythroid promoter of the human porphobilinogen deaminase gene". Proc. Natl. Acad. Sci. USA. 86 (17): 6548–52. Bibcode:1989PNAS...86.6548M. doi:10.1073/pnas.86.17.6548. PMC 297881. PMID 2771941.
- Romeo, PH (1990). "Megakaryocytic and erythrocytic lineages share specific transcription factors". Nature. 344 (6265): 447–9. Bibcode:1990Natur.344..447R. doi:10.1038/344447a0. PMID 2320113. S2CID 4277397.
- Newman, JR (2003). "Comprehensive identification of human bZIP interactions with coiled-coil arrays". Science. 300 (5628): 2097–101. Bibcode:2003Sci...300.2097N. doi:10.1126/science.1084648. PMID 12805554. S2CID 36715183.
- Motohashi, H (2000). "MARE-dependent transcriptional regulation is determined by the abundance of small Maf proteins". Cell. 103 (6): 865–75. doi:10.1016/s0092-8674(00)00190-2. PMID 11136972. S2CID 16876406.
- Shavit, J (1998). "Impaired megakaryopoiesis and behavioral defects in mafG-null mutant mice". Genes Dev. 12 (14): 2164–74. doi:10.1101/gad.12.14.2164. PMC 317009. PMID 9679061.
- Onodera, K (2000). "Perinatal synthetic lethality and hematopoietic defects in compound mafG::mafK mutant mice". EMBO J. 19 (6): 1335–45. doi:10.1093/emboj/19.6.1335. PMC 305674. PMID 10716933.
- Shivdasani, RA (1995). "Transcription factor NF-E2 is required for platelet formation independent of the actions of thrombopoietin/MGDF in megakaryocyte development". Cell. 81 (5): 695–704. doi:10.1016/0092-8674(95)90531-6. PMID 7774011. S2CID 14195541.
- Fujita, R (2013). "NF-E2 p45 is important for establishing normal function of platelets". Mol. Cell. Biol. 33 (14): 2659–70. doi:10.1128/MCB.01274-12. PMC 3700136. PMID 23648484.
- Katsuoka, F (2003). "Small Maf compound mutants display central nervous system neuronal degeneration, aberrant transcription, and Bach protein mislocalization coincident with myoclonus and abnormal startle response". Mol. Cell. Biol. 23 (4): 1163–74. doi:10.1128/mcb.23.4.1163-1174.2003. PMC 141134. PMID 12556477.
- Kobayashi, A (2011). "Central nervous system-specific deletion of transcription factor Nrf1 causes progressive motor neuronal dysfunction". Genes Cells. 16 (6): 692–703. doi:10.1111/j.1365-2443.2011.01522.x. PMID 21554501.
- Lee, CS (2011). "Loss of nuclear factor E2-related factor 1 in the brain leads to dysregulation of proteasome gene expression and neurodegeneration". Proc. Natl. Acad. Sci. USA. 108 (20): 8408–13. Bibcode:2011PNAS..108.8408L. doi:10.1073/pnas.1019209108. PMC 3100960. PMID 21536885.
- Hirotsu, Y (2012). "NF-E2-related factor 1 (Nrf1) serves as a novel regulator of hepatic lipid metabolism through regulation of the Lipin1 and PGC-1β genes". Mol. Cell. Biol. 32 (14): 2760–70. doi:10.1128/MCB.06706-11. PMC 3416188. PMID 22586274.
- Katsuoka, F (2005). "Genetic evidence that small maf proteins are essential for the activation of antioxidant response element-dependent genes". Mol. Cell. Biol. 25 (18): 8044–51. doi:10.1128/MCB.25.18.8044-8051.2005. PMC 1234339. PMID 16135796.
- Sun, J (2002). "Hemoprotein Bach1 regulates enhancer availability of heme oxygenase-1 gene". EMBO J. 21 (19): 5216–24. doi:10.1093/emboj/cdf516. PMC 129038. PMID 12356737.
- Muto, A (1998). "Identification of Bach2 as a B-cell-specific partner for small maf proteins that negatively regulate the immunoglobulin heavy chain gene 3' enhancer". EMBO J. 17 (19): 5734–43. doi:10.1093/emboj/17.19.5734. PMC 1170901. PMID 9755173.
- Muto, A (2004). "The transcriptional programme of antibody class switching involves the repressor Bach2". Nature. 429 (6991): 566–71. Bibcode:2004Natur.429..566M. doi:10.1038/nature02596. hdl:2241/1881. PMID 15152264. S2CID 4430935.
- Agrawal, SA (2015). "Compound mouse mutants of bZIP transcription factors Mafg and Mafk reveal a regulatory network of non-crystallin genes associated with cataract". Hum. Genet. 134 (7): 717–35. doi:10.1007/s00439-015-1554-5. PMC 4486474. PMID 25896808.
- Yamazaki, H (2012). "Embryonic lethality and fetal liver apoptosis in mice lacking all three small Maf proteins". Mol. Cell. Biol. 32 (4): 808–16. doi:10.1128/MCB.06543-11. PMC 3272985. PMID 22158967.
- Leung, L (2003). "Deficiency of the Nrf1 and Nrf2 transcription factors results in early embryonic lethality and severe oxidative stress". J. Biol. Chem. 278 (48): 48021–9. doi:10.1074/jbc.M308439200. PMID 12968018.
- Martínez-Hernández, A (2014). "Small MAF genes variants and chronic myeloid leukemia". Eur. J. Haematol. 92 (1): 35–41. doi:10.1111/ejh.12211. PMID 24118457.
- Wang, X (2010). "Genetic variation and antioxidant response gene expression in the bronchial airway epithelium of smokers at risk for lung cancer". PLOS ONE. 5 (8): e11934. Bibcode:2010PLoSO...511934W. doi:10.1371/journal.pone.0011934. PMC 2914741. PMID 20689807.
- Mimura, J (2015). "Role of Nrf2 in the pathogenesis of atherosclerosis". Free Radic. Biol. Med. 88 (Pt B): 221–32. doi:10.1016/j.freeradbiomed.2015.06.019. PMID 26117321.
- Yamazaki, H (2015). "Role of the Keap1/Nrf2 pathway in neurodegenerative diseases". Pathol. Int. 65 (5): 210–9. doi:10.1111/pin.12261. PMID 25707882.
- Suzuki, T (2015). "Molecular basis of the Keap1-Nrf2 system". Free Radic. Biol. Med. 88 (Pt B): 93–100. doi:10.1016/j.freeradbiomed.2015.06.006. PMID 26117331.
