Tetramethylethylene
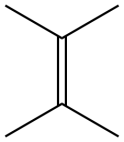
Tetramethylethylene is a hydrocarbon with the formula Me2C=CMe2 (Me = methyl). A colorless liquid, it is the simplest tetrasubstituted alkene.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2,3-Dimethyl-2-butene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.008.422 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H12 | |
| Molar mass | 84.162 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.7075 g/cm3 (at 20 °C) |
| Melting point | −74.6 °C (−102.3 °F; 198.6 K) |
| Boiling point | 73.3 °C (163.9 °F; 346.4 K) |
| Hazards | |
| GHS pictograms |   |
| GHS Signal word | Danger |
| H225, H304 | |
| P210, P233, P240, P241, P242, P243, P280, P301+310, P303+361+353, P331, P370+378, P403+235, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis and reactions
It is prepared by base-catalyzed isomerization of 2,3-dimethyl-1-butene.[1]
Tetramethylethylene forms metal-alkene complexes with low-valent metals and reacts with diborane to give the monoalkyborane known as thexylborane.[2][3]
References
- Hattori, Hideshi (2001). "Solid base catalysts: Generation of basic sites and application to organic synthesis". Applied Catalysis A: General. 222 (1–2): 247–259. doi:10.1016/S0926-860X(01)00839-0.
- Negishi, Ei-Ichi; Brown, Herbert C. (1974). "Thexylborane-A Highly Versatile Reagent for Organic Synthesis via Hydroboration". Synthesis. 1974 (2): 77–89. doi:10.1055/s-1974-23248.
- Giordano, G.; Crabtree, R. H. (1990). "Di-μ-Chloro-Bis(η4 -1,5-Cyclooctadiene)-Dirhodium(I)". Inorganic Syntheses. 28. Event occurs at Di-μ-Chloro-Bis(η4-1,5-Cyclooctadiene)-Dirhodium(I). pp. 88–90. doi:10.1002/9780470132593.ch22. ISBN 9780470132593.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.