Zeolitic imidazolate framework
Zeolitic imidazolate frameworks (ZIFs) are a class of metal-organic frameworks (MOFs) that are topologically isomorphic with zeolites. ZIFs are composed of tetrahedrally-coordinated transition metal ions (e.g. Fe, Co, Cu, Zn) connected by imidazolate linkers. Since the metal-imidazole-metal angle is similar to the 145° Si-O-Si angle in zeolites, ZIFs have zeolite-like topologies.[1] As of 2010, 105 ZIF topologies have been reported in the literature.[2][3] Due to their robust porosity, resistance to thermal changes, and chemical stability, ZIFs are being investigated for applications such as carbon capture.[4]
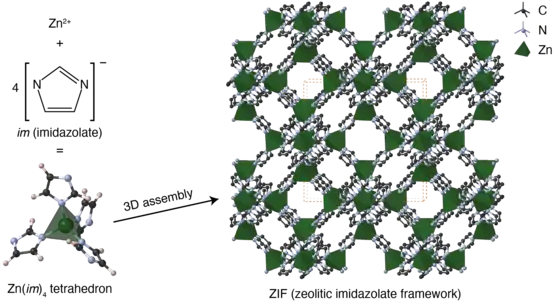
Synthesis
ZIFs are prepared by solvothermal or hydrothermal techniques. Crystals slowly grow from a heated solution of a hydrated metal salt, an ImH (imidazole with acidic proton), a solvent, and base.[5] Functionalized ImH linkers allow for control of ZIF structure.[6] This process is ideal for generating monocrystalline materials for single-crystal X-ray diffraction.[7][8] A wide range of solvents, bases, and conditions have been explored, with an eye towards improving crystal functionality, morphology, and dispersity. Prototypically, an amide solvent such as N,N-dimethylformamide (DMF) is used. The heat applied decomposes the amide solvent to generate amines, which in turn generate the imidazolate from the imidazole species. Methanol,[9][10] ethanol,[11] isopropanol,[12] and water[13][14][15] have also been explored as alternative solvents for ZIF formation but require bases such as pyridine,[16] TEA,[17] sodium formate,[18] and NaOH.[19] Polymers such as poly(ethylene oxide)–poly(propylene oxide)–poly(ethylene oxide),[20] polyvinylpyrrolidone,[21] and poly-(diallyldimethylammonium chloride)[22] have been found to act as crystal dispersants, imparting particle-size and morphology control.
Due to their promising material properties, significant interest lies in economical large-scale production methods. Sonochemical synthesis, which allows nucleation reactions to proceed rapidly through acoustic generation of localized heat and pressure, has been explored as a way to shorten synthesis times.[23][24] As with the case of zeolites, microwave-assisted synthesis has also been of interest for the rapid synthesis of ZIFs.[25][26] Both methods have been shown to reduce reaction times from days to hours, or from hours to minutes. Solvent-free methods, such as ball-milling or chemical vapor deposition, have also been described to produce high-quality ZIF-8.[27][28] Chemical vapor deposition is of particular promise due to the high degree of uniformity and aspect ratio control it can offer, and its ability to be integrated into traditional lithographic workflows for functional thin films (e.g. microelectronics). Environmentally-friendly synthesis based on supercritical carbon dioxide (scCO2) have been also reported as a feasible procedure for the preparation of ZIF-8 at an industrial scale.[29] Working under stoichiometric conditions, ZIF-8 could be obtained in 10 hours and does not require the use of ligand excess, additives, organic solvents or cleaning steps.
Applications of ZIFs
Applications to carbon capture
ZIFs exhibit some properties relevant to carbon capture,[30] while commercial technology still centers around amine solvents.[31]
Zeolites are known to have tunable pores – ranging between 3-12 Angstroms – which allows them to separate carbon dioxide. Because a molecule is about 5.4 Angstroms in length, zeolites with a pore size of 4-5 Angstroms can be well-suited for carbon capture. However, other factors also need to be considered when determining how effective zeolites will be at carbon capture. The first is basicity, which can be created by doing an alkali metal cation exchange. The second is the Si/Al ratio which impacts the cation exchange capacity. To get a higher adsorption capacity, there must be a lower Si/Al ratio in order to increase the cation exchange capacity.
ZIFs 68, 69, 70, 78, 81, 82, 95, and 100 have been found to have very high uptake capacity, meaning that they can store a lot of carbon dioxide, though their affinity to it is not always strong. Of those, 68, 69, and 70 show high affinities for carbon dioxide, evidenced by their adsorption isotherms, which show steep uptakes at low pressures. One liter of ZIF can hold 83 liters of CO
2. This could also be useful for pressure-swing adsorption.[32]
Other separation applications
Much ZIF research focuses on the separation of hydrogen and carbon dioxide because a well-studied ZIF, ZIF-8, has a very high separation factor for hydrogen and carbon dioxide mixtures. It is also very good for the separation of hydrocarbon mixtures, like the following:
- Ethane-propane = 80
- Ethylene- propylene = 10
- Ethylene- propane = 167
In addition to gas separations, ZIF’s have the potential to separate components of biofuels, specifically, water and ethanol. Of all of the ZIF’s that have been tested, ZIF-8 shows high selectivity. ZIF’s have also shown potential in separating other alcohols, like propanol and butanol, from water. Typically, water and ethanol (or other alcohols) are separated using distillation, however ZIF’s offer a potential lower-energy separation option.[33]
Catalysis
ZIF’s also have great potential as heterogeneous catalysts; ZIF-8 has been shown to act as good catalysts for the transesterification of vegetable oils, the Friedel-Crafts acylation reaction between benzoyl chloride and anisole, and for the formation of carbonates. ZIF-8 nanoparticles can also be used to enhance the performance in the Knoevenagel condensation reaction between benzaldehyde and malononitrile.[34] ZIF’s have also been shown to work well in oxidation and epoxidation reactions; ZIF-9 has been shown to catalyze the aerobic oxidation of tetralin and the oxidation of many other small molecules. It can also catalyze reactions to produce hydrogen at room temperature, specifically the dehydrogenation of dimethylamine borane and NaBH4 hydrolysis.
The table below gives a more comprehensive list of ZIF’s that can act as catalysts for different organic reactions.[2]
| ZIF Material | Additional Materials | Reaction (s) Catalyzed |
|---|---|---|
| ZIF-8 | gold nanoparticles | Oxidation of CO
Oxidation of aldehyde groups |
| ZIF-8 | gold and silver core shell nanoparticles | Reduction of 4-nitrophenol |
| ZIF-8 | gold, silver, and platinum nanoparticles | Oxidation of CO
Hydrogenation of n-hexene |
| ZIF-8 | platinum nanoparticles | Hydrogenation of alkene |
| ZIF-8 | platinum and titanium dioxide nanotubes | Degradation of phenol |
| ZIF-8 | palladium nanoparticles | Aminocarbonylation |
| ZIF-8 | iridium nanoparticles | Hydrogenation of cyclohexene and phenylacetene |
| ZIF-8 | ruthenium nanoparticles | Asymmetric hydrogenation of acetophonone |
| ZIF-8 | iron oxide microspheres | Knoevenagel condensation |
| ZIF-8 | Zn2GeO4 nanorods | Conversion of CO2 |
| ZIF-65 | Molybdenum Oxide | Degradation of methyl orange and orange II dyes |
Sensing and electronic devices
ZIF’s are also good candidates for chemical sensors because of their tunable adsorbance properties. ZIF-8 exhibits sensitivity when exposed to the vapor of ethanol and water mixtures, and this response is dependent on the concentration of ethanol in the mixture.[35] Additionally, ZIF’s are attractive materials for matrices for biosensors, like electrochemical biosensors, for in-vivo electrochemical measurements. They also have potential applications as luminescent probes for the detection of metal ions and small molecules. ZIF-8 luminescence is highly sensitive to , and ions as well as acetone. ZIF nanoparticles can also sense fluorescently tagged single stranded pieces of DNA.[35]
Drug delivery
Because ZIF’s are porous, chemically stable, thermally stable, and tunable, they are potentially a platform for drug delivery and controlled drug release. ZIF-8 is very stable in water and aqueous sodium hydroxide solutions but decompose quickly in acidic solutions, indicating a pH sensitivity that could aid in the development of ZIF-based drug-release platforms.[35]
Comparing ZIFs with other compounds
ZIFS vs MOFs
While ZIFs are a subset of the MOF hybrids that combine organic and metal frameworks to create hybrid microporous and crystalline structures, they are much more restricted in their structure. Similar to MOFs, most ZIF properties are largely dependent on the properties of the metal clusters, ligands, and synthesis conditions in which they were created.[36]
Most ZIF alterations up to this point have involved changing the linkers — bridging O2 — anions and imizazolate-based ligands[31] - or combining two types of linkers to change bond angles or pore size due to limitations in synthesizing methods and production.[37] A large portion of changing linkers included adding functional groups with various polarities and symmetries to the imidazolate ligands to alter the ZIFs carbon dioxide adsorption ability without changing the transitional-metal cations.[38] Compare this to MOFs, which have a much larger degree of variety in the types of their building units.
Despite these similarities with other MOFs, ZIFs have significant properties that distinguish these structures as uniquely applicable to carbon capture processes. Because ZIFs tend to resemble the crystalline framework of zeolites, their thermal and chemical stability are higher than those of other MOFs, allowing them to work at a wider range in temperatures, making them suitable to chemical processes.[36]
Perhaps the most important difference is the ZIFs' hydrophobic properties and water stability. A main issue with zeolites and MOFs, to a certain extent, was their adsorption of water along with CO
2. Water vapor is often found in carbon-rich exhaust gases, and MOFs would absorb the water, lowering the amount of CO
2 required to reach saturation.[36] MOFs are also less stable in moist and oxygen-rich environments due to metal-oxygen bonds performing hydrolysis. ZIFs, however, have nearly identical performance in dry vs humid conditions, showing much higher CO
2 selectivity over water, allowing the adsorbent to store more carbon before saturation is reached.[37]
ZIFs vs commercially available products
Even in comparison with other materials, the ZIFs most attractive quality is still its hydrophobic properties. When compared to ZIFs in dry conditions, activated carbon was nearly identical with its uptake capacity.[37] However, once the conditions were changed to wet, the activated carbon’s uptake was halved. When this saturation and regeneration tests were run at these conditions, ZIFs also showed minimal to no structural degradation, a good indication of the adsorbent’s re-usability.[37]
However, ZIFs tend to be expensive to synthesize. MOFs require synthesis methods with long reaction periods, high pressures, and high temperatures, which aren’t methods that are easy to scale-up.[36] ZIFs do tend to be more affordable than commercially available non-ZIF MOFs.
When combined with polymer-sorbent materials, research determined that hybrid polymer-ZIF sorbent membranes no longer following the upper bound of the Robeson plot, which is a plot of selectivity as a function of permeation for membrane gas separation.[31]
See also
References
- Park, KS; et al. (2006). "Exceptional chemical and thermal stability of zeolitic imidazolate frameworks" (PDF). PNAS. 103 (27): 10186–10191. Bibcode:2006PNAS..10310186P. doi:10.1073/pnas.0602439103. PMC 1502432. PMID 16798880.
- Phan, A.; Doonan, C. J.; Uribe-Romo, F. J.; et al. (2010). "Synthesis, Structure, and Carbon Dioxide Capture Properties of Zeolitic Imidazolate Frameworks". Acc. Chem. Res. 43: 58–67. doi:10.1021/ar900116g. PMID 19877580.
- Zhang, J.-P.; Zhang, Y.-B.; Lin, J.-B.; Chen, X.-M. (2012). "Metal Azolate Frameworks: From Crystal Engineering to Functional Materials". Chem. Rev. 112: 1001–1033. doi:10.1021/cr200139g.
- Yaghi, Omar M. (January 2010). "Synthesis, Structure, and Carbon Dioxide Capture Properties of Zeolitic Imidazolate Frameworks" (PDF). Accounts of Chemical Research. 43 (1): 58–67. doi:10.1021/ar900116g. PMID 19877580.
- Park, Kyo Sung; Ni, Zheng; Côté, Adrien P.; et al. (2006-07-05). "Exceptional chemical and thermal stability of zeolitic imidazolate frameworks". Proceedings of the National Academy of Sciences. 103 (27): 10186–10191. Bibcode:2006PNAS..10310186P. doi:10.1073/pnas.0602439103. ISSN 0027-8424. PMC 1502432. PMID 16798880.
- Hayashi, Hideki; Côté, Adrien P.; Furukawa, Hiroyasu; et al. (2007-07-01). "Zeolite A imidazolate frameworks". Nature Materials. 6 (7): 501–506. Bibcode:2007NatMa...6..501H. doi:10.1038/nmat1927. ISSN 1476-1122. PMID 17529969.
- Banerjee, Rahul; Phan, Anh; Wang, Bo; et al. (2008-02-15). "High-Throughput Synthesis of Zeolitic Imidazolate Frameworks and Application to CO2 Capture". Science. 319 (5865): 939–943. Bibcode:2008Sci...319..939B. doi:10.1126/science.1152516. ISSN 0036-8075. PMID 18276887.
- Wang, Bo; Côté, Adrien P.; Furukawa, Hiroyasu; et al. (2008-05-08). "Colossal cages in zeolitic imidazolate frameworks as selective carbon dioxide reservoirs". Nature. 453 (7192): 207–211. Bibcode:2008Natur.453..207W. doi:10.1038/nature06900. ISSN 0028-0836. PMID 18464739.
- Huang, Xiao-Chun; Lin, Yan-Yong; Zhang, Jie-Peng; Chen, Xiao-Ming (2006-02-27). "Ligand-Directed Strategy for Zeolite-Type Metal–Organic Frameworks: Zinc(II) Imidazolates with Unusual Zeolitic Topologies". Angewandte Chemie International Edition. 45 (10): 1557–1559. doi:10.1002/anie.200503778. ISSN 1521-3773. PMID 16440383.
- Cravillon, Janosch; Münzer, Simon; Lohmeier, Sven-Jare; et al. (2009-04-28). "Rapid Room-Temperature Synthesis and Characterization of Nanocrystals of a Prototypical Zeolitic Imidazolate Framework". Chemistry of Materials. 21 (8): 1410–1412. doi:10.1021/cm900166h. ISSN 0897-4756.
- He, Ming; Yao, Jianfeng; Li, Lunxi; et al. (2013-10-01). "Synthesis of Zeolitic Imidazolate Framework-7 in a Water/Ethanol Mixture and Its Ethanol-Induced Reversible Phase Transition". ChemPlusChem. 78 (10): 1222–1225. doi:10.1002/cplu.201300193. ISSN 2192-6506.
- Bennett, Thomas D.; Saines, Paul J.; Keen, David A.; et al. (2013-05-27). "Ball-Milling-Induced Amorphization of Zeolitic Imidazolate Frameworks (ZIFs) for the Irreversible Trapping of Iodine". Chemistry – A European Journal. 19 (22): 7049–7055. doi:10.1002/chem.201300216. ISSN 1521-3765. PMID 23576441.
- Pan, Yichang; Liu, Yunyang; Zeng, Gaofeng; et al. (2011-02-01). "Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system". Chemical Communications. 47 (7): 2071–3. doi:10.1039/C0CC05002D. ISSN 1364-548X. PMID 21206942.
- Tanaka, Shunsuke; Kida, Koji; Okita, Muneyuki; et al. (2012-10-05). "Size-controlled Synthesis of Zeolitic Imidazolate Framework-8 (ZIF-8) Crystals in an Aqueous System at Room Temperature". Chemistry Letters. 41 (10): 1337–1339. doi:10.1246/cl.2012.1337. ISSN 0366-7022.
- Kida, Koji; Okita, Muneyuki; Fujita, Kosuke; et al. (2013-02-07). "Formation of high crystalline ZIF-8 in an aqueous solution". CrystEngComm. 15 (9): 1794. doi:10.1039/C2CE26847G. ISSN 1466-8033.
- Yang, Tingxu; Chung, Tai-Shung (2013-04-23). "Room-temperature synthesis of ZIF-90 nanocrystals and the derived nano-composite membranes for hydrogen separation". Journal of Materials Chemistry A. 1 (19): 6081. doi:10.1039/C3TA10928C. ISSN 2050-7496.
- "Solvothermal synthesis of mixed-ligand metal–organic framework ZIF-78 with controllable size and morphology". ResearchGate. Retrieved 2017-05-01.
- Cravillon, Janosch; Schröder, Christian A.; Bux, Helge; et al. (2011-12-12). "Formate modulated solvothermal synthesis of ZIF-8 investigated using time-resolved in situ X-ray diffraction and scanning electron microscopy". CrystEngComm. 14 (2): 492–498. doi:10.1039/C1CE06002C. ISSN 1466-8033.
- Peralta, David; Chaplais, Gérald; Simon-Masseron, Angélique; Barthelet, Karin; Pirngruber, Gerhard D. (2012-05-01). "Synthesis and adsorption properties of ZIF-76 isomorphs" (PDF). Microporous and Mesoporous Materials. 153: 1–7. doi:10.1016/j.micromeso.2011.12.009.
- Yao, Jianfeng; He, Ming; Wang, Kun; et al. (2013-04-16). "High-yield synthesis of zeolitic imidazolate frameworks from stoichiometric metal and ligand precursor aqueous solutions at room temperature". CrystEngComm. 15 (18): 3601. doi:10.1039/C3CE27093A. ISSN 1466-8033.
- Shieh, Fa-Kuen; Wang, Shao-Chun; Leo, Sin-Yen; Wu, Kevin C.-W. (2013-08-19). "Water-Based Synthesis of Zeolitic Imidazolate Framework-90 (ZIF-90) with a Controllable Particle Size". Chemistry – A European Journal. 19 (34): 11139–11142. doi:10.1002/chem.201301560. ISSN 1521-3765. PMID 23832867.
- Nune, Satish K.; Thallapally, Praveen K.; Dohnalkova, Alice; et al. (2010-06-29). "Synthesis and properties of nano zeolitic imidazolate frameworks". Chemical Communications. 46 (27): 4878–80. doi:10.1039/C002088E. ISSN 1364-548X. PMID 20585703.
- Seoane, Beatriz; Zamaro, Juan M.; Tellez, Carlos; Coronas, Joaquin (2012-04-02). "Sonocrystallization of zeolitic imidazolate frameworks (ZIF-7, ZIF-8, ZIF-11 and ZIF-20)". CrystEngComm. 14 (9): 3103. doi:10.1039/C2CE06382D. ISSN 1466-8033.
- Cho, Hye-Young; Kim, Jun; Kim, Se-Na; Ahn, Wha-Seung (2013-03-15). "High yield 1-L scale synthesis of ZIF-8 via a sonochemical route". Microporous and Mesoporous Materials. 169: 180–184. doi:10.1016/j.micromeso.2012.11.012.
- Bux, Helge; Liang, Fangyi; Li, Yanshuo; et al. (2009). "Zeolitic Imidazolate Framework Membrane with Molecular Sieving Properties by Microwave-Assisted Solvothermal Synthesis". Journal of the American Chemical Society. 131 (44): 16000–16001. doi:10.1021/ja907359t. PMID 19842668.
- Hillman, Febrian; Zimmerman, John M.; Paek, Seung-Min; et al. (2017-03-28). "Rapid microwave-assisted synthesis of hybrid zeolitic–imidazolate frameworks with mixed metals and mixed linkers". Journal of Materials Chemistry A. 5 (13): 6090–6099. doi:10.1039/C6TA11170J. ISSN 2050-7496.
- Bennett, Thomas D.; Cao, Shuai; Tan, Jin Chong; et al. (2011). "Facile Mechanosynthesis of Amorphous Zeolitic Imidazolate Frameworks". Journal of the American Chemical Society. 133 (37): 14546–14549. doi:10.1021/ja206082s. PMID 21848328.
- Stassen, Ivo; Styles, Mark; Grenci, Gianluca; et al. (2016-03-01). "Chemical vapour deposition of zeolitic imidazolate framework thin films". Nature Materials. 15 (3): 304–310. Bibcode:2016NatMa..15..304S. doi:10.1038/nmat4509. ISSN 1476-1122. PMID 26657328.
- López-Domínguez, Pedro; López-Periago, Ana M.; Fernández-Porras, Francisco J.; et al. (2017-03-01). "Supercritical CO2 for the synthesis of nanometric ZIF-8 and loading with hyperbranched aminopolymers. Applications in CO2 capture". Journal of CO2 Utilization. 18: 147–155. doi:10.1016/j.jcou.2017.01.019.
- Venna, Surendar R.; Carreon, Moises A. (2010-01-13). "Highly Permeable Zeolite Imidazolate Framework-8 Membranes for CO2/CH4 Separation". Journal of the American Chemical Society. 132 (1): 76–78. doi:10.1021/ja909263x. ISSN 0002-7863. PMID 20014839.
- Smit, Bernard; Reimer, Jeffrey A.; Oldenburg, Curtis M.; Bourg, Ian C. (2014). Introduction to Carbon Capture and Sequestration (1 ed.). Hackensack, NJ: Imperial College Press. ISBN 978-1-78326-328-8.
- Phan, Anh; Doonan, Christian J.; Uribe-Romo, Fernando J.; et al. (2010-01-19). "Synthesis, structure, and carbon dioxide capture properties of zeolitic imidazolate frameworks". Accounts of Chemical Research. 43 (1): 58–67. doi:10.1021/ar900116g. ISSN 1520-4898. PMID 19877580.
- Zhang, Kang; Nalaparaju, Anjaiah; Chen, Yifei; Jiang, Jianwen (2014-04-23). "Biofuel purification in zeolitic imidazolate frameworks: the significant role of functional groups". Physical Chemistry Chemical Physics. 16 (20): 9643–55. Bibcode:2014PCCP...16.9643Z. doi:10.1039/C4CP00739E. ISSN 1463-9084. PMID 24727907.
- Guan, Yebin; Shi, Juanjuan; Xia, Ming; et al. (2017-11-30). "Monodispersed ZIF-8 particles with enhanced performance for CO2 adsorption and heterogeneous catalysis". Applied Surface Science. 423: 349–353. Bibcode:2017ApSS..423..349G. doi:10.1016/j.apsusc.2017.06.183.
- Chen, Binling; Yang, Zhuxian; Zhu, Yanqiu; Xia, Yongde (2014-09-23). "Zeolitic imidazolate framework materials: recent progress in synthesis and applications". Journal of Materials Chemistry A. 2 (40): 16811–16831. doi:10.1039/C4TA02984D. ISSN 2050-7496.
- Basnayake, Sajani A.; Su, Jie; Zou, Xiadong; Balkus, Kenneth J. (2015-02-04). "Carbonate-Based Zeolitic Imidazolate Frame for Highly Selective CO2 Capture". Inorganic Chemistry. 54 (4): 1816–1821. doi:10.1021/ic5027174. PMID 25650775.
- Nguyen, Nhung T. T.; Lo, Tien N. H.; Kim, Jaheon (2016-04-04). "Mixed-Metal Zeolitic Imidazolate Frameworks and their Selective Capture of Wet Carbon Dioxide over Methane" (PDF). Inorganic Chemistry. 55 (12): 6201–6207. doi:10.1021/acs.inorgchem.6b00814. PMID 27248714.
- Wang, Sibo; Wang, Xinchen (2015-12-08). "Imidazolium Ionic Liquids, Imidazolylidene Heterocyclic Carbenes, and Zeolitic Imidazolate Frameworks for CO2 Capture and Photochemical Reduction". Angewandte Chemie. 55 (7): 2308–2320. doi:10.1002/anie.201507145. PMID 26683833.