Bisoctrizole
Bisoctrizole (INN[1]/USAN,[2] marketed by BASF as Tinosorb M, by DSM Nutritional Products as Parsol Max , by Everlight Chemical as Eversorb M, and by MPI as Milestab 360, INCI methylene bis-benzotriazolyl tetramethylbutylphenol) is a phenolic benzotriazole that is added to sunscreens to absorb UV rays.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
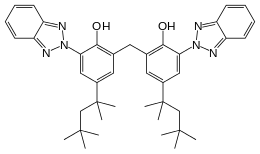
2,2′-methanediylbis[6-(2H-benzotriazol-2-yl)-4-(2,4,4-trimethylpentan-2-yl)phenol] | |
| Other names
UV-360 | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.100.550 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C41H50N6O2 | |
| Molar mass | 658.88 g/mol |
| Melting point | 195.7 °C (384.3 °F; 468.8 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Bisoctrizole is a broad-spectrum ultraviolet radiation absorber, absorbing UVB as well as UVA rays. It also reflects and scatters UV. Bisoctrizole is a hybrid UV absorber, an organic UV filter produced in microfine organic particles (< 200 nm),[3][4] like microfine zinc oxide and titanium dioxide. Where other organic UV absorbers need to be dissolved in either the oil or water phase, bisoctrizole dissolves poorly in both and is applied as invisible particle.
Bisoctrizole is added to the water phase of a sunscreen as a 50% suspension, whereas mineral micropigments are usually added to the oil phase. The bisoctrizole particles are stabilized by the surfactant decyl glucoside.
Bisoctrizole shows very little photodegradation and has a stabilizing effect on other UV absorbers, octyl methoxycinnamate (octinoxate) in particular.
When formulated into a sunscreen, bisoctrizole has minimal skin penetration[5] and it shows no estrogenic effects in vitro.[6]
Bisoctrizole is not approved by the U.S. Food and Drug Administration (FDA), but is approved in the EU and other parts of the world.[7][8][9]
References
- "International Nonproprietary Names for Pharmaceutical Substances (INN)". World Health Organization.
- "Archived copy". Archived from the original on 2007-09-29. Retrieved 2007-08-19.CS1 maint: archived copy as title (link)
- Ciba TINOSORB M
- Herzog, B.; Mongiat, S.; Deshayes, C.; Neuhaus, M.; Sommer, K.; Mantler, A. (2002). "In vivo and in vitro assessment of UVA protection by sunscreen formulations containing either butyl methoxy dibenzoyl methane, methylene bis-benzotriazolyl tetramethylbutylphenol, or microfine ZnO". International Journal of Cosmetic Science. 24 (3): 170–85. doi:10.1046/j.1467-2494.2002.00137.x. PMID 18498509.
- Mavon A, Miquel C, Lejeune O, Payre B, Moretto P (2007). "In vitro percutaneous absorption and in vivo stratum corneum distribution of an organic and a mineral sunscreen". Skin Pharmacol Physiol. 20 (1): 10–20. doi:10.1159/000096167. PMID 17035717.
- Ashby J, Tinwell H, Plautz J, Twomey K, Lefevre PA (December 2001). "Lack of binding to isolated estrogen or androgen receptors, and inactivity in the immature rat uterotrophic assay, of the ultraviolet sunscreen filters Tinosorb M-active and Tinosorb S". Regul Toxicol Pharmacol. 34 (3): 287–91. doi:10.1006/rtph.2001.1511. PMID 11754532.
- Manage Account - Modern Medicine
- CL1976L0768EN0150010.0001 1..107
- Australian Regulatory Guidelines for OTC Medicines - Chapter 10 Archived 2007-08-31 at the Wayback Machine
External links
- Ciba® TINOSORB® M – Microfine UV absorber with Triple Action
- http://www.dermatologytimes.com/dermatologytimes/article/articleDetail.jsp?id=159652
- NEW-WAVE SUNSCREENS – Active ingredient makers are frustrated by the long list of sunscreens and UV-A testing protocols that are still awaiting FDA decisions, C&EN Cover Story
- https://www.fda.gov/ohrms/dockets/dockets/05n0446/05n-0446-bkg0001-03-Tab-01-vol2.pdf