Centrosome cycle
Centrosomes are the major microtubule organizing centers (MTOC) in mammalian cells.[2] Failure of centrosome regulation can cause mistakes in chromosome segregation and is associated with aneuploidy. A centrosome is composed of two orthogonal cylindrical proteins, called centrioles, which are surrounded by an electron and protein dense amorphous cloud of pericentriolar material (PCM).[3] The PCM is essential for nucleation and organization of microtubules.[3] The centrosome cycle is important to ensure that daughter cells receive a centrosome after cell division. As the cell cycle progresses, the centrosome undergoes a series of morphological and functional changes. Initiation of the centrosome cycle occurs early in the cell cycle in order to have two centrosomes by the time mitosis occurs.
Since the centrosome organizes the microtubules of a cell, t has to do with the formation of the mitotic spindle, polarity and, therefore, cell shape, as well as all other processes having to do with the mitotic spindle.[2] The centriole is the inner core of the centrosome, and its conformation is typically somewhat like that of spokes on a wheel. It has a somewhat different conformation amount different organisms, but its overall structure is similar. Plants, on the other hand, do not typically have centrioles.[4]
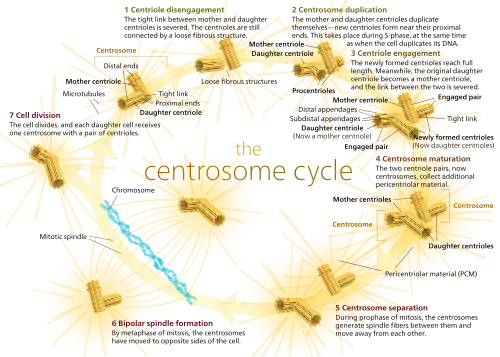
The centrosome cycle consists of four phases that are synchronized to the cell cycle. These include: centrosome duplication during the G1 phase and S Phase, centrosome maturation in the G2 phase, centrosome separation in the mitotic phase, and centrosome disorientation in the late mitotic phase—G1 phase.
Centriole synthesis
Centrioles are generated in new daughter cells through duplication of pre-existing centrioles in the mother cells. Each daughter cell inherits two centrioles (one centrosome) surrounded by pericentriolar material as a result of cell division. However, the two centrioles are of different ages. This is because one centriole originates from the mother cell while the other is replicated from the mother centriole during the cell cycle. It is possible to distinguish between the two preexisting centrioles because the mother and daughter centriole differ in both shape and function.[5] For example, the mother centriole can nucleate and organize microtubules, whereas the daughter centriole can only nucleate.
First, procentrioles begin to form near each preexisting centriole as the cell moves from the G1 phase to the S phase.[6][7][8] During S and G2 phases of the cell cycle, the procentrioles elongate until they reach the length of the older mother and daughter centrioles. At this point, the daughter centriole which takes on characteristics of a mother centriole. Once they reach full length, the new centriole and its mother centriole form a diplosome. A diplosome is a rigid complex formed by an orthogonal mother and newly formed centriole (now a daughter centriole) that aids in the processes of mitosis. As mitosis occurs, the distance between mother and daughter centriole increases until, congruent with anaphase, the diplosome breaks down and each centriole is surrounded by its own pericentriolar material.[6]
Centrosome duplication
Cell cycle regulation of centrosome duplication
Centrosomes are only supposed to replicate once in each cell cycle and are therefore highly regulated.[9] The centrosome cycle has been found to be regulated by multiple things, including reversible phosphorylation and proteolysis.[2] It also undergoes specific processes in each step of cell division due to the heavy regulation, which is why the process is so efficient.[9]
Centrosome duplication is heavily regulated by cell cycle controls. This link between the cell cycle and the centrosome cycle is mediated by cyclin-dependent kinase 2 (Cdk2). Cdk2 is a protein kinase (an enzyme) known to regulate the cell cycle.[10] There has been ample evidence [11][12][13][14] that Cdk2 is necessary for both DNA replication and centrosome duplication, which are both key events in S phase. It has also been shown [13][15][16] that Cdk2 complexes with both cyclin A and cyclin E and this complex is critical for centrosome duplication.[10] Three Cdk2 substrates have been proposed to be responsible for regulation of centriole duplication: nucleophosmin (NPM/B23), CP110, and MPS1.[3] Nucleophosmin is only found in unreplicated centrosomes and its phosphorylation by Cdk2/cyclin E removes NPM from the centrosomes, initiating procentriole formation.[17][18] CP110 is an important centrosomal protein that is phosphorylated by both mitotic and interphase Cdk/cyclin complexes and is thought to influence centrosome duplication in the S phase. [19] MPS1 is a protein kinase that is essential to the spindle assembly checkpoint,[19] and it is thought to possibly remodel an SAS6-cored intermediate between severed mother and daughter centrioles into a pair of cartwheel protein complexes onto which procentrioles assemble.[20]
Centrosome maturation
Centrosome maturation is defined as the increase or accumulation of γ-tubulin ring complexes and other PCM proteins at the centrosome.[2] This increase in γ -tubulin gives the mature centrosome greater ability to nucleate microtubules. Phosphorylation plays a key regulatory role in centrosome maturation, and it is thought that Polo-like kinases (Plks) and Aurora kinases are responsible for this phosphorylation. [21] The phosphorylation of downstream targets of Plks and Aurora A lead to the recruitment of γ –tubulin and other proteins that form PCM around the centrioles. [23]
Centrosome separation
In early mitosis, several motor proteins drive the separation of centrosomes. With the onset of prophase, the motor protein dynein provides the majority of the force required to pull the two centrosomes apart. The separation event actually occurs at the G2/M transition and happens in two steps. In the first step, the connection between the two parental centrioles is destroyed. In the second step, the centrosomes are separated via microtubule motor proteins.[2]
Centrosome disorientation
Centrosome disorientation refers to the loss of orthogonality between the mother and daughter centrioles.[2] Once disorientation occurs, the mature centriole begins to move toward the cleave furrow. It has been proposed that this movement is a key step in abscission, the terminal phase of cell division.[21]
Centrosome reduction
Centrosome reduction is the gradual loss of centrosomal components that takes place after mitosis and during differentiation[22] In cycling cells, after mitosis the centrosome has lost most of its pericentriolar material (PCM) and its microtubule nucleation capacity. In sperm, centriole structure is also changed in addition to the loss of PCM and its microtubule nucleation capacity.[23]
Dysregulation of the centrosome cycle
Improper progression through the centrosome cycle can lead to incorrect numbers of centrosomes and aneuploidy, which could eventually lead to cancer. The role of centrosomes in tumor progression is unclear. The mis-expression of genes such as p53, BRCA1, Mdm2, Aurora-A and survivin causes an increase in the amount of centrosomes present in a cell. However, it is not well understood how these genes influence the centrosome or how an increase in centrosomes influences tumor progression.[24]
The Centrosome Cycle and Disease
Issues with the centrosome can have detrimental effects on the cell, which can lead to diseases in the organisms hosting the cells. Cancer is a heavily studied disease that has been found to have a relation to the cell's centrosome.[2] Dwarfism, microcephaly, and ciliopathies have also recently been genetically linked to centrosome proteins.[25]
Centrosomes are believed to be related to cancer due to the fact that they contain tumor suppressor proteins and oncogenes. These proteins have been found to cause detrimental alterations in the centrosome of various tumor cells.[26] There are two main categories of the centrosome alteration: structural and functional. The structural changes can lead to different shapes, sizes, numbers, positions, or composition, while the functional changes can lead to issues with the microtubules and mitotic spindles, therefore becoming detrimental in cell division.[26] Researchers are hopeful that the targeting of carious centrosomal proteins may be a possible treatment to or prevention of cancer.[26]
References
- "Figure 1". Aurora-A: the maker and breaker of spindle poles. Journal of Cell Science. Archived from the original on 11 May 2012. Retrieved 11 December 2012.
- Meraldi P, Nigg EA (June 2002). "The centrosome cycle". FEBS Letters. 521 (1–3): 9–13. doi:10.1016/S0014-5793(02)02865-X. PMID 12067716.
- Loncarek J, Khodjakov A (February 2009). "Ab ovo or de novo? Mechanisms of centriole duplication". Molecules and Cells. 27 (2): 135–42. doi:10.1007/s10059-009-0017-z. PMC 2691869. PMID 19277494.
- Fu J, Hagan IM, Glover DM (February 2015). "The centrosome and its duplication cycle". Cold Spring Harbor Perspectives in Biology. 7 (2): a015800. doi:10.1101/cshperspect.a015800. PMID 25646378.
- Piel M, Nordberg J, Euteneuer U, Bornens M (February 2001). "Centrosome-dependent exit of cytokinesis in animal cells". Science. 291 (5508): 1550–3. Bibcode:2001Sci...291.1550P. doi:10.1126/science.1057330. PMID 11222861.
- Chrétien D, Buendia B, Fuller SD, Karsenti E (November 1997). "Reconstruction of the centrosome cycle from cryoelectron micrographs". Journal of Structural Biology. 120 (2): 117–33. doi:10.1006/jsbi.1997.3928. PMID 9417977.
- Kuriyama R, Borisy GG (December 1981). "Centriole cycle in Chinese hamster ovary cells as determined by whole-mount electron microscopy". The Journal of Cell Biology. 91 (3 Pt 1): 814–21. doi:10.1083/jcb.91.3.814. PMC 2112828. PMID 7328123.
- Vorobjev IA (June 1982). "Centrioles in the cell cycle. I. Epithelial cells". The Journal of Cell Biology. 93 (3): 938–49. doi:10.1083/jcb.93.3.938. PMC 2112136. PMID 7119006.
- Fujita H, Yoshino Y, Chiba N (March 2016). "Regulation of the centrosome cycle". Molecular & Cellular Oncology. 3 (2): e1075643. doi:10.1080/23723556.2015.1075643. PMC 4905396. PMID 27308597.
- "CDK2 cyclin dependent kinase 2 [Homo sapiens (human)]". Gene - NCBI. Retrieved 1 December 2019.
- Hinchcliffe EH, Li C, Thompson EA, Maller JL, Sluder G (February 1999). "Requirement of Cdk2-cyclin E activity for repeated centrosome reproduction in Xenopus egg extracts". Science. 283 (5403): 851–4. Bibcode:1999Sci...283..851H. doi:10.1126/science.283.5403.851. PMID 9933170.
- Matsumoto Y, Hayashi K, Nishida E (April 1999). "Cyclin-dependent kinase 2 (Cdk2) is required for centrosome duplication in mammalian cells". Current Biology. 9 (8): 429–32. doi:10.1016/S0960-9822(99)80191-2. PMID 10226033.
- Meraldi P, Lukas J, Fry AM, Bartek J, Nigg EA (June 1999). "Centrosome duplication in mammalian somatic cells requires E2F and Cdk2-cyclin A". Nature Cell Biology. 1 (2): 88–93. doi:10.1038/10054. PMID 10559879.
- Lacey KR, Jackson PK, Stearns T (March 1999). "Cyclin-dependent kinase control of centrosome duplication". Proceedings of the National Academy of Sciences of the United States of America. 96 (6): 2817–22. Bibcode:1999PNAS...96.2817L. doi:10.1073/pnas.96.6.2817. PMC 15852. PMID 10077594.
- Hinchcliffe EH, Sluder G (September 2001). "Centrosome duplication: three kinases come up a winner!". Current Biology. 11 (17): R698-701. doi:10.1016/S0960-9822(01)00412-2. PMID 11553343.
- Matsumoto Y, Maller JL (October 2004). "A centrosomal localization signal in cyclin E required for Cdk2-independent S phase entry". Science. 306 (5697): 885–8. Bibcode:2004Sci...306..885M. doi:10.1126/science.1103544. PMID 15514162.
- Okuda M, Horn HF, Tarapore P, Tokuyama Y, Smulian AG, Chan PK, et al. (September 2000). "Nucleophosmin/B23 is a target of CDK2/cyclin E in centrosome duplication". Cell. 103 (1): 127–40. doi:10.1016/S0092-8674(00)00093-3. PMID 11051553.
- Tokuyama Y, Horn HF, Kawamura K, Tarapore P, Fukasawa K (June 2001). "Specific phosphorylation of nucleophosmin on Thr(199) by cyclin-dependent kinase 2-cyclin E and its role in centrosome duplication". The Journal of Biological Chemistry. 276 (24): 21529–37. doi:10.1074/jbc.M100014200. PMID 11278991.
- Stucke VM, Silljé HH, Arnaud L, Nigg EA (April 2002). "Human Mps1 kinase is required for the spindle assembly checkpoint but not for centrosome duplication". The EMBO Journal. 21 (7): 1723–32. doi:10.1093/emboj/21.7.1723. PMC 125937. PMID 11927556.
- Pike AN, Fisk HA (April 2011). "Centriole assembly and the role of Mps1: defensible or dispensable?". Cell Division. 6: 9. doi:10.1186/1747-1028-6-9. PMC 3094359. PMID 21492451.
- Rusan NM, Rogers GC (May 2009). "Centrosome function: sometimes less is more". Traffic. 10 (5): 472–81. doi:10.1111/j.1600-0854.2009.00880.x. PMID 19192251.
- Avidor-Reiss T, Khire A, Fishman EL, Jo KH (2015). "Atypical centrioles during sexual reproduction". Frontiers in Cell and Developmental Biology. 3: 21. doi:10.3389/fcell.2015.00021. PMC 4381714. PMID 25883936.
- Khire A, Vizuet AA, Davila E, Avidor-Reiss T (November 2015). "Asterless Reduction during Spermiogenesis Is Regulated by Plk4 and Is Essential for Zygote Development in Drosophila". Current Biology. 25 (22): 2956–63. doi:10.1016/j.cub.2015.09.045. PMC 4654664. PMID 26480844.
- Cunha-Ferreira I, Bento I, Bettencourt-Dias M (May 2009). "From zero to many: control of centriole number in development and disease". Traffic. 10 (5): 482–98. doi:10.1111/j.1600-0854.2009.00905.x. PMID 19416494.
- Nigg EA, Čajánek L, Arquint C (August 2014). "The centrosome duplication cycle in health and disease". FEBS Letters. 588 (15): 2366–72. doi:10.1016/j.febslet.2014.06.030. PMID 24951839.
- Rivera-Rivera Y, Saavedra HI (December 2016). "Centrosome - a promising anti-cancer target". Biologics. 10: 167–176. doi:10.2147/BTT.S87396. PMC 5167523. PMID 28008224.
