Chaetoglobosin A
Chaetoglobosin A is a fungal isolate with anticancer activity in vitro.[1] Derivatives of the compound include MBJ-0038, MBJ-0039, and MBJ-0040.[2]
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C32H36N2O5 | |
| Molar mass | 528.649 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
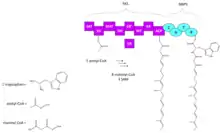
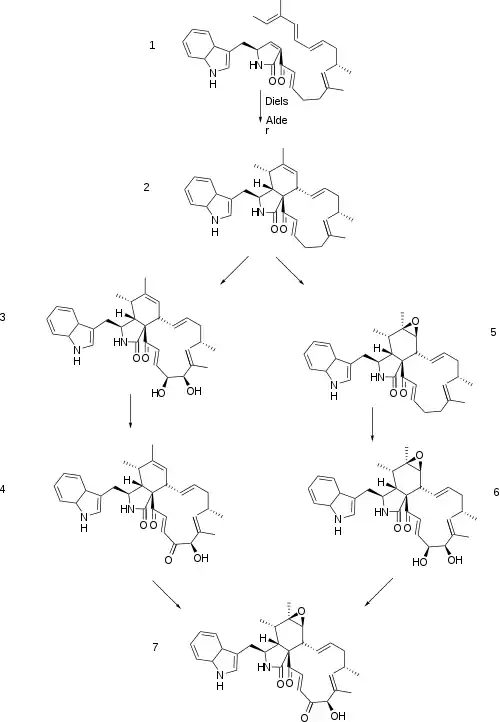
Biosynthesis
Chaetoglobosin A biosynthesis begins with a product from hybrid PKS-NRPS encoded by the gene CHGG_01239, followed by multiple oxidations which form different intermediates depending on the order of functional groups oxidized. The PKS-NRPS product undergoes a diels alder, to form prochaetoglobosin I (2) and is subsequently oxidized in different paths as shown in the scheme. Either the epoxide is created first to form prochaetoglobosin IV (5), followed by di-hydroxylation to form 20-dihydrochaetoglobosin A (6), and a final oxidation of one hydroxyl to ketone to form chaetoglobosin A, or di-hydroxylation of (2) occurs first, forming cytoglobosin D (3), followed by one hydroxyl oxidation to form chaetoglobosin J (4), and lastly epoxidation to form chaetoglobosin A. Epoxidation of (3) can also occur prior to hydroxyl oxidation to form (6). [3]
References
- Sekita, S.; Yoshihara, K.; Kuwano, H. (1973). "Structures of chaetoglobosin A and B, cytotoxic metabolites of Chaetomium globosum". Tetrahedron Lett. 14 (23): 2109–2112. doi:10.1016/S0040-4039(01)86820-9.
- Kawahara, T; Itoh, M; Izumikawa, M; Sakata, N; Tsuchida, T; Shin-Ya, K (2013). "New chaetoglobosin derivatives, MBJ-0038, MBJ-0039 and MBJ-0040, isolated from the fungus Chaetomium sp. f24230". The Journal of Antibiotics. 66 (12): 727–30. doi:10.1038/ja.2013.75. PMID 23881215.
- Ishiuchi, Kan’ichiro; Nakazawa, Takehito; Yagishita, Fumitoshi; Mino, Takashi; Noguchi, Hiroshi; Hotta, Kinya; Watanabe, Kenji (2013). "Combinatorial Generation of Complexity by Redox Enzymes in the Chaetoglobosin A Biosynthesis". Journal of the American Chemical Society. 135 (19): 7371–7377. doi:10.1021/ja402828w. PMID 23611317.