Megachile rotundata
Megachile rotundata, the alfalfa leafcutting bee,[1] is a European bee that has been introduced to various regions around the world. As a solitary bee species, it does not build colonies or store honey, but is a very efficient pollinator of alfalfa, carrots, other vegetables and some fruits. Because of this, farmers often use M. rotundata as a pollination aid by distributing M. rotundata prepupae around their crops.[2] Each female will construct and provision her own nest, which is built in old trees or log tunnels.[3] Being a leafcutter bee, these nests are lined with cut leaves.[4] These bees feed on pollen and nectar and display sexual dimorphism. This species has been known to bite and sting, however it poses no overall danger unless it is threatened or harmed and its sting has been described as half as painful as a honey bee's.[5]
| Megachile rotundata | |
|---|---|
 | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Hymenoptera |
| Family: | Megachilidae |
| Genus: | Megachile |
| Subgenus: | Megachile (Eutricharaea) |
| Species: | M. rotundata |
| Binomial name | |
| Megachile rotundata (Fabricius, 1787) | |
Taxonomy and phylogeny
Megachile rotundata is a member of the subfamily Megachilinae, which includes more than 4000 bee species; this currently makes this family the second largest among all other bee families. This subfamily is one of four other subfamilies of Megachilidae, the other three being Fideliinae, Pararhophitinae, and Lithurginae. Phylogenetic studies suggest that this subfamily is monophyletic.[6] More specifically, it belongs to the genus Megachile, which contains 52 subgenera and 1,478 species. This genus Megachile consists of bees which cut leaf pieces to line their nests.[5]
.jpg.webp)
Description and identification
.jpg.webp)
Megachile rotundata is a European leaf-cutting bee placed in the sub-genus Eutricharia, the "small leaf-cutting bees"; they are 6–9 mm (0.24–0.35 in) in length.[7][8] They are partially bivoltine, meaning that under the right conditions they can produce two generations per year.[9] These bees present a sexual dimorphism, in which the males are smaller than the females and differently marked.[10] M. rotundata bees are a dark grey color. Females have white hairs all over their bodies, including on their scopae. In contrast, males have white and yellow spots on their abdomens.[5]
Distribution and habitat
Megachile rotundata is currently found on all continents except for Antarctica.[6] In North America, the species was deliberately imported to assist in the pollination of food crops, but has now become feral and widespread.[11] In New Zealand and Australia, M. rotundata was also introduced to assist in the pollination of alfalfa (known locally as lucerne), in 1971 in New Zealand,[12][13] and 1987 in Australia.[14][15]
Nest construction
Females construct tubular-shaped nests in a variety of sites including rotting wood, flower stems, reeds, and soda straws. In the wild, females will also create nests in small holes in the ground or in available cracks/crevices in trees or buildings. The nests are composed of a string of individual cells, as many as the space will allow. When managed for pollination, the females are induced to nest in paper cylinders similar to drinking straws or drilled blocks of wood.
Each cell is made from circular disks cut from plant leaves using the bee's mandibles, hence the name "leafcutter".[11] Females use approximately 15 leaves per cell, called a concave bottom, overlapping the leaves to produce the thimble-shaped cell.[3] While the bees do not store honey, females do collect pollen and nectar which they store in the cells of their nests.[11] Specifically, females will first regurgitate the nectar they have provisioned into the cell and then transfer the pollen that is attached to their scopa on top of the nectar.[3] Each cell contains one pollen and nectar ball, and one egg[11] with each cell containing a 2:1 nectar-to-pollen ratio.[3] The completion of one cell in the nest requires between 15 and 20 provisioning trips. After the female lays her eggs, she seals the cell with circular leaf pieces.[3]
Nest dimensions and sex ratio
Studies reveal that positioning of male and female progeny in the nest is strategic and that cell size plays a major role in the size of progeny, independent of the mother's size. Females have been observed to lay female eggs in the inner cells and male eggs in the outer cells. With respect to sex ratios, larger cell provisions are correlated with a greater production of female offspring. Two explanations exist for these behaviors in terms of mother's foraging behaviors: 1) The mother brings more provisions to the inner cells because she expects that female progeny will be produced there and 2) the mother chooses to fertilize her egg, and therefore promotes the production of female progeny, because she has to bring larger provision proportions to a larger cell.[16] The sex ratio changes depending on nest size, length, and nesting material. This ratio is controlled by the female. These observations have been made for females who make their nests in tunnels. For example, a 5.5 mm tunnel diameter is associated with a 3:1 ratio and a 6.0 mm tunnel diameter is associated with a 2:1 ratio of males to females. Shorter tunnels, those that are below 5.0 cm long, are less favorable.[17]
Nest recognition
During nest construction, females transfer olfactory cues onto their nests to facilitate nest recognition. Specifically, they will transfer these hormones by running their abdomens along the nest or excreting liquid from the tip of the abdomen. The olfactory cues are especially concentrated around the nest entrance. When these olfactory cues were experimentally removed, female M. rotundata were unable to identify their own nests, revealing the importance of these chemicals. The chemical composition of these olfactory cues includes hydrocarbons, wax esters, fatty aldehydes, and fatty alcohol acetate esters.[18]
Colony cycle
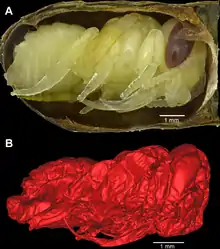
As a member of the Hymenoptera order, this bee is haplodiploid.[19] Adults emerge by the end of the summer through one of two developmental pathways: larvae will develop by the end of one summer and proceed through the a pre-pupal diapause phase until the next summer or larvae, known as "second-generation" bees, will skip this phase and emerge as adults in the same summer.[9]
The larva transitions through four instar stages before emerging as an adult. During its development, which occurs rapidly, the larva consumes the pollen ball and enters diapause when the pollen is fully consumed. In its progression into the diapause phase, the larva will defecate pellets in a ring formation and then spins its cocoon out of silk threads. The next spring, the mature larva pupates, a process that lasts 3–4 weeks, and completes its development. Once the bee is developed, it cuts its way out from the nest by chewing itself out of its cocoon.[3]
Upon emergence, females will mate immediately and begin constructing their nests after one week.[4]
Progeny released via the two alternative pathways for emergence display different sex ratios as well as sizes. Among adults who emerge during the summer of the same year, the sex-ratio is biased towards males. Among the "second-generation bees," however, the sex ratio is female biased. Further, spring-emergent adults weigh more than summer-emergent ("second-generation") adults. These differences have been proposed to be attributed to the chances of survival to mating of the two sexes and the metabolic costs involved in development. Bees that undergo diapause and emerge in the spring must endure the long winter, and thus require more food stores. As a result, they will be larger when they mature. Another explanation has been that smaller bees mature faster and thus are able to mate more quickly when they emerge in the summer to avoid the cold, harsh conditions of the winter.[20]
The sex ratio of the offspring also depends on the distance between nesting and foraging sites. Females have been observed and determined to bias their offspring sex ratio to males at larger flight distances from the nest.[10]
Kin recognition and fratricide
Research on second-generation bees illustrates that kin recognition of nest mates is not a genetically-based behavior. Further, fratricide has been hypothesized to be a non-discriminatory behavior; emerging individuals treat developing siblings and non-siblings similarly. When developing in cells behind diapausing bees, inner-nesting bees either bypass the nest in front of them, retreat back to their nests until diapausing individuals emerge, or chew through the nesting cells in front of them, killing the diapausing bees. These decisions are contingent on the extent of development of delay of nestmates. For example, emerging individuals would remain in their cells when they were delayed from emerging for a relatively short period of time. When delayed for longer periods of times, however, emerging individuals would use their mandibles to destroy the cells of their nest mates. With respect to gender differences, males have been observed to bypass nests more frequently than females due to the small size of males.[21]
Sexual behavior
Mating
Magachile rotundata has been found to be a monandrous bee species.[22] During the mating season, males attempt to obtain mates by positioning themselves at sites where female are likely to be, including foraging sites and nests. While females can mate several times, they resist male advances by restricting their mating to one sexual interaction and fleeing from these males. This behavior is attributed to female productivity during the construction of her nest and egg-laying. When harassed, females are unable to build their nests efficiently, making less foraging trips and spending a longer time overall in nest construction. Fleeing allows females to avoid being mounted. In the process of fleeing, however, females may lose their nesting materials, such as leaves and must then make an additional trip to make up for the lost materials.[19]
Sexual pheromones
Sexual pheromones in epicuticular waxes are released by females to assist in attracting males as mates. Studies of the emissions from these waxes reveal that alkenes are the primary compound detected by males. Further, females of different ages emit different pheromones, containing different alkene compositions. In young females, substances that classify as either 7-pentacosene or 9-pentacosene are present in the highest concentrations and, in older females, 5-monoene compounds predominate. During mating season, males are able to detect these pheromones and preferentially select young females to copulate with.[23]
Interaction with other species
Diet
M. rotundata can feed on nectar and pollen from a variety of plants[24] but prefer Medicago sativa.[5] Females will immediately begin feeding after emergence during the maturation period of their eggs.[4] During feeding, the bee will insert its proboscis into the keel of the plant. In the process, pollen is brushed onto its scopa.[5]

Predators
Currently, there are 28 species known to prey on M. rotundata, all of which have unique ways of invading and destroying their host nests. Some species include Trogoderma glabrum, Trogoderma variabile, Tribolium audax, Tribolium brevicornis and Trichodes ornatus. T. ornatus is the most formidable predator, with females laying their eggs in cracks between leaf pieces. During development, T. ornatus larvae will kill host bee larvae and consume pollen from nest provisions. When it reaches the 3rd stage of development, it will burrow into adjacent cells and consume host larvae irrespective of host larvae instar stage. In another case, female T. glabrum will lays her eggs in cracks between cells at the back of the nest. As they develop, the larvae will begin to consume host larvae, moving towards the front of the nest and eating females first. In a third case, T. variabile attack M. rotundata when it is in its cocoon or the pupae stage.[25]
Parasites
Chalcid wasps are the primary parasites of M. rotundata. Some species include Pteromalus venustus, Monodontomerus obscurus, Melittobia chalybii, Diachys confusus,[3] and Sapyga pumila.[4] The most formidable predator of this species is P. venustus, a parasitoid wasp in which the female will use its ovipositor to sting a developing M. rotundata larva or pupa.[3] After stinging to paralyze its host, the female lays her eggs on the host's surface and the development of the parasite offspring takes place. After about 15–20 weeks, adult P. venustus emerge.[3]
Other parasites include several species of Coelioxys and Nemognatha lutea. Coelioxys takes advantages of instances when female M. rotundata are away from the nest, such as during foraging, to lay its egg in the host nest cells. Once Coelioxys reaches the larval stage, it will kill the developing, host larva and consume all of the provisions that the host female placed in the nest. N. lutea, in contrast, initiate parasitism of hosts at foraging sites. Specifically, female N. lutea will lay their eggs on flowers and, when a bee arrives at this flower during provisioning, will secure itself, as a triungulin stage, to the bee. The triungulin will then, upon the host bee's arrival to its nest, remove itself from the bee, entering a cell and consuming all the provisions gathered by the host female.[3]
Mating
Males of the species may, presumably by mistake, attempt to mate with other bee species which are using the same foraging areas as female leaf cutters. They follow several inches behind their intended partner, whether a leaf cutter or bumble bee, which is moving around a plant seeking a flower; then when the target does alight, the male leaf cutter darts rapidly onto the target's back. This causes bumble bees and nonreceptive female leaf cutters to move to another flower and in the process shed the male leaf cutter. The male may then move right to the next prospect, or may first stop for a sip of nectar.
Disease
Chalkbrood disease in M. rotundata, specifically larvae, was discovered in 1974 in Nevada. This disease occurs from exposure of M. rotundata to Ascosphaera aggregata, which is introduced into the nest cells through nectar and pollen provisions that females bring back from foraging visits.[26] Signs of chalkbrood infection have been observed to occur in the fifth instar larva, leading to the milky appearance of the larval hemolymph and development of a pink, tan, or gray cast in the head or abdominal region. These changes are accompanied by the spread of pink, tan, or gray color throughout the body of the larva and dark-colored, fungal cysts appear under the cuticle.[27]
A variety of microorganisms have been isolated from the alimentary canals and frass of M. rotundata. Bacteria include Bacillus firmus, B. licheniformis, B. megaterium, B. pumilus, and Streptomyces and fungus includes Trichosporonoides megachiliensis.[10] With respect to the development of chalkbrood, it has been suggested that the bacteria and fungi promote the inhabitation of A. aggregata. This is supported by observations in chalkbrood diseased larvae, which contain higher amounts of bacteria and fungi in their gut than in healthy larvae.[26]
In controlling chalkbrood, such methods as decontamination of nest cells and materials and shelters was initially achieved using household bleach. Current methods include paraformaldehyde fumigation.[3]
Defense
Female alfalfa leafcutter bees have stingers, but both sexes will usually use their mandibles as a defensive mechanism, usually only defending themselves when squeezed or antagonized. Therefore, bee suits, such as those required with honey bees, are not necessary when dealing with these bees. When these bees do sting, however, they do not lose their stingers or die after stinging.[5]
Human importance
Human assistance in pest control
In managing M. rotundata for pollination, such methods as dichlorvos pest strips and ultraviolet lamps placed above liquid traps are effective in killing parasitoids but not harming M. rotundata.[4] Other methods of preventing parasitoid invasion include physical barriers. For example, the presence of thick artificial cavities separating the exterior from the nest and application of a felt cloth sealing to the back of the nesting board prevents parasitoid access to the nest.[3] Another way that pest are controlled is by opening up the nesting material and cleaning the cocoons.[28]
Pollination

M. rotundata was intentionally introduced into the United States during the 1940s, and its management as a pollinator leads to increases in seed production of some crops in the U.S. It has been shown to pollinate the following plant species:[4]
Such behaviors as gregarious nesting, utilization of leaves and nesting materials that have been mass-produced by humans, efficient pollination of M. sativa, and synchronous emergence of adult bees during alfalfa blooming period provide positive benefits for the use of these bees in agricultural development.
When these bees are supplied to farmers for pollinating crops, they are usually supplied in a dormant state called pre-pupa, and kept in such state by a constant temperature of about 7 °C(44.6 °F). At a time that is appropriate for the crop's flowers, the farmer puts the pre-pupal form in an incubating environment, a constant temperature of about 27 °C(80.6 °F) The adult bees emerge from the pupal form after about 25 days at that temperature. Then, the farmer brings the bees to the field.[2][29]
See also
References
- "ITIS Bees: World Bee Checklist". Species 2000 & ITIS Catalogue of Life, 2019 Annual Checklist. Integrated Taxonomic Information System. 2019. Retrieved 2019-06-20.
- "How to Manage Alfalfa Leafcutting Bees", a publication of the University of Maine Agricultural Cooperative Extension, year 2007.
- Goettel, Mark S. (2008-01-01). "Alfalfa Leafcutting Bee, Megachile rotundata (Hymenoptera: Megachilidae)". In Capinera, John L. (ed.). Encyclopedia of Entomology. Springer Netherlands. pp. 98–101. doi:10.1007/978-1-4020-6359-6_135. ISBN 978-1-4020-6242-1.
- Pitts-Singer, Theresa L.; Cane, James H. (2011-01-01). "The Alfalfa Leafcutting Bee, Megachile rotundata: The World's Most Intensively Managed Solitary Bee". Annual Review of Entomology. 56 (1): 221–237. doi:10.1146/annurev-ento-120709-144836. PMID 20809804.
- "Megachile rotundata (alfalfa leafcutting bee)". Animal Diversity Web. Retrieved 2015-10-15.
- Gonzalez, Victor H.; Griswold, Terry; Praz, Christophe J.; Danforth, Bryan N. (2012-04-01). "Phylogeny of the bee family Megachilidae (Hymenoptera: Apoidea) based on adult morphology" (PDF). Systematic Entomology. 37 (2): 261–286. doi:10.1111/j.1365-3113.2012.00620.x. ISSN 1365-3113.
- https://bugguide.net/node/view/210925 BugGuide, Megachille rotundata
- "Megachile rotundata (Fabricius, 1787) Alfalfa Leafcutting Bee". Discover Life. Retrieved 9 January 2016.
- Pitts-Singer, Theresa L.; Bosch, Jordi (2010). "Nest Establishment, Pollination Efficiency, and Reproductive Success of Megachile rotundata (Hymenoptera: Megachilidae) in Relation to Resource Availability in Field Enclosures". Environmental Entomology. 39 (1): 149–158. doi:10.1603/en09077. PMID 20146851.
- Peterson, Jason H.; Roitberg, Bernard D.; Peterson, J. H. (2005-10-22). "Impacts of flight distance on sex ratio and resource allocation to offspring in the leafcutter bee, Megachile rotundata". Behavioral Ecology and Sociobiology. 59 (5): 589–596. doi:10.1007/s00265-005-0085-9. ISSN 0340-5443.
- Milius, Susan (January 6, 2007). "Most Bees Live Alone: No hives, no honey, but maybe help for crops". Science News. 171 (1): 11–3. doi:10.1002/scin.2007.5591710110.
- "Introduced Bees in New Zealand" Archived 2010-05-27 at the Wayback Machine, New Zealand Entomologist Vol 33: 92-101 (February 2010)
- "Selection and importation of new pollinators to New Zealand" Archived 2008-10-15 at the Wayback Machine, B. J. Donovan, DSIR Plant Protection
- "There have been several importations of the alfalfa leafcutting bee, beginning in 1987...", Australian Honey Industry Monthly Review, January 2001
- "Introducing the Alfalfa Leafcutting Bee into Australia - A Case Study, R.M. Bitner and S.S. Peterson"
- Klostermeyer, E. C.; Mech, Stephen J.; Rasmussen, Wm. B. (1973). "Sex and Weight of Megachile rotundata (Hymenoptera: Megachilidae) Progeny Associated with Provision Weights". Journal of the Kansas Entomological Society. 46 (4): 536–548. JSTOR 25082604.
- Stephen, W. P.; Osgood, Charles E. (1965-10-01). "Influence of Tunnel Size and Nesting Medium on Sex Ratios in a Leaf-Cutter Bee, Megachile rotundata". Journal of Economic Entomology. 58 (5): 965–968. doi:10.1093/jee/58.5.965. ISSN 0022-0493.
- Guédot, Christelle; Buckner, James S.; Hagen, Marcia M.; Bosch, Jordi; Kemp, William P.; Pitts-Singer, Theresa L. (2013). "Nest Marking Behavior and Chemical Composition of Olfactory Cues Involved in Nest Recognition in Megachile rotundata". Environmental Entomology. 42 (4): 779–789. doi:10.1603/en13015. PMID 23905742.
- Rossi, Benjamin H.; Nonacs, Peter; Pitts-Singer, Theresa L. (2010-01-01). "Sexual harassment by males reduces female fecundity in the alfalfa leafcutting bee, Megachile rotundata". Animal Behaviour. 79 (1): 165–171. doi:10.1016/j.anbehav.2009.10.023.
- Tepedino, V. J.; Parker, F. D. (1988-05-01). "Alternation of Sex Ratio in a Partially Bivoltine Bee, Megachile rotundata (Hymenoptera: Megachilidae)". Annals of the Entomological Society of America. 81 (3): 467–476. doi:10.1093/aesa/81.3.467. ISSN 0013-8746.
- Tepedino, V. J.; Frohlich, D. R. (1984-01-01). "Fratricide in Megachile rotundata, a Non-Social Megachilid Bee: Impartial Treatment of Sibs and Non-Sibs". Behavioral Ecology and Sociobiology. 15 (1): 19–23. doi:10.1007/bf00310210. JSTOR 4599689.
- Paxton, Robert John (2005). "Male mating behaviour and mating systems of bees: an overview" (PDF). Apidologie. 36 (2): 145–156. doi:10.1051/apido:2005007.
- Paulmier, Ivan; Bagnères, Anne-Geneviève; Afonso, Carlos M. M.; Dusticier, Georges; Rivière, Germaine; Clément, Jean-Luc (1999-03-01). "Alkenes as a Sexual Pheromone in the Alfalfa Leaf-Cutter Bee Megachile rotundata". Journal of Chemical Ecology. 25 (3): 471–490. doi:10.1023/A:1020993518226. ISSN 0098-0331.
- O'Neill, Kevin M.; O'Neill, Ruth P.; Blodgett, Sue; Fultz, Jessica (2004). "Composition of Pollen Loads of Megachile rotundata in Relation to Flower Diversity (Hymenoptera: Megachilidae)". Journal of the Kansas Entomological Society. 77 (4): 619–625. doi:10.2317/E-24.1. JSTOR 25086246.
- Eves, Jack D.; Mayer, Daniel F. (1980). Parasite, predators, and nest destroyers of the alfalfa leafcutting bee, Megachile rotundata. Western Regional Extension Publication.
- Inglis, G. Douglas; Sigler, Lynne; Goette, Mark S. (1993-09-01). "Aerobic microorganisms associated with alfalfa leafcutter bees (Megachile rotundata)". Microbial Ecology. 26 (2): 125–143. doi:10.1007/BF00177048. ISSN 0095-3628. PMID 24190009.
- Vandenberg, John D.; Stephen, W. P. (1982-03-01). "Etiology and symptomatology of chalkbrood in the alfalfa leafcutting bee, Megachile rotundata". Journal of Invertebrate Pathology. 39 (2): 133–137. doi:10.1016/0022-2011(82)90002-7.
- "Pests, Chemicals, & Drilled Wood". crownbees.com. Retrieved 2017-04-18.
- Alfalfa Leafcutter Bee (Megachile rotundata)
