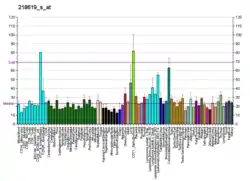
SUV39H1
Histone-lysine N-methyltransferase SUV39H1 is an enzyme that in humans is encoded by the SUV39H1 gene.[5]
Function
This gene is a member of the suppressor of variegation 3-9 homolog family and encodes a protein with a chromodomain and a C-terminal SET domain. This nuclear protein moves to the centromeres during mitosis and functions as a histone methyltransferase, methylating Lys-9 of histone H3. Overall, it plays a vital role in heterochromatin organization, chromosome segregation, and mitotic progression.[6]
References
- GRCh38: Ensembl release 89: ENSG00000101945 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000039231 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Aagaard L, Laible G, Selenko P, Schmid M, Dorn R, Schotta G, Kuhfittig S, Wolf A, Lebersorger A, Singh PB, Reuter G, Jenuwein T (June 1999). "Functional mammalian homologues of the Drosophila PEV-modifier Su(var)3-9 encode centromere-associated proteins which complex with the heterochromatin component M31". EMBO J. 18 (7): 1923–38. doi:10.1093/emboj/18.7.1923. PMC 1171278. PMID 10202156.
- "Entrez Gene: SUV39H1 suppressor of variegation 3-9 homolog 1 (Drosophila)".
- Zhang CL, McKinsey TA, Olson EN (October 2002). "Association of class II histone deacetylases with heterochromatin protein 1: potential role for histone methylation in control of muscle differentiation". Mol. Cell. Biol. 22 (20): 7302–12. doi:10.1128/mcb.22.20.7302-7312.2002. PMC 139799. PMID 12242305.
- Fujita N, Watanabe S, Ichimura T, Tsuruzoe S, Shinkai Y, Tachibana M, Chiba T, Nakao M (June 2003). "Methyl-CpG binding domain 1 (MBD1) interacts with the Suv39h1-HP1 heterochromatic complex for DNA methylation-based transcriptional repression". J. Biol. Chem. 278 (26): 24132–8. doi:10.1074/jbc.M302283200. PMID 12711603.
- Rual JF, Venkatesan K, Hao T, Hirozane-Kishikawa T, Dricot A, Li N, Berriz GF, Gibbons FD, Dreze M, Ayivi-Guedehoussou N, Klitgord N, Simon C, Boxem M, Milstein S, Rosenberg J, Goldberg DS, Zhang LV, Wong SL, Franklin G, Li S, Albala JS, Lim J, Fraughton C, Llamosas E, Cevik S, Bex C, Lamesch P, Sikorski RS, Vandenhaute J, Zoghbi HY, Smolyar A, Bosak S, Sequerra R, Doucette-Stamm L, Cusick ME, Hill DE, Roth FP, Vidal M (October 2005). "Towards a proteome-scale map of the human protein-protein interaction network". Nature. 437 (7062): 1173–8. Bibcode:2005Natur.437.1173R. doi:10.1038/nature04209. PMID 16189514. S2CID 4427026.
- Fuks F, Hurd PJ, Deplus R, Kouzarides T (May 2003). "The DNA methyltransferases associate with HP1 and the SUV39H1 histone methyltransferase". Nucleic Acids Res. 31 (9): 2305–12. doi:10.1093/nar/gkg332. PMC 154218. PMID 12711675.
- Vaute O, Nicolas E, Vandel L, Trouche D (January 2002). "Functional and physical interaction between the histone methyl transferase Suv39H1 and histone deacetylases". Nucleic Acids Res. 30 (2): 475–81. doi:10.1093/nar/30.2.475. PMC 99834. PMID 11788710.
- Chakraborty S, Sinha KK, Senyuk V, Nucifora G (August 2003). "SUV39H1 interacts with AML1 and abrogates AML1 transactivity. AML1 is methylated in vivo". Oncogene. 22 (34): 5229–37. doi:10.1038/sj.onc.1206600. PMID 12917624.
- Nielsen SJ, Schneider R, Bauer UM, Bannister AJ, Morrison A, O'Carroll D, Firestein R, Cleary M, Jenuwein T, Herrera RE, Kouzarides T (August 2001). "Rb targets histone H3 methylation and HP1 to promoters". Nature. 412 (6846): 561–5. Bibcode:2001Natur.412..561N. doi:10.1038/35087620. PMID 11484059. S2CID 4378296.
- Vandel L, Nicolas E, Vaute O, Ferreira R, Ait-Si-Ali S, Trouche D (October 2001). "Transcriptional repression by the retinoblastoma protein through the recruitment of a histone methyltransferase". Mol. Cell. Biol. 21 (19): 6484–94. doi:10.1128/mcb.21.19.6484-6494.2001. PMC 99795. PMID 11533237.
- Firestein R, Cui X, Huie P, Cleary ML (July 2000). "Set domain-dependent regulation of transcriptional silencing and growth control by SUV39H1, a mammalian ortholog of Drosophila Su(var)3-9". Mol. Cell. Biol. 20 (13): 4900–9. doi:10.1128/mcb.20.13.4900-4909.2000. PMC 85941. PMID 10848615.
- Khanal, Prem; Kim, Garam; Lim, Sung‐Chul; Yun, Hyo‐Jeong; Lee, Kwang Youl; Choi, Hoo‐Kyun; Choi, Hong Seok (2013). "Prolyl isomerase Pin1 negatively regulates the stability of SUV39H1 to promote tumorigenesis in breast cancer". The FASEB Journal. 27 (11): 4606–4618. doi:10.1096/fj.13-236851. PMID 23934277. S2CID 5259616.
Further reading
- Schotta G, Ebert A, Reuter G (2003). "SU(VAR)3-9 is a conserved key function in heterochromatic gene silencing". Genetica. 117 (2–3): 149–58. doi:10.1023/A:1022923508198. PMID 12723694. S2CID 39517859.
- Hijmans EM, Voorhoeve PM, Beijersbergen RL, van 't Veer LJ, Bernards R (1995). "E2F-5, a new E2F family member that interacts with p130 in vivo". Mol. Cell. Biol. 15 (6): 3082–9. doi:10.1128/mcb.15.6.3082. PMC 230539. PMID 7760804.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Aagaard L, Schmid M, Warburton P, Jenuwein T (2000). "Mitotic phosphorylation of SUV39H1, a novel component of active centromeres, coincides with transient accumulation at mammalian centromeres". J. Cell Sci. 113 (5): 817–29. PMID 10671371.
- Melcher M, Schmid M, Aagaard L, Selenko P, Laible G, Jenuwein T (2000). "Structure-function analysis of SUV39H1 reveals a dominant role in heterochromatin organization, chromosome segregation, and mitotic progression". Mol. Cell. Biol. 20 (10): 3728–41. doi:10.1128/MCB.20.10.3728-3741.2000. PMC 85674. PMID 10779362.
- Firestein R, Cui X, Huie P, Cleary ML (2000). "Set domain-dependent regulation of transcriptional silencing and growth control by SUV39H1, a mammalian ortholog of Drosophila Su(var)3-9". Mol. Cell. Biol. 20 (13): 4900–9. doi:10.1128/MCB.20.13.4900-4909.2000. PMC 85941. PMID 10848615.
- Fraser ME, James MN, Bridger WA, Wolodko WT (2000). "Phosphorylated and dephosphorylated structures of pig heart, GTP-specific succinyl-CoA synthetase". J. Mol. Biol. 299 (5): 1325–39. doi:10.1006/jmbi.2000.3807. PMID 10873456.
- Rea S, Eisenhaber F, O'Carroll D, Strahl BD, Sun ZW, Schmid M, Opravil S, Mechtler K, Ponting CP, Allis CD, Jenuwein T (2000). "Regulation of chromatin structure by site-specific histone H3 methyltransferases". Nature. 406 (6796): 593–9. Bibcode:2000Natur.406..593R. doi:10.1038/35020506. PMID 10949293. S2CID 205008015.
- Lachner M, O'Carroll D, Rea S, Mechtler K, Jenuwein T (2001). "Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins". Nature. 410 (6824): 116–20. Bibcode:2001Natur.410..116L. doi:10.1038/35065132. PMID 11242053. S2CID 4331863.
- Vandel L, Trouche D (2001). "Physical association between the histone acetyl transferase CBP and a histone methyl transferase". EMBO Rep. 2 (1): 21–6. doi:10.1093/embo-reports/kve002. PMC 1083799. PMID 11252719.
- Nielsen SJ, Schneider R, Bauer UM, Bannister AJ, Morrison A, O'Carroll D, Firestein R, Cleary M, Jenuwein T, Herrera RE, Kouzarides T (2001). "Rb targets histone H3 methylation and HP1 to promoters". Nature. 412 (6846): 561–5. Bibcode:2001Natur.412..561N. doi:10.1038/35087620. PMID 11484059. S2CID 4378296.
- Vandel L, Nicolas E, Vaute O, Ferreira R, Ait-Si-Ali S, Trouche D (2001). "Transcriptional repression by the retinoblastoma protein through the recruitment of a histone methyltransferase". Mol. Cell. Biol. 21 (19): 6484–94. doi:10.1128/MCB.21.19.6484-6494.2001. PMC 99795. PMID 11533237.
- Vaute O, Nicolas E, Vandel L, Trouche D (2002). "Functional and physical interaction between the histone methyl transferase Suv39H1 and histone deacetylases". Nucleic Acids Res. 30 (2): 475–81. doi:10.1093/nar/30.2.475. PMC 99834. PMID 11788710.
- Schotta G, Ebert A, Krauss V, Fischer A, Hoffmann J, Rea S, Jenuwein T, Dorn R, Reuter G (2002). "Central role of Drosophila SU(VAR)3-9 in histone H3-K9 methylation and heterochromatic gene silencing". EMBO J. 21 (5): 1121–31. doi:10.1093/emboj/21.5.1121. PMC 125909. PMID 11867540.
- Sewalt RG, Lachner M, Vargas M, Hamer KM, den Blaauwen JL, Hendrix T, Melcher M, Schweizer D, Jenuwein T, Otte AP (2002). "Selective interactions between vertebrate polycomb homologs and the SUV39H1 histone lysine methyltransferase suggest that histone H3-K9 methylation contributes to chromosomal targeting of Polycomb group proteins". Mol. Cell. Biol. 22 (15): 5539–53. doi:10.1128/MCB.22.15.5539-5553.2002. PMC 133945. PMID 12101246.
- Zhang CL, McKinsey TA, Olson EN (2002). "Association of class II histone deacetylases with heterochromatin protein 1: potential role for histone methylation in control of muscle differentiation". Mol. Cell. Biol. 22 (20): 7302–12. doi:10.1128/MCB.22.20.7302-7312.2002. PMC 139799. PMID 12242305.
- Yamamoto K, Sonoda M (2003). "Self-interaction of heterochromatin protein 1 is required for direct binding to histone methyltransferase, SUV39H1". Biochem. Biophys. Res. Commun. 301 (2): 287–92. doi:10.1016/S0006-291X(02)03021-8. PMID 12565857.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.