Titanium butoxide
Titanium butoxide is an metal-organic chemical compound with the formula Ti(OBu)4 (Bu = CH2CH2CH2CH3). It is a colorless odorless liquid, although aged samples are yellowish with a weak alcohol-like odor. It is soluble in many organic solvents.[1][3] It hydrolyzes to give titanium dioxide, which allows deposition of TiO2 coatings of various shapes and sizes down to the nanoscale.[4][5]
 gas phase structure | |
| Names | |
|---|---|
| IUPAC name
titanium(4+) butan-1-olate | |
| Other names | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.024.552 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| UN number | 2920 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C16H36O4Ti | |
| Molar mass | 340.32164 |
| Odor | weak alcohol-like[1] |
| Density | 0.998 g/cm3[1] |
| Melting point | -55 °C[1] |
| Boiling point | 312 °C[1] |
| decomposes[1] | |
| Solubility | most organic solvents except ketones[1] |
Refractive index (nD) |
1.486[1] |
| Thermochemistry | |
Heat capacity (C) |
711 J/(mol·K)[2] |
Std enthalpy of formation (ΔfH⦵298) |
-1670 kJ/mol[2] |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
3122 mg/kg (rat, oral) and 180 mg/kg (mouse, intravenal).[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure and synthesis
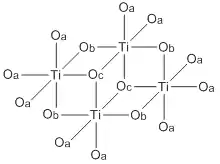
Like most titanium alkoxides (exception: titanium isopropoxide), Ti(OBu)4 is not a monomer but exists as a cluster (see titanium ethoxide). Nonetheless it is often depicted as a simple monomer.
It is produced by treating titanium tetrachloride with butanol:
- TiCl4 + 4 HOBu → Ti(OBu)4 + 4 HCl
The reaction requires base to proceed to completion.
Reactions
Like other titanium alkoxides, titanium butoxide exchanges alkoxide groups:
- Ti(OBu)4 + HOR → Ti(OBu)3(OR) + HOBu
- Ti(OBu)3(OR) + HOR → Ti(OBu)2(OR)2 + HOBu
etc. For this reason, titanium butoxide is not compatible with alcohol solvents.
Analogous to the alkoxide exchange, titanium butoxide hydrolyzes readily. The reaction details are complex, but can be summarized with this balanced equation.
- Ti(OBu)4 + 2 H2O → TiO2 + 4 HOBu
Pyrolysis also affords the dioxide:
- Ti(OBu)4 → TiO2 + 2 Bu2O
Reactions and hazard

Tetrabutyl orthotitanate reacts with alkylcyclosiloxanes. With ocatamethylcyclotetrasiloxane it produces dibutoxydimethylsilane, 1,5-dibutoxyhexamethyltrisiloxane, 1,7-dibutoxyoctamethyltetrasiloxane, 1,3-dibutoxytetramethyldisiloxane and polymers. With hexamethylcyclotrisiloxane it also produces dibutoxydimethylsilane.[6]
References
- Butyl titanate. pubchem.ncbi.nlm.nih.gov
- Tetrabutyl titanate. nist.gov
- Pohanish, Richard P.; Greene, Stanley A. (2009). Wiley Guide to Chemical Incompatibilities. John Wiley & Sons. p. 1010. ISBN 978-0-470-52330-8.
- Wang, Cui (2015). "Hard-templating of chiral TiO2 nanofibres with electron transition-based optical activity". Science and Technology of Advanced Materials. 16 (5): 054206. Bibcode:2015STAdM..16e4206W. doi:10.1088/1468-6996/16/5/054206. PMC 5070021. PMID 27877835.
- Wu, Limin; Baghdachi, Jamil (2015). Functional Polymer Coatings: Principles, Methods, and Applications. Wiley. p. 10. ISBN 978-1-118-88303-7.
- K. A. Andrianov, Sh. V. Pichkhadze, V. V. Komarova, Ts. N. Vardosanidze (1962). "Reactions of organocyclosiloxanes with tetrabutyl orthotitanate". Bulletin of the Academy of Sciences of the USSR Division of Chemical Science. 11 (5): 776–779. doi:10.1007/BF00905301. ISSN 0568-5230.CS1 maint: uses authors parameter (link)