Water dimer
The water dimer consists of two water molecules loosely bound by a hydrogen bond. It is the smallest water cluster. Because it is the simplest model system for studying hydrogen bonding in water, it has been the target of many theoretical [1][2][3] (and later experimental) studies that it has been called a "theoretical Guinea pig".[4]
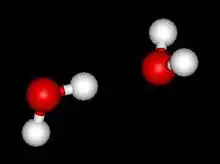
Structure and properties
The ab initio binding energy between the two water molecules is estimated to be 5-6 kcal/mol, although values between 3 and 8 have been obtained depending on the method. The experimentally measured dissociation energy (including nuclear quantum effects) of (H2O)2 and (D2O)2 are 3.16 ± 0.03 kcal/mol (13.22 ± 0.12 kJ/mol)[5] and 3.56 ± 0.03 kcal/mol (14.88 ± 0.12 kJ/mol),[6] respectively. The values are in excellent agreement with calculations.[7][8] The O-O distance of the vibrational ground-state is experimentally measured at ca. 2.98 Å;[9] the hydrogen bond is almost linear, but the angle with the plane of the acceptor molecule is about 57°. The vibrational ground-state is known as the linear water dimer (shown in the figure to the right), which is a near prolate top (viz., in terms of rotational constants, A > B ≈ C). Other configurations of interest include the cyclic dimer and the bifurcated dimer.
History and relevance
The first theoretical study of the water dimer was an ab initio calculation published in 1968 by Morokuma and Pedersen.[10] Since then, the water dimer has been the focus of sustained interest by theoretical chemists concerned with hydrogen bonding—a search of the CAS database up to 2006 returns over 1100 related references (73 of them in 2005). The water dimer is a hotly studied topic in Physical Chemistry for several reasons. (H2O)2 is thought to play a significant role in many atmospheric processes, including acid rain formation, absorption of excess solar radiation, condensation of water droplets, and chemical reactions. In addition, a complete understanding of the water dimer is thought to play a key role in a more thorough understanding of hydrogen bonding in liquid and solid forms of water.
References
- Buckingham, A. D. The hydrogen bond, and the structure and properties of water and the water dimer. Journal of Molecular Structure 1991, 250, 111-18.
- Goldman, N., Leforestier, C., and Saykally, R. J., Water Dimers in the Atmosphere II: Results from the VRT(ASP-W)III Potential Surface, Journal of Physical Chemistry A, 2004, 108, p. 787-794.
- Schütz, M.; Brdarski, S.; Widmark, P.-O.; Lindh, R.; Karlström, G. The water dimer interaction energy: Convergence to the basis set limit at the correlated level, Journal Chemical Physics, 1997, 107, 4597-4605.
- Jeffrey, G. A.; An Introduction to Hydrogen Bonding (Topics in Physical Chemistry). Oxford University Press, USA (March 13, 1997). ISBN 0-19-509549-9
- Rocher-Casterline, B. E.; Ch'ng, L. C.; Mollner, A. K.; Reisler, H. Journal of Chemical Physics 2011, 115, 6903-6909 doi:10.1063/1.3598339
- Ch'ng, L. C.; Samanta, A. K.; Czakó, G.; Bowman, J. M.; Reisler, H. Journal of American Chemical Society 2012, 134, 15430 doi:10.1021/ja305500x
- Shank, A.; Wang, Y.; Kaledin, A.; Braams, B. J.; Bowman. J. M. Journal of Chemical Physics 2009, 130, 144314 doi:10.1063/1.3112403
- Leforestier, C.; Szalewicz, K.; van der Avoird, A. Journal of Chemical Physics 2012, 137, 014305 doi:10.1063/1.4722338
- Scheiner, S. Ab initio studies of hydrogen bonds: the water dimer paradigm. Annual Review of Physical Chemistry 1994, 45, 23-56.
- Morokuma, K.; Pedersen, L. Molecular-orbital studies of hydrogen bonds. An ab initio calculation for dimeric water. Journal of Chemical Physics 1968, 48, 3275-3282.
