Azurite
Azurite is a soft, deep-blue copper mineral produced by weathering of copper ore deposits. During the early 19th century, it was also known as chessylite, after the type locality at Chessy-les-Mines near Lyon, France.[2] The mineral, a basic carbonate with the chemical formula Cu3(CO3)2(OH)2, has been known since ancient times, and was mentioned in Pliny the Elder's Natural History under the Greek name kuanos (κυανός: "deep blue," root of English cyan) and the Latin name caeruleum.[4] Since antiquity, azurite's exceptionally deep and clear blue has been associated with low-humidity desert and winter skies. The modern English name of the mineral reflects this association, since both azurite and azure are derived via Arabic from the Persian lazhward (لاژورد), an area known for its deposits of another deep-blue stone, lapis lazuli ("stone of azure").
| Azurite | |
|---|---|
 Azurite from Burra Mine, South Australia | |
| General | |
| Category | Carbonate mineral |
| Formula (repeating unit) | Cu3(CO3)2(OH)2 |
| Strunz classification | 5.BA.05 |
| Crystal system | Monoclinic |
| Crystal class | Prismatic (2/m) (same H-M symbol) |
| Space group | P21/c |
| Unit cell | a = 5.01 Å, b = 5.85 Å c = 10.35 Å; β = 92.43°; Z = 2 |
| Identification | |
| Formula mass | 344.67 g/mol |
| Color | Azure-blue, dark to pale blue; pale blue in transmitted light |
| Crystal habit | Massive, prismatic, stalactitic, tabular |
| Twinning | Rare, twin planes {101}, {102} or {001} |
| Cleavage | Perfect on {011}, fair on {100}, poor on {110} |
| Fracture | Conchoidal |
| Tenacity | brittle |
| Mohs scale hardness | 3.5 to 4 |
| Luster | Vitreous |
| Streak | Light blue |
| Diaphaneity | Transparent to translucent |
| Specific gravity | 3.773 (measured), 3.78 (calculated) |
| Optical properties | Biaxial (+) |
| Refractive index | nα = 1.730 nβ = 1.758 nγ = 1.838 |
| Birefringence | δ = 0.108 |
| Pleochroism | Visible shades of blue |
| 2V angle | Measured: 68°, calculated: 64° |
| Dispersion | relatively weak |
| References | [1][2][3] |
Mineralogy
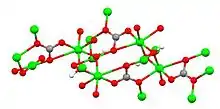
Azurite is one of two basic copper(II) carbonate minerals, the other being bright green malachite. Simple copper carbonate (CuCO3) is not known to exist in nature. Azurite has the formula Cu3(CO3)2(OH)2, with the copper(II) cations linked to two different anions, carbonate and hydroxide. Small crystals of azurite can be produced by rapidly stirring a few drops of copper sulfate solution into a saturated solution of sodium carbonate and allowing the solution to stand overnight. Azurite crystals are monoclinic.[5] Large crystals are dark blue, often prismatic.[2][3][6] Azurite specimens can be massive to nodular. They are often stalactitic in form. Specimens tend to lighten in color over time due to weathering of the specimen surface into malachite. Azurite is soft, with a Mohs hardness of only 3.5 to 4. The specific gravity of azurite is 3.77 to 3.89. Azurite is destroyed by heat, losing carbon dioxide and water to form black, copper(II) oxide powder. Characteristic of a carbonate, specimens effervesce upon treatment with hydrochloric acid.
Color
The optical properties (color, intensity) of minerals such as azurite and malachite are characteristic of copper(II). Many coordination complexes of copper(II) exhibit similar colors. As explained within the context of crystal field theory, the color results from low energy d-d transitions associated with the d9 metal center.
Weathering
Azurite is unstable in open air compared to malachite, and often is pseudomorphically replaced by malachite. This weathering process involves the replacement of some of the carbon dioxide (CO2) units with water (H2O), changing the carbonate:hydroxide ratio of azurite from 1:1 to the 1:2 ratio of malachite:
- 2 Cu3(CO3)2(OH)2 + H2O → 3 Cu2(CO3)(OH)2 + CO2
From the above equation, the conversion of azurite into malachite is attributable to the low partial pressure of carbon dioxide in air. Azurite is also incompatible with aquatic media, such as saltwater aquariums.
Uses
Pigments
Azurite is unstable in air. However it was used as a blue pigment in antiquity.[7] Azurite is naturally occurring in Sinai and the Eastern Desert of Egypt. It was reported by F. C. J. Spurrell (1895) in the following examples; a shell used as a pallet in a Fourth Dynasty (2613 to 2494 BCE) context in Meidum, a cloth over the face of a Fifth Dynasty (2494 to 2345 BCE) mummy also at Meidum and a number of Eighteenth Dynasty (1543–1292 BCE) wall paintings.[8] Depending on the degree of fineness to which it was ground, and its basic content of copper carbonate, it gave a wide range of blues. It has been known as mountain blue or Armenian stone, in addition it was formerly known as Azurro Della Magna (from Italian). When mixed with oil it turns slightly green. When mixed with egg yolk it turns green-grey. It is also known by the names blue bice and blue verditer, though verditer usually refers to a pigment made by chemical process. Older examples of azurite pigment may show a more greenish tint due to weathering into malachite. Much azurite was mislabeled lapis lazuli, a term applied to many blue pigments. As chemical analysis of paintings from the Middle Ages improves, azurite is being recognized as a major source of the blues used by medieval painters. Lapis lazuli (the pigment ultramarine) was chiefly supplied from Afghanistan during the Middle Ages, whereas azurite was a common mineral in Europe at the time. Sizable deposits were found near Lyons, France. It was mined since the 12th century in Saxony, in the silver mines located there.[9]
Heating can be used to distinguish azurite from purified natural ultramarine blue, a more expensive but more stable blue pigment, as described by Cennino D'Andrea Cennini. Ultramarine withstands heat, whereas azurite converts to black copper oxide. However, gentle heating of azurite produces a deep blue pigment used in Japanese painting techniques.
 Ground azurite for use as a pigment
Ground azurite for use as a pigment The background of Lady with a Squirrel by Hans Holbein the Younger was painted with Azurite
The background of Lady with a Squirrel by Hans Holbein the Younger was painted with Azurite The greenish tint of the Madonna's mantle in Raphael's Madonna and Child Enthroned with Saints is due to azurite weathering to malachite
The greenish tint of the Madonna's mantle in Raphael's Madonna and Child Enthroned with Saints is due to azurite weathering to malachite
Jewelry
Azurite is used occasionally as beads and as jewelry, and also as an ornamental stone. However, its softness and tendency to lose its deep blue color as it weathers limit such uses. Heating destroys azurite easily, so all mounting of azurite specimens must be done at room temperature.
Collecting
The intense color of azurite makes it a popular collector's stone. However, bright light, heat, and open air all tend to reduce the intensity of its color over time. To help preserve the deep blue color of a pristine azurite specimen, collectors should use a cool, dark, sealed storage environment similar to that of its original natural setting.
Prospecting
While not a major ore of copper itself, the presence of azurite is a good surface indicator of the presence of weathered copper sulfide ores. It is usually found in association with the chemically similar malachite, producing a striking color combination of deep blue and bright green that is strongly indicative of the presence of copper ores.
History
The use of azurite and malachite as copper ore indicators led indirectly to the name of the element nickel in the English language. Nickeline, a principal ore of nickel that is also known as niccolite, weathers at the surface into a green mineral (annabergite) that resembles malachite. This resemblance resulted in occasional attempts to smelt nickeline in the belief that it was copper ore, but such attempts always ended in failure due to high smelting temperatures needed to reduce nickel. In Germany this deceptive mineral came to be known as kupfernickel, literally "copper demon." The Swedish alchemist Baron Axel Fredrik Cronstedt (who had been trained by Georg Brandt, the discoverer of the nickel-like metal cobalt) realized that there was probably a new metal hiding within the kupfernickel ore, and in 1751 he succeeded in smelting kupfernickel to produce a previously unknown (except in certain meteorites) silvery white, iron-like metal. Logically, Cronstedt named his new metal after the nickel part of kupfernickel.
Gallery of azurite mineral specimens
 Azurite crystals from China
Azurite crystals from China Azurite from Arizona, collected by Dr John Hunter in the 18th century, Hunterian Museum, Glasgow
Azurite from Arizona, collected by Dr John Hunter in the 18th century, Hunterian Museum, Glasgow Fresh, unweathered azurite crystals showing the deep blue of unaltered azurite. From Špania Dolina, Slovakia
Fresh, unweathered azurite crystals showing the deep blue of unaltered azurite. From Špania Dolina, Slovakia Azurite with Malachite, Copper Queen mine, Bisbee, Arizona
Azurite with Malachite, Copper Queen mine, Bisbee, Arizona Azurite from Touissit, Morocco
Azurite from Touissit, Morocco Azurite, Morenci, Arizona
Azurite, Morenci, Arizona Azurite in siltstone, Malbunka mine, Northern Territory, Australia
Azurite in siltstone, Malbunka mine, Northern Territory, Australia Azurite from Tsumeb, Namibia
Azurite from Tsumeb, Namibia Azurite, cross-section through merged stalactites, Bisbee, Arizona
Azurite, cross-section through merged stalactites, Bisbee, Arizona Azurite crystal, from the minerals' collection at the Natural History Museum, London.
Azurite crystal, from the minerals' collection at the Natural History Museum, London.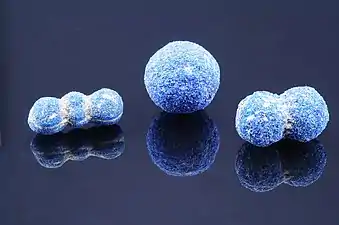 Spheroidal azurite specimens from Utah
Spheroidal azurite specimens from Utah
References
- Handbook of Mineralogy
- Mindat.org
- Webmineral.com Webmineral Data
- The Ancient Library: Smith, Dictionary of Greek and Roman Antiquities, p.321, right col., under BLUE Archived December 20, 2005, at the Wayback Machine
- Zigan, F.; Schuster, H.D. (1972). "Verfeinerung der Struktur von Azurit, Cu3(OH)2(CO3)2, durch Neutronenbeugung". Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie. 135 (5–6): 416–436. Bibcode:1972ZK....135..416Z. doi:10.1524/zkri.1972.135.5-6.416.
- Hurlbut, Cornelius S.; Klein, Cornelis, 1985, Manual of Mineralogy, 20th ed., Wiley, ISBN 978-0-471-00042-6
- Gettens, R.J. and Fitzhugh, E.W., Azurite and Blue Verditer, in Artists’ Pigments. A Handbook of Their History and Characteristics, Vol. 2: A. Roy (Ed.) Oxford University Press 1993, p. 23–24
- Nicholson, Paul; Shaw, Ian (2000). Ancient Egyptian Materials and Technology. Cambridge University Press. ISBN 978-0521452571.
- Andersen, Frank J. Riches of the Earth. W.H. Smith Publishers, New York, 1981, ISBN 0-8317-7739-7
Further reading
- Gettens, R.J. and Fitzhugh, E.W., "Azurite and Blue Verditer", in Artists' Pigments: A Handbook of Their History and Characteristics, Vol. 2: A. Roy (Ed.) Oxford University Press 1993, p. 23–35
External links
| Wikimedia Commons has media related to Azurite. |
- Spencer, Leonard James (1911). . Encyclopædia Britannica. 3 (11th ed.). p. 86.
- Azurite, Colourlex