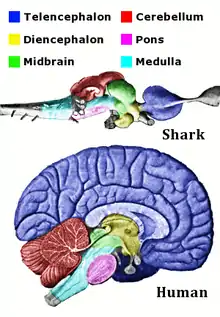
Cephalization
Cephalization is an evolutionary trend in which, over many generations, the mouth, sense organs, and nerve ganglia become concentrated at the front end of an animal, producing a head region. This is associated with movement and bilateral symmetry, such that the animal has a definite head end. This led to the formation of a highly sophisticated brain in three groups of animals, namely the arthropods, cephalopod molluscs, and vertebrates.

Animals without bilateral symmetry
Cnidaria, such as the radially symmetrical Hydrozoa, show some degree of cephalization. The Anthomedusae have a head end with their mouth, photoreceptive cells, and a concentration of neural cells.[1]
Bilateria
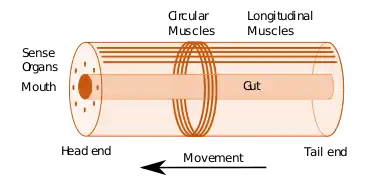
Cephalization is a characteristic feature of the Bilateria, a large group containing the majority of animal phyla.[2] These have the ability to move, using muscles, and a body plan with a front end that encounters stimuli first as the animal moves forwards, and accordingly has evolved to contain many of the body's sense organs, able to detect light, chemicals, and often sound. There is often also a collection of nerve cells able to process the information from these sense organs, forming a brain in several phyla and one or more ganglia in others.[3]
Acoela
The Acoela are basal bilaterians, part of the Xenacoelomorpha. They are small and simple animals, and have very slightly more nerve cells at the head end than elsewhere, not forming a distinct and compact brain. This represents an early stage in cephalization.[4]
Flatworms
_(cropped).jpg.webp)
The Platyhelminthes (flatworms) have a more complex nervous system than the Acoela, and are lightly cephalized, for instance having an eyespot above the brain, near the front end.[4]
Complex active bodies
The philosopher Michael Trestman noted that three bilaterian phyla, namely the arthropods, the molluscs in the shape of the cephalopods, and the chordates, were distinctive in having "complex active bodies", something that the acoels and flatworms did not have. Any such animal, whether predator or prey, has to be aware of its environment—to catch its prey, or to evade its predators. These groups are exactly those that are most highly cephalized.[5][6] These groups, however, are not closely related: in fact, they represent widely separated branches of the Bilateria, as shown on the phylogenetic tree; their lineages split hundreds of millions of years ago. Other (less cephalized) phyla are not shown, for clarity.[7][8][9]
| Planulozoa |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| 680 mya |
Arthropods
In arthropods, cephalization progressed with increasing incorporation of trunk segments into the head region. This was advantageous because it allowed for the evolution of more effective mouth-parts for capturing and processing food. Insects are strongly cephalized, their brain made of three fused ganglia attached to the ventral nerve cord, which in turn has a pair of ganglia in each segment of the thorax and abdomen. The insect head is an elaborate structure made of several segments fused rigidly together, and equipped with both simple and compound eyes, and multiple appendages including sensory antennae and complex mouthparts (maxillae and mandibles).[4]

Cephalopods
Cephalopod molluscs including octopus, squid, cuttlefish and nautilus are the most intelligent and highly cephalized invertebrates, with well-developed senses, including advanced 'camera' eyes and large brains.[10][11]
Vertebrates
Cephalization in vertebrates, the group that includes mammals, birds, and fishes, has been studied extensively.[4] The heads of vertebrates are complex structures, with distinct sense organs; a large, multi-lobed brain; jaws, often with teeth; and a tongue. Cephalochordates like Branchiostoma (the lancelet, a small fishlike animal with very little cephalization), are closely related to vertebrates but do not have these structures. In the 1980s, the new head hypothesis proposed that the vertebrate head is an evolutionary novelty resulting from the emergence of neural crest and cranial placodes (thickened areas of ectoderm).[12][13] However, in 2014, a transient larva tissue of the lancelet was found to be virtually indistinguishable from the neural crest-derived cartilage which forms the vertebrate skull, suggesting that persistence of this tissue and expansion into the entire head space could be a viable evolutionary route to formation of the vertebrate head.[14] Advanced vertebrates have increasingly elaborate brains.[4]
See also
References
- Satterlie, Richard (February 2017). Byrne, John H (ed.). "The Oxford Handbook of Invertebrate Neurobiology". 1. Oxford: 184–218. doi:10.1093/oxfordhb/9780190456757.013.7. ISBN 9780190456757.
Ocelli located at the base of the many tentacles represent one input to the B system, whereas the neurons of the O system are directly photosensitive. Many hydromedusae have ocelli of different levels of complexity (Singla, 1974). In addition, other marginal sensory structures associated with the outer nerve ring include statocysts (Singla, 1975), and mechanoreceptors, such as the tactile combs of Aglantha, which are located at the tentacle bases, and can activate the escape swimming circuitry (Arkett & Mackie, 1988; Mackie, 2004b).
Cite journal requires|journal=(help);|chapter=ignored (help) - "Trends in evolution". University of California Museum of Paleontology. Retrieved 10 January 2019.
- Brusca, Richard C. (2016). Introduction to the Bilateria and the Phylum Xenacoelomorpha | Triploblasty and Bilateral Symmetry Provide New Avenues for Animal Radiation (PDF). Invertebrates. Sinauer Associates. pp. 345–372. ISBN 978-1605353753.
- Çabej, Nelson (2013). Rise of the Animal Kingdom and Epigenetic Mechanisms of Evolution. Building the most complex structure on Earth : an epigenetic narrative of development and evolution of animals. Elsevier. pp. 239–298. ISBN 978-0-12-401667-5.
- Trestman, Michael (April 2013). "The Cambrian Explosion and the Origins of Embodied Cognition" (PDF). Biological Theory. 8 (1): 80–92. doi:10.1007/s13752-013-0102-6. S2CID 84629416.
- Godfrey-Smith, Peter (2017). Other Minds: The Octopus and the Evolution of Intelligent Life. HarperCollins Publishers. p. 38. ISBN 978-0-00-822628-2.
- Peterson, Kevin J.; Cotton, James A.; Gehling, James G.; Pisani, Davide (27 April 2008). "The Ediacaran emergence of bilaterians: congruence between the genetic and the geological fossil records". Philosophical Transactions of the Royal Society of London B: Biological Sciences. 363 (1496): 1435–1443. doi:10.1098/rstb.2007.2233. PMC 2614224. PMID 18192191.
- Laura Wegener Parfrey; Daniel J G Lahr; Andrew H Knoll; Laura A Katz (16 August 2011). "Estimating the timing of early eukaryotic diversification with multigene molecular clocks" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 108 (33): 13624–9. doi:10.1073/PNAS.1110633108. ISSN 0027-8424. PMC 3158185. PMID 21810989. Wikidata Q24614721.
- "Raising the Standard in Fossil Calibration". Fossil Calibration Database. Retrieved 3 March 2018.
- Tricarico, E.; Amodio, P.; Ponte, G.; Fiorito, G. (2014). "Cognition and recognition in the cephalopod mollusc Octopus vulgaris: coordinating interaction with environment and conspecifics". In Witzany, G. (ed.). Biocommunication of Animals. Springer. pp. 337–349. ISBN 978-94-007-7413-1.
- Solomon, Eldra; Berg, Linda; Martin, Diana W. (2010). Biology. Cengage Learning. p. 884. ISBN 978-1-133-17032-7.
- Gans, C.; Northcutt, R. G. (1983). "Neural crest and the origin of vertebrates: a new head". Science. 220 (4594): 268–273. Bibcode:1983Sci...220..268G. doi:10.1126/science.220.4594.268. PMID 17732898. S2CID 39290007.
- Diogo, R.; et al. (2015). "A new heart for a new head in vertebrate cardiopharyngeal evolution". Nature. 520 (7548): 466–73. Bibcode:2015Natur.520..466D. doi:10.1038/nature14435. PMC 4851342. PMID 25903628.
- Jandzik, D.; Garnett, A. T.; Square, T. A.; Cattell, M. V.; Yu, J. K.; Medeiros, D. M. (26 February 2015). "Evolution of the new vertebrate head by co-option of an ancient chordate skeletal tissue". Nature. 518 (7540): 534–537. Bibcode:2015Natur.518..534J. doi:10.1038/nature14000. PMID 25487155. S2CID 4449267. For laysummary see: "Evolution: How vertebrates got a head". Research. Nature (paper). 516 (7530): 171. 11 December 2014.