Lead carbonate
Lead(II) carbonate is the chemical compound PbCO3. It is a white solid with several practical uses, despite its toxicity.[2] It occurs naturally as the mineral cerussite.[3]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Lead(II) carbonate | |
| Other names | |
| Identifiers | |
| ECHA InfoCard | 100.009.041 |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| Properties | |
| PbCO3 | |
| Molar mass | 267.21 g/mol |
| Appearance | White powder |
| Density | 6.582 g/cm3 |
| Melting point | 315 °C (599 °F; 588 K) (decomposes) |
| 0.00011 g/100 mL (20 °C) | |
Solubility product (Ksp) |
1.46 x 10−13 |
| Solubility | insoluble in alcohol, ammonia; soluble in acid, alkali |
| −61.2·10−6 cm3/mol | |
Refractive index (nD) |
1.804 [1] |
| Hazards | |
| Safety data sheet | External MSDS |
EU classification (DSD) (outdated) |
Repr. Cat. 1/3 Toxic (T) Harmful (Xn) Dangerous for the environment (N) |
| R-phrases (outdated) | R61, R20/22, R33, R62, R50/53 |
| S-phrases (outdated) | S53, S45, S60, S61 |
| Flash point | Non-flammable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Structure
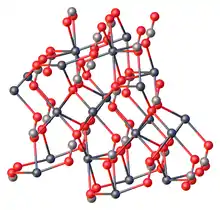
Like all metal carbonates, lead(II) carbonate adopts a dense, highly crosslinked structure consisting of intact CO32- and metal cation sites. As verified by X-ray crystallography, the Pb(II) centers are seven-coordinate, being surrounded by multiple carbonate ligands. The carbonate centers are bonded to bidentate to a single Pb and bridge to five other Pb sites.[4]
Production and use
Lead carbonate is manufactured by passing carbon dioxide into a cold dilute solution of lead(II) acetate, or by shaking a suspension of a lead salt more soluble than the carbonate with ammonium carbonate at a low temperature to avoid formation of basic lead carbonate.[2]
- Pb(CH3COO)2 + (NH4)2CO3 → PbCO3 + 2 NH4(CH3COO)
Lead carbonate is used as a catalyst to polymerize formaldehyde to poly(oxymethylene). It improves the bonding of chloroprene to wire.[2]
Regulations
The supply and use of this compound is restricted in Europe.[5]
Other lead carbonates
A number of lead carbonates are known:
- White lead, a basic lead carbonate, 2PbCO3·Pb(OH)2
- Shannonite, PbCO3·PbO
- Plumbonacrite, 3PbCO3·Pb(OH)2·PbO[6]
- PbCO3·2PbO
- Abellaite, NaPb2(OH)(CO3)2
- Leadhillite, 2PbCO3·PbSO4·Pb(OH)2
References
- Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- Carr, Dodd S. (2005). "Lead Compounds". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a15_249.
- Inorganic Chemistry, Egon Wiberg, Arnold Frederick Holleman Elsevier 2001 ISBN 0-12-352651-5
- Sahl, Kurt (1974). "Verfeinerung der Kristallstruktur von Cerussit, PbCO3". Zeitschrift für Kristallographie. 139 (3–5): 215–222. Bibcode:1974ZK....139..215S. doi:10.1524/zkri.1974.139.3-5.215.
- "EU law - EUR-Lex".
- S.V. Krivovichev and P.C. Burns, "Crystal chemistry of basic lead carbonates. II. Crystal structure of synthetic 'plumbonacrite'." Mineralogical Magazine, 64(6), pp. 1069-1075, December 2000. "Archived copy" (PDF). Archived from the original (PDF) on 2009-05-21. Retrieved 2009-05-21.CS1 maint: archived copy as title (link)