Tetrairidium dodecacarbonyl
Tetrairidium dodecacarbonyl is the chemical compound with the formula Ir4(CO)12. This tetrahedral cluster is the most common and most stable "binary" carbonyl of iridium. This air-stable species is only poorly soluble in organic solvents. It has been used to prepare bimetallic clusters and catalysts, e.g. for the water gas shift reaction, and reforming, but these studies are of purely academic interest.
12.svg.png.webp) | |
 | |
| Names | |
|---|---|
| IUPAC names
dodecacarbonyl-1κ3C,2κ3C,3κ3C,4κ3C-[Td-(13)-Δ4-closo]-tetrairidium(6 Ir—Ir) tetrahedro-tetrakis(tricarbonyliridium)(6 Ir—Ir) | |
| Other names
iridium(0) carbonyl; iridium carbonyl; iridium dodecacarbonyl | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.038.718 |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| Ir4(CO)12 | |
| Molar mass | 1104.92 g/mol |
| Appearance | Canary-yellow crystals |
| Melting point | 195 °C (383 °F; 468 K) |
| Solubility | Chlorocarbons, toluene, tetrahydrofuran |
| Related compounds | |
Related compounds |
Tetrarhodium dodecacarbonyl |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
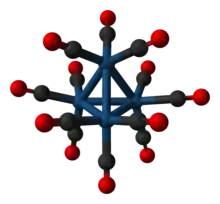
Structure
Each Ir center is octahedral, being bonded to 3 other iridium atoms and three terminal CO ligands. Ir4(CO)12 has Td symmetry with an average Ir-Ir distances of 2.693 Å.[1] The related clusters Rh4(CO)12 and Co4(CO)12 have C3v symmetry because of the presence of three bridging CO ligands in each.
Preparation
It is prepared in two steps by reductive carbonylation of hydrated iridium trichloride. The first step gives [Ir(CO)2Cl2]−.[2]
- IrCl3 + 3 CO + H2O → [Ir(CO)2Cl2]− + CO2 + 2 H+ + Cl−
- 4 [Ir(CO)2Cl2]− + 6 CO + 2 H2O → Ir4(CO)12 + 2 CO2 + 4 H+ + 8 Cl−
References
- Churchill, Melvyn Rowen; Hutchinson, John P. (1978). "Crystal Structure of tetrairidium dodecacarbonyl, Ir4(CO)12. An Unpleasant Case of Disorder". Inorganic Chemistry. 17 (12): 3528–35. doi:10.1021/ic50190a040.CS1 maint: uses authors parameter (link)
- Pergola, R. D.; Garlaschelli, L.; Matinengo, S. (1990). "Dodecacarbonyltetrairidium: Ir4(CO)12". Inorganic Syntheses. 28: 245–247. doi:10.1002/9780470132593.ch63. ISBN 9780470132593.