Agrin
Agrin is a large proteoglycan whose best-characterised role is in the development of the neuromuscular junction during embryogenesis. Agrin is named based on its involvement in the aggregation of acetylcholine receptors during synaptogenesis. In humans, this protein is encoded by the AGRN gene.[5][6][7]
| Agrin NtA domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | NtA | ||||||||
| Pfam | PF03146 | ||||||||
| InterPro | IPR004850 | ||||||||
| SCOP2 | 1jc7 / SCOPe / SUPFAM | ||||||||
| |||||||||
This protein has nine domains homologous to protease inhibitors.[8] It may also have functions in other tissues and during other stages of development. It is a major proteoglycan component in the glomerular basement membrane and may play a role in the renal filtration and cell-matrix interactions.[9]
Agrin-targeted autoantibodies have been infrequently observed in myasthenia gravis.
Discovery
Agrin was first identified by the U.J. McMahan laboratory, Stanford University.[10]
Mechanism of action
During development in humans, the growing end of motor neuron axons secrete a protein called agrin.[11] When secreted, agrin binds to several receptors on the surface of skeletal muscle. The receptor which appears to be required for the formation of the neuromuscular junction (NMJ) is called the MuSK receptor (Muscle specific kinase).[12][13] MuSK is a receptor tyrosine kinase - meaning that it induces cellular signaling by causing the addition of phosphate molecules to particular tyrosines on itself and on proteins that bind the cytoplasmic domain of the receptor.
In addition to MuSK, agrin binds several other proteins on the surface of muscle, including dystroglycan and laminin. It is seen that these additional binding steps are required to stabilize the NMJ.
The requirement for Agrin and MuSK in the formation of the NMJ was demonstrated primarily by knockout mouse studies. In mice that are deficient for either protein, the neuromuscular junction does not form.[14] Many other proteins also comprise the NMJ, and are required to maintain its integrity. For example, MuSK also binds a protein called "dishevelled" (Dvl), which is in the Wnt signalling pathway. Dvl is additionally required for MuSK-mediated clustering of AChRs, since inhibition of Dvl blocks clustering.
Signaling
The nerve secretes agrin, resulting in phosphorylation of the MuSK receptor.
It seems that the MuSK receptor recruits casein kinase 2, which is required for clustering.[15]
A protein called rapsyn is then recruited to the primary MuSK scaffold, to induce the additional clustering of acetylcholine receptors (AChR). This is thought of as the secondary scaffold. A protein called Dok-7 has shown to be additionally required for the formation of the secondary scaffold; it is apparently recruited after MuSK phosphorylation and before acetylcholine receptors are clustered.
Structure
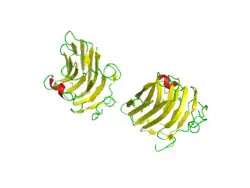
There are three potential heparan sulfate (HS) attachment sites within the primary structure of agrin, but it is thought that only two of these actually carry HS chains when the protein is expressed.
In fact, one study concluded that at least two attachment sites are necessary by inducing synthetic agents. Since agrin fragments induce acetylcholine receptor aggregation as well as phosphorylation of the MuSK receptor, researchers spliced them and found that the variant did not trigger phosphorylation. It has also been shown that the G3 domain of agrin is very plastic, meaning it can discriminate between binding partners for a better fit.[16]
Heparan sulfate glycosaminoglycans covalently linked to the agrin protein have been shown to play a role in the clustering of AChR. Interference in the correct formation of heparan sulfate through the addition of chlorate to skeletal muscle cell culture results in a decrease in the frequency of spontaneous acetylcholine receptor (AChR) clustering. It may be that rather than solely binding directly to the agrin protein core a number of components of the secondary scaffold may also interact with its heparan sulfate side-chains.[17]
A role in the retention of anionic macromolecules within the vasculature has also been suggested for agrin-linked HS at the glomerular or alveolar basement membrane.
Research
Agrin is investigated in relation with osteoarthritis.[18][19] In addition, by its ability to activate the Hippo signaling pathway, agrin is emerging as a key proteoglycan in the tumor microenvironment.[20]
References
- GRCh38: Ensembl release 89: ENSG00000188157 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000041936 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Rupp F, Payan DG, Magill-Solc C, Cowan DM, Scheller RH (May 1991). "Structure and expression of a rat agrin". Neuron. 6 (5): 811–23. doi:10.1016/0896-6273(91)90177-2. PMID 1851019. S2CID 44440186.
- Kröger S, Schröder JE (October 2002). "Agrin in the developing CNS: new roles for a synapse organizer". News Physiol. Sci. 17 (5): 207–12. doi:10.1152/nips.01390.2002. PMID 12270958.
- Groffen AJ, Buskens CA, van Kuppevelt TH, Veerkamp JH, Monnens LA, van den Heuvel LP (May 1998). "Primary structure and high expression of human agrin in basement membranes of adult lung and kidney". Eur. J. Biochem. 254 (1): 123–8. doi:10.1046/j.1432-1327.1998.2540123.x. PMID 9652404.
- Tsen G, Halfter W, Kröger S, Cole GJ (1995). "Agrin is a heparan sulfate proteoglycan". J Biol Chem. 270 (7): 3392–3399. doi:10.1074/jbc.270.7.3392. PMID 7852425.
- Groffen AJ, Ruegg MA, Dijkman H, van de Velden TJ, Buskens CA, van den Born J, Assmann KJ, Monnens LA, Veerkamp JH, van den Heuvel LP (1998). "Agrin is a major heparan sulfate proteoglycan in the human glomerular basement membrane". J Histochem Cytochem. 46 (1): 19–27. doi:10.1177/002215549804600104. PMID 9405491.
- Magill C, Reist NE, Fallon JR, Nitkin RM, Wallace BG, McMahan UJ (1987). "Agrin". Prog. Brain Res. Progress in Brain Research. 71: 391–6. doi:10.1016/S0079-6123(08)61840-3. ISBN 978-0-444-80814-1. PMID 3035610.
- Sanes JR, Lichtman JW (November 2001). "Induction, assembly, maturation and maintenance of a postsynaptic apparatus". Nat. Rev. Neurosci. 2 (11): 791–805. doi:10.1038/35097557. PMID 11715056. S2CID 52802445.
- Glass DJ, Bowen DC, Stitt TN, Radziejewski C, Bruno J, Ryan TE, Gies DR, Shah S, Mattsson K, Burden SJ, DiStefano PS, Valenzuela DM, DeChiara TM, Yancopoulos GD (May 1996). "Agrin acts via a MuSK receptor complex". Cell. 85 (4): 513–23. doi:10.1016/S0092-8674(00)81252-0. PMID 8653787. S2CID 14930468.
- Sanes JR, Apel ED, Gautam M, Glass D, Grady RM, Martin PT, Nichol MC, Yancopoulos GD (May 1998). "Agrin receptors at the skeletal neuromuscular junction". Ann. N. Y. Acad. Sci. 841 (1): 1–13. Bibcode:1998NYASA.841....1S. doi:10.1111/j.1749-6632.1998.tb10905.x. PMID 9668217. S2CID 20097480.
- Gautam M, Noakes PG, Moscoso L, Rupp F, Scheller RH, Merlie JP, Sanes JR (May 1996). "Defective neuromuscular synaptogenesis in agrin-deficient mutant mice". Cell. 85 (4): 525–35. doi:10.1016/S0092-8674(00)81253-2. PMID 8653788. S2CID 12517490.
- Cheusova T, Khan MA, Schubert SW, Gavin AC, Buchou T, Jacob G, Sticht H, Allende J, Boldyreff B, Brenner HR, Hashemolhosseini S (July 2006). "Casein kinase 2-dependent serine phosphorylation of MuSK regulates acetylcholine receptor aggregation at the neuromuscular junction". Genes Dev. 20 (13): 1800–16. doi:10.1101/gad.375206. PMC 1522076. PMID 16818610.
- PDB: 1PZ7; Stetefeld, J.; Alexandrescu, A.T.; Maciejewski, M.W.; Jenny, M.; Rathgeb-Szabo, K.; Schulthess, T.; Landwehr, R.; Frank, S.; Ruegg, M.A.; Kammerer, R.A. (2004). "Modulation of agrin function by alternative splicing and Ca2+ binding". Structure. 12 (3): 503–515. doi:10.1016/j.str.2004.02.001. PMID 15016366.
- McDonnell KM, Grow WA (2004). "Reduced glycosaminoglycan sulfation diminishes the agrin signal transduction pathway". Dev. Neurosci. 26 (1): 1–10. doi:10.1159/000080706. PMID 15509893. S2CID 42558266.
- Thorup A, Dell'Accio F, Eldridge SE. "Regrowing knee cartilage: new animal studies show promise". The Conversation. Retrieved 2020-10-12.
- Eldridge SE, Barawi A, Wang H, Roelofs AJ, Kaneva M, Guan Z, et al. (September 2020). "Agrin induces long-term osteochondral regeneration by supporting repair morphogenesis". Science Translational Medicine. 12 (559): eaax9086. doi:10.1126/scitranslmed.aax9086. hdl:2164/15360. PMID 32878982. S2CID 221469142.
- Chakraborty S, Hong W (February 2018). "Linking Extracellular Matrix Agrin to the Hippo Pathway in Liver Cancer and Beyond". Cancers. 10 (2): 45. doi:10.3390/cancers10020045. PMC 5836077. PMID 29415512.
External links
- Human AGRN genome location and AGRN gene details page in the UCSC Genome Browser.