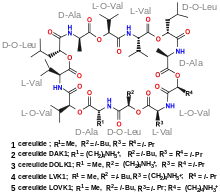
Cereulide
Cereulide is a toxin produced by some strains of Bacillus cereus. It is a potent cytotoxin that destroys mitochondria. It also causes nausea and vomiting.
 | |
| Names | |
|---|---|
| IUPAC name
cyclo[D-alanyl-N-oxa-L-valyl-L-valyl-N-oxa-D-leucyl-D-alanyl-N-oxa-L-valyl-L-valyl-N-oxa-D-leucyl-D-alanyl-N-oxa-L-valyl-L-valyl-N-oxa-D-leucyl] | |
| Other names
1,7,13,19,25,31-Hexaoxa-4,10,16,22,28,34-hexaazacyclohexatriacontane, cyclic peptide derivate; Cyclo(D-alanyl-3-methyl-L-2-hydroxybutanoyl-L-valyl-4-methyl-D-2-hydroxypentanoyl-D-alanyl-3-methyl-L-2-hydroxybutanoyl-L-valyl-4-methyl-D-2-hydroxypentanoyl-D-alanyl-3-methyl-L-2-hydroxybutanoyl-L-valyl-4-methyl-D-2-hydroxypentanoyl) | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C57 H96 N6 O18 (D-Ala-D-O-Leu-L-Val)3 | |
| Molar mass | 1152 |
| extremely low | |
| Hazards | |
| Main hazards | Neurotoxicant |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cereulide acts as ionophore with a high affinity to potassium cations. Exposure to cereulide causes loss of the membrane potential and uncoupling of oxidative phosphorylation in the mitochondria.[1][2] The nausea and vomiting is believed to be caused by cereulide's binding and activation of 5-HT3 receptors, leading to increased afferent vagus nerve stimulation.[3]
Cereulide is a cyclic dodecadepsipeptide resembling valinomycin; it contains three repeats of four amino acids: D-Oxy-Leu—D-Ala—L-Oxy-Val—L-Val. It is produced by a dedicated non-ribosomal peptide synthesis (NRPS) system in B. cereus.[4]
The spores of cereulide-producing strains of B. cereus are manyfold more heat resistant than spores of cereulide non-producers. The toxin has no loss of activity upon autoclaving, cooking, or baking.[1]
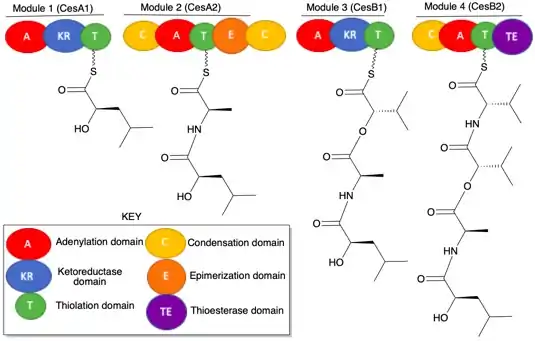
Biosynthesis
In Bacillus cereus, cereulide biosynthesis occurs by the non-ribosomal peptide synthetase of the heterodimer proteins CesA and CesB. In non-ribosomal peptide synthetase, individual amino acids are added, modified, and linked. Addition is facilitated by the adenylation (A) domain. Modification is accomplished by the ketoreductase (KR) and epimerization (E) domains. Finally, the growing peptides are linked by condensation domains. The transportation between domains is facilitated by a peptide carrier protein or thiolation (T) domain, which houses the growing peptide chain. Additionally, a thioesterase (TE) domain is used by the final module to cleave and cyclize the final peptide product.[4]
The peptides produced from both CesA and CesB are linked with an ester rather than amide bond; given the cyclic structure of cereulide, this cyclic ester (or lactone) linkage makes cereulide a depsipeptide.[4]
CesA is a 387 kDa heterodimer protein composed of CesA1 and CesA2 modules. CesA1 adds ketoisocaproic acid to the adenylation domain. The thiolation domain will then move the ketoisocaproic acid along the ketoreductase domain, which reduces ketoisocaproic acid into D-α-hydroxyisocaproic acid with the cofactor NADPH. In module CesA2, L-alanine is added to the adenylation domain. The condensation domain will facilitate a nucleophilic attack by the free amine on L-alanine onto the thioester of D-α-hydroxyisocaproic acid (D-HIC) on the CesA1 module. This event links the peptides and situates the growing peptide molecule on the thiolation domain of CesA2. Next, an epimerization domain changes the stereochemistry of L-alanine (L-Ala) into D-alanine (D-Ala).[4]
CesB is a 305 kDa heterodimer protein composed of CesB1 and CesB2 modules. CesB1 behaves almost identically to CesA1, where ketoisocaproic acid was added and reduced; however, the substrate α-ketoisovaleric acid is reduced to L- α-hydroxyisovaleric acid (L-HIV). Additionally, a condensation domain at the end of CesA (beyond CesA2) facilitates the ester formation between L-HIV and the D-HIC-D-Ala peptide.[4]
Next, CesB2 adds L-valine (L-Val) to the adenylation domain, and the condensation domain facilitates the nucleophilic attack of the amine on L-Val onto the D-HIC-D-Ala-L-HIV thioester, which creates a D-HIC-D-Ala-L-HIV-L-Val tetrapeptide on the thiolation domain of CesB2. Finally, the final thioesterase domain combines three units of the aforementioned tetrapeptide between the α-hydroxyl group of D-HIC and the thioester of a L-Val of another tetrapeptide. Ultimately, three esters are formed during this cyclization of 3 tetrapeptides. The resulting cyclic depsipeptide, which contains alternating units of esters and amides, is cereulide.[4]
References
- News on cereulide, the emetic toxin of Bacillus Cereus
- M. A. Andersson; R. Mikkola; J. Helin; M. C. Andersson; M. Salkinoja-Salonen (April 1998). "A Novel Sensitive Bioassay for Detection of Bacillus cereus Emetic Toxin and Related Depsipeptide Ionophores". Applied and Environmental Microbiology. 64 (4): 1338–1343. doi:10.1128/AEM.64.4.1338-1343.1998.
- Agata N, Ohta M, Mori M, Isobe M (1995). "A novel dodecadepsipeptide, cereulide, is an emetic toxin of Bacillus cereus". FEMS Microbiol Lett. 129 (1): 17–20. doi:10.1016/0378-1097(95)00119-P. PMID 7781985.
- Alonzo, Diego A.; Magarvey, Nathan A.; Schmeing, T. Martin (2015). "Characterization of Cereulide Synthetase, a Toxin-Producing Macromolecular Machine". PLOS ONE. 10 (6): e0128569. Bibcode:2015PLoSO..1028569A. doi:10.1371/journal.pone.0128569. PMC 4455996. PMID 26042597.
External links
- Peltola; et al. (2004). "News on cereulide, the emetic toxin of Bacillus Cereus".
- S. Pitchayawasin; M. Isobe; M. Kuse; T. Franz; N. Agata; M. Ohta (May 19, 2004). "Molecular diversity of cereulide" (PDF). Archived from the original (PDF) on March 13, 2005. Cite journal requires
|journal=(help) Archive.org
