Growth hormone-binding protein
Growth hormone-binding protein (GHBP) is a soluble carrier protein for growth hormone (GH).[1] The function of GHBP is still unknown.[2][3] Current research suggests that the protein is associated with regulation of the GH supply in the circulatory system as well as GH receptor function.[4]
| growth hormone receptor | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Symbol | GHR | ||||||
| NCBI gene | 2690 | ||||||
| HGNC | 4263 | ||||||
| OMIM | 600946 | ||||||
| RefSeq | NM_000163 | ||||||
| UniProt | P10912 | ||||||
| Other data | |||||||
| Locus | Chr. 5 p13-p12 | ||||||
| |||||||
Expression
In humans, GHBP is formed by post-translational modification after the complete transcription and translation of the growth hormone receptor (GHR) gene into the cell-surface receptor protein. The gene that codes for GHR (and inherently GHBP) is on Chromosome 5.[5] A precursor messenger RNA (mRNA) from the complete gene first is transcribed and then spliced to encode the full receptor protein. This mature mRNA is composed of exons. Exons are peptide encoding regions of DNA genes that remain in the transcript after splicing and during the maturation of mRNA. The mRNA transcript encodes for a receptor protein that is made up of three distinct parts: an intracellular domain, a transmembrane domain, and an extracellular domain.[6] Specifically, part of exon 2 and exons 3-7 of the GHR gene will translate to amino acids that make up the extracellular domain of GHR. This extracellular domain physically binds GH in the receptor-ligand interaction.[4]
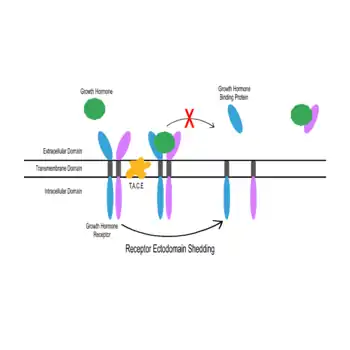
Receptor ectodomain shedding
When the extracellular domain of GHR is proteolytically cleaved (see: proteolytic cleavage) from the rest of the receptor protein, the extracellular domain is released as the water-soluble, carrier protein GHBP.[8] As the extracellular domain alone, the polypeptide consists of 246 amino acids[4] and is roughly 60 kDA in size.[9] This cleaving process is called “receptor ectodomain shedding.[10] In humans and rabbits, tumor-necrosis factor alpha converting enzyme (T.A.C.E.) is postulated to play a significant role in the post-translational processing activity that sheds GHBP from GHR.[11][12] Studies show that this activity primarily occurs in the liver.[13] When growth hormone is bound to two dimerized GH receptors, the shedding activity is inhibited. This occurs because when the ligand binds to the receptors, a conformational change occurs in them that potentially blocks the proteolytic activity of T.A.C.E.[12][14]
Alternative splicing
In humans, studies have shown that alternative splicing of the GHR gene can lead to increased rates of proteolysis. For example, a deletion within the mRNA that encodes part of the transmembrane domain of the protein effectively leads to non-translation of the intracellular domain due to the presence of a stop codon.[15] This truncated version of GHR is cleaved more frequently into GHBP and may potentially explain the reasoning behind increased concentrations of GHBP present in some tissues.[16]
In mouse and rat models, the extracellular domain is formed primarily through alternative splicing of the precursor GHR mRNA to form a mature transcript that translate GHBP alone. These animals can potentially shed GHBP via post-translational modification as well, although this activity is minimal.[12][17]
Function
The fully characterized, physiological consequences of GHBP binding GH is not known at this time.[18]
Binding stoichiometry
GHBP and GHR bind growth hormone by using the ligand’s C-terminal disulfide bridges. In GH, two disulfide bridges link four alpha-helices, and these helices make direct contact with the extracellular domain of GHR.[19] Two receptor molecules are pre-dimerized upon GH binding, so it always binds in a 1:2 ratio.[20] Assays estimate that growth hormone and growth hormone binding protein form a natural complex at a 1:1 ratio for transport and preservation of the ligand through the bloodstream.[21][22] However, some sources have shown that high physiological concentration of GHBP will result in a 1:2 ratio.[23][24] When the cysteine amino acids in GHBP are mutated and the disulfide bridges are disrupted, the stability of the ligand to bind to the active site of the GHBP is significantly lessened.[25]
Activation
The clearance rate, or the rate at which the carrier protein is broken down, for GHBP alone is much faster than when it is bound to its ligand.[4] Additionally, current literature provides evidence that the carrier-protein prolongs the half-life of growth hormone through its binding with the ligand.[26] One purpose of GHBP can be inferred: to maintain the level of GH in the blood, as roughly half of its concentration is complexed with GHBP.[27] Yet this could be confounded by the fact that GH binding to GHBP prevents the ligand from binding to GHR and ultimately proteolytic activity.[28] Another function is that GHBP displays competitive inhibition for GH against the GHR receptor.[13]
Studies elucidate another aspect of GHBP physiological role: The proteolytic cleavage activity that forms GHBP ultimately regulates GHR production in humans as well as rats.[14][29] If there is low GHBP concentration then there are high levels of GHR expression. Conversely, high levels of GHBP protein show negative correlation with levels of growth hormone receptor expression.[13][30]
Isoforms
Exon 3
Studies have identified a GHBP isoform that exists due to gene polymorphism, or variable expression of the allele. These isoforms differ based on whether or not the extracellular domain of GHR includes the amino acids encoded by exon 3 - exon 3 is either kept (dominant) or spliced out (recessive).[31] As human are diploid, they may genotype as homozygous dominant (two copies of the allele retain exon 3), heterozygous (one copy with Exon 3, and one without), or homozygous recessive (two copies of the allele without exon 3).[32] Studies have shown that the two isoforms can co-exist as dimerized GH receptors, as E3+/E3+, E3+/E3-, or E3-/E3-.[31]
Furthermore, the two isoforms both exist in the blood as GHBP. However, they may have separate functions that are poorly understood. The presence or absence of exon 3 in humans is individual-specific, but one study suggests that gender may play a role in this variable splicing, as females were shown to express higher levels of deleted-exon 3 GHBP in their blood.[33] The evolutionary reason for exon-3 variable GHBP expression has not clearly be defined, and the isoforms in the blood have not been shown to differ with respect to GH affinity, which is unusual for an isoform that is missing an entire exon.[34]
References
- Baumann G (Dec 2002). "Growth hormone binding protein. The soluble growth hormone receptor". Minerva Endocrinologica. 27 (4): 265–76. PMID 12511849.
- Baumann G, Stolar MW, Amburn K, Barsano CP, DeVries BC (Jan 1986). "A specific growth hormone-binding protein in human plasma: initial characterization". The Journal of Clinical Endocrinology and Metabolism. 62 (1): 134–41. doi:10.1210/jcem-62-1-134. PMID 3940261.
- Baumann G (1999). "Growth Hormone Binding Proteins". In Bengtsson B (ed.). Growth Hormone. Endocrine Updates. 4. Springer. pp. 37–57. doi:10.1007/978-1-4615-5163-8_3. ISBN 978-1-4613-7351-3.
- Schilbach K, Bidlingmaier M (2015). "Growth hormone binding protein - physiological and analytical aspects". Best Practice & Research. Clinical Endocrinology & Metabolism. 29 (5): 671–83. doi:10.1016/j.beem.2015.06.004. PMID 26522453.
- Barton DE, Foellmer BE, Wood WI, Francke U (1989). "Chromosome mapping of the growth hormone receptor gene in man and mouse". Cytogenetics and Cell Genetics. 50 (2–3): 137–41. doi:10.1159/000132743. PMID 2776481.
- "GHR growth hormone receptor [Homo sapiens (human)]". NCBI.
- Fisker S (Feb 2006). "Physiology and pathophysiology of growth hormone-binding protein: methodological and clinical aspects". Growth Hormone & IGF Research. 16 (1): 1–28. doi:10.1016/j.ghir.2005.11.001. PMID 16359897.
- Leung DW, Spencer SA, Cachianes G, Hammonds RG, Collins C, Henzel WJ, Barnard R, Waters MJ, Wood WI (1987). "Growth hormone receptor and serum binding protein: purification, cloning and expression". Nature. 330 (6148): 537–43. doi:10.1038/330537a0. PMID 2825030. S2CID 4370204.
- Fisker S (2006). "Physiology and pathophysiology of growth hormone-binding protein: methodological and clinical aspects". Growth Hormone & IGF Research. 16 (1): 1–28. doi:10.1016/j.ghir.2005.11.001. PMID 16359897.
- Alele J, Jiang J, Goldsmith JF, Yang X, Maheshwari HG, Black RA, Baumann G, Frank SJ (1998). "Blockade of growth hormone receptor shedding by a metalloprotease inhibitor". Endocrinology. 139 (4): 1927–35. doi:10.1210/endo.139.4.5906. PMID 9528979.
- Black RA, Rauch CT, Kozlosky CJ, Peschon JJ, Slack JL, Wolfson MF, Castner BJ, Stocking KL, Reddy P, Srinivasan S, Nelson N, Boiani N, Schooley KA, Gerhart M, Davis R, Fitzner JN, Johnson RS, Paxton RJ, March CJ, Cerretti DP (1997). "A metalloproteinase disintegrin that releases tumour-necrosis factor-alpha from cells". Nature. 385 (6618): 729–33. doi:10.1038/385729a0. PMID 9034190. S2CID 4251053.
- Schantl JA, Roza M, Van Kerkhof P, Strous GJ (Jan 2004). "The growth hormone receptor interacts with its sheddase, the tumour necrosis factor-alpha-converting enzyme (TACE)". The Biochemical Journal. 377 (Pt 2): 379–84. doi:10.1042/BJ20031321. PMC 1223864. PMID 14519102.
- Amit T, Youdim MB, Hochberg Z (2000). "Clinical review 112: Does serum growth hormone (GH) binding protein reflect human GH receptor function?". The Journal of Clinical Endocrinology and Metabolism. 85 (3): 927–32. doi:10.1210/jcem.85.3.6461. PMID 10720017.
- Zhang Y, Guan R, Jiang J, Kopchick JJ, Black RA, Baumann G, Frank SJ (Jul 2001). "Growth hormone (GH)-induced dimerization inhibits phorbol ester-stimulated GH receptor proteolysis". The Journal of Biological Chemistry. 276 (27): 24565–73. doi:10.1074/jbc.M101281200. PMID 11309389.
- Ross RJ, Esposito N, Shen XY, Von Laue S, Chew SL, Dobson PR, Postel-Vinay MC, Finidori J (Mar 1997). "A short isoform of the human growth hormone receptor functions as a dominant negative inhibitor of the full-length receptor and generates large amounts of binding protein". Molecular Endocrinology. 11 (3): 265–73. doi:10.1210/mend.11.3.9901. PMID 9058373.
- Iida K, Takahashi Y, Kaji H, Nose O, Okimura Y, Abe H, Chihara K (1998). "Growth hormone (GH) insensitivity syndrome with high serum GH-binding protein levels caused by a heterozygous splice site mutation of the GH receptor gene producing a lack of intracellular domain". The Journal of Clinical Endocrinology and Metabolism. 83 (2): 531–7. doi:10.1210/jcem.83.2.4601. PMID 9467570.
- Edens A, Southard JN, Talamantes F (Dec 1994). "Mouse growth hormone-binding protein and growth hormone receptor transcripts are produced from a single gene by alternative splicing". Endocrinology. 135 (6): 2802–5. doi:10.1210/endo.135.6.7988474. PMID 7988474.
- Postel-Vinay MC (1996). "Growth hormone- and prolactin-binding proteins: soluble forms of receptors". Hormone Research. 45 (3–5): 178–81. doi:10.1159/000184783. PMID 8964579.
- Gráf L, Borvendég J, Barát E, Hermann I, Patthy A (Jul 1976). "Reactivity and biological importance of the disulfide bonds in human growth hormone". FEBS Letters. 66 (2): 233–7. doi:10.1016/0014-5793(76)80511-X. PMID 955085. S2CID 32938660.
- Brooks AJ, Waters MJ (Sep 2010). "The growth hormone receptor: mechanism of activation and clinical implications". Nature Reviews. Endocrinology. 6 (9): 515–25. doi:10.1038/nrendo.2010.123. PMID 20664532. S2CID 23639819.
- Sundström M, Lundqvist T, Rödin J, Giebel LB, Milligan D, Norstedt G (Dec 1996). "Crystal structure of an antagonist mutant of human growth hormone, G120R, in complex with its receptor at 2.9 A resolution". The Journal of Biological Chemistry. 271 (50): 32197–203. doi:10.1074/jbc.271.50.32197. PMID 8943276.
- Goodwill D (2004). "Growth Hormone". RCSB Protein Data Bank. doi:10.2210/rcsb_pdb/mom_2004_4.
- Baumann G, Lowman HB, Mercado M, Wells JA (May 1994). "The stoichiometry of growth hormone-binding protein complexes in human plasma: comparison with cell surface receptors". The Journal of Clinical Endocrinology and Metabolism. 78 (5): 1113–8. doi:10.1210/jcem.78.5.8175967. PMID 8175967.
- de Vos AM, Ultsch M, Kossiakoff AA (Jan 1992). "Human growth hormone and extracellular domain of its receptor: crystal structure of the complex". Science. 255 (5042): 306–12. doi:10.1126/science.1549776. PMID 1549776.
- Junnila RK, Wu Z, Strasburger CJ. The role of human growth hormone's C-terminal disulfide bridge. Growth Horm IGF Res. 2013;23(3):62-7
- Carlsson LM, Rosberg S, Vitangcol RV, Wong WL, Albertsson-Wikland K (Aug 1993). "Analysis of 24-hour plasma profiles of growth hormone (GH)-binding protein, GH/GH-binding protein-complex, and GH in healthy children". The Journal of Clinical Endocrinology and Metabolism. 77 (2): 356–61. doi:10.1210/jcem.77.2.8345039. PMID 8345039.
- Baumann G, Amburn K, Shaw MA (Mar 1988). "The circulating growth hormone (GH)-binding protein complex: a major constituent of plasma GH in man". Endocrinology. 122 (3): 976–84. doi:10.1210/endo-122-3-976. PMID 3342762.
- Barnard R, Waters MJ (1997). "The serum growth hormone binding protein: pregnant with possibilities". The Journal of Endocrinology. 153 (1): 1–14. doi:10.1677/joe.0.1530001. PMID 9135564.
- Wang X, Jiang J, Warram J, Baumann G, Gan Y, Menon RK, Denson LA, Zinn KR, Frank SJ (Jun 2008). "Endotoxin-induced proteolytic reduction in hepatic growth hormone (GH) receptor: a novel mechanism for GH insensitivity". Molecular Endocrinology. 22 (6): 1427–37. doi:10.1210/me.2007-0561. PMC 2422827. PMID 18323468.
- Maheshwari H, Lillioja S, Castillo CE, Mercado M, Baumann G (1995). "Growth hormone-binding protein in human lymph". The Journal of Clinical Endocrinology and Metabolism. 80 (12): 3582–4. doi:10.1210/jcem.80.12.8530602. PMID 8530602.
- Stallings-Mann ML, Ludwiczak RL, Klinger KW, Rottman F (Oct 1996). "Alternative splicing of exon 3 of the human growth hormone receptor is the result of an unusual genetic polymorphism". Proceedings of the National Academy of Sciences of the United States of America. 93 (22): 12394–9. doi:10.1073/pnas.93.22.12394. PMC 38002. PMID 8901592.
- Seidel B, Glasow A, Schutt M, Kiess W, Wu Z, Strasburger CJ, Kratzsch J (Mar 2003). "Association between the GH receptor/exon 3 genotype and the level of exon 3-positive GH-binding protein in human serum". European Journal of Endocrinology. 148 (3): 317–24. doi:10.1530/eje.0.1480317. PMID 12611612.
- Kratzsch J, Wu Z, Kiess W, Dehmel B, Bosse-Henck A, Reuter W, Pflaum CD, Strasburger CJ (2001). "The exon 3-retaining and the exon 3-deleted forms of the growth hormone-binding protein (GHBP) in human serum are regulated differently". Clinical Endocrinology. 54 (1): 61–8. doi:10.1046/j.1365-2265.2001.01177.x. PMID 11167927. S2CID 23577976.
- Kucukhuseyin O, Toptas B, Timirci-Kahraman O, Isbir S, Karsidag K, Isbir T (2015-06-01). "The Effect of GHR/exon-3 Polymorphism and Serum GH, IGF-1 and IGFBP-3 Levels in Diabetes and Coronary Heart Disease". In Vivo. 29 (3): 371–8. PMID 25977383.
External links
- somatotropin-binding+protein at the US National Library of Medicine Medical Subject Headings (MeSH)
- Baumann G, Shaw MA, Winter RJ (Oct 1987). "Absence of the plasma growth hormone-binding protein in Laron-type dwarfism". The Journal of Clinical Endocrinology and Metabolism. 65 (4): 814–6. doi:10.1210/jcem-65-4-814. PMID 3654924.