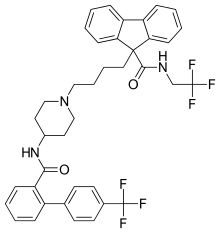
Lomitapide
Lomitapide , sold under the brand name Juxtapid in the US and as Lojuxta in the EU) is a medication used as a lipid-lowering agent for the treatment of familial hypercholesterolemia, developed by Aegerion Pharmaceuticals.[3] It has been tested in clinical trials as single treatment and in combinations with atorvastatin, ezetimibe and fenofibrate.[4][5]
 | |
| Clinical data | |
|---|---|
| Trade names | Juxtapid (US), Lojuxta (EU) |
| Other names | AEGR-773, BMS-201038 |
| License data |
|
| Routes of administration | Oral (capsules) |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C39H37F6N3O2 |
| Molar mass | 693.734 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
The US Food and Drug Administration (FDA) approved lomitapide on 21 December 2012, as an orphan drug to reduce LDL cholesterol, total cholesterol, apolipoprotein B, and non-high-density lipoprotein (non-HDL) cholesterol in patients with homozygous familial hypercholesterolemia (HoFH).[6]
On 31 May 2013 the European Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion with a unanimous vote recommending a marketing authorization for lomitapide.[2] On 31 July 2013 the European Commission approved lomitapide as an adjunct to a low-fat diet and other lipid-lowering medicinal products with or without low density lipoprotein (LDL) apheresis in adult patients with HoFH.
Mechanism of action
Lomitapide inhibits the microsomal triglyceride transfer protein (MTP or MTTP) which is necessary for very low-density lipoprotein (VLDL) assembly and secretion in the liver.[3][7]
On 24 December 2012, drug manufacturer Aegerion announced they had been approved by the FDA to as "an adjunct to a low-fat diet and other lipid-lowering treatments...in patients with homozygous familial hypercholesterolemia (HoFH)."[8][9]
Side effects
In a Phase III study, lomitapide led to elevated aminotransferase levels and fat accumulation in the liver.[7]
References
- "Juxtapid- lomitapide mesylate capsule". DailyMed. Retrieved 27 January 2021.
- "Lojuxta EPAR". European Medicines Agency (EMA). Retrieved 27 January 2021.
- H. Spreitzer (12 March 2007). "Neue Wirkstoffe – BMS-201038". Österreichische Apothekerzeitung (in German) (6/2007): 268.
- Horwich TB, Fonarow GC (July 2008). "Measures of obesity and outcomes after myocardial infarction". Circulation. 118 (5): 469–71. doi:10.1161/CIRCULATIONAHA.108.792689. PMID 18663098.
- "Aegerion Pharmaceuticals, Inc. Announces AEGR-733 Phase II Data Demonstrates Significant Lowering of LDL Cholesterol with Promising Hepatic Safety Profile". Business Wire. 9 November 2008. Archived from the original on 2012-02-29.
- "FDA approves new orphan drug for rare cholesterol disorder". U.S. Food and Drug Administration. Archived from the original on 28 January 2013.
- Cuchel M, Bloedon LT, Szapary PO, Kolansky DM, Wolfe ML, Sarkis A, et al. (January 2007). "Inhibition of microsomal triglyceride transfer protein in familial hypercholesterolemia". The New England Journal of Medicine. 356 (2): 148–56. doi:10.1056/NEJMoa061189. PMID 17215532.
- "FDA Approves Juxtapid for Homozygous Familial Hypercholesteolemia". 26 December 2012. Archived from the original on 29 December 2012. Retrieved 1 January 2013.
- "FDA Approves Aegerion Pharmaceuticals' Juxtapid (lomitapide) Capsules for Homozygous Familial Hypercholesterolemia (HoFH)" (Press release). Aegerion Pharmaceuticals. 24 December 2012. Archived from the original on 22 September 2016. Retrieved 1 January 2013.
External links
- "Lomitapide". Drug Information Portal. U.S. National Library of Medicine.