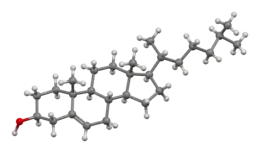
Cholesterol
Cholesterol (from the Ancient Greek chole- (bile) and stereos (solid), followed by the chemical suffix -ol for an alcohol) is an organic molecule. It is a sterol (or modified steroid),[3] a type of lipid.[1] Cholesterol is biosynthesized by all animal cells and is an essential structural component of animal cell membranes.
 | |
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
cholest-5-en-3β-ol | |
| Systematic IUPAC name
(3S,8S,9S,10R,13R,14S,17R)-10,13-dimethyl-17-[(2R)-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol | |
| Other names
Cholesterin, Cholesteryl alcohol[1] | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.321 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H46O | |
| Molar mass | 386.65 g/mol |
| Appearance | white crystalline powder[2] |
| Density | 1.052 g/cm3 |
| Melting point | 148 to 150 °C (298 to 302 °F; 421 to 423 K) [2] |
| Boiling point | 360 °C (680 °F; 633 K) (decomposes) |
| 0.095 mg/L (30 °C)[1] | |
| Solubility | soluble in acetone, benzene, chloroform, ethanol, ether, hexane, isopropyl myristate, methanol |
| -284.2·10−6 cm3/mol | |
| Hazards | |
| Flash point | 209.3 ±12.4 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
| Types of fats in food |
|---|
| See also |
Cholesterol also serves as a precursor for the biosynthesis of steroid hormones, bile acid[4] and vitamin D. Cholesterol is the principal sterol synthesized by all animals. In vertebrates, hepatic cells typically produce the greatest amounts. It is absent among prokaryotes (bacteria and archaea), although there are some exceptions, such as Mycoplasma, which require cholesterol for growth.[5]
François Poulletier de la Salle first identified cholesterol in solid form in gallstones in 1769. However, it was not until 1815 that chemist Michel Eugène Chevreul named the compound "cholesterine".[6][7]
Physiology
Cholesterol is essential for all animal life, with each cell capable of synthesizing it by way of a complex 37-step process. This begins with the mevalonate or HMG-CoA reductase pathway, the target of statin drugs, which encompasses the first 18 steps. This is followed by 19 additional steps to convert the resulting lanosterol into cholesterol.
A human male weighing 68 kg (150 lb) normally synthesizes about 1 gram (1,000 mg) of cholesterol per day, and his body contains about 35 g, mostly contained within the cell membranes. Typical daily cholesterol dietary intake for a man in the United States is 307 mg.[8]
Most ingested cholesterol is esterified, which causes it to be poorly absorbed by the gut. The body also compensates for absorption of ingested cholesterol by reducing its own cholesterol synthesis.[9] For these reasons, cholesterol in food, seven to ten hours after ingestion, has little, if any effect on concentrations of cholesterol in the blood.[10] However, during the first seven hours after ingestion of cholesterol, as absorbed fats are being distributed around the body within extracellular water by the various lipoproteins (which transport all fats in the water outside cells), the concentrations increase.[11]
Plants make cholesterol in very small amounts. [12] In larger quantities they produce phytosterols, chemically similar substances which can compete with cholesterol for reabsorption in the intestinal tract, thus potentially reducing cholesterol reabsorption.[13] When intestinal lining cells absorb phytosterols, in place of cholesterol, they usually excrete the phytosterol molecules back into the GI tract, an important protective mechanism. The intake of naturally occurring phytosterols, which encompass plant sterols and stanols, ranges between ≈200–300 mg/day depending on eating habits.[14] Specially designed vegetarian experimental diets have been produced yielding upwards of 700 mg/day.[15]
Membranes
Cholesterol composes about 30% of all animal cell membranes. It is required to build and maintain membranes and modulates membrane fluidity over the range of physiological temperatures. The hydroxyl group of each cholesterol molecule interacts with water molecules surrounding the membrane, as do the polar heads of the membrane phospholipids and sphingolipids, while the bulky steroid and the hydrocarbon chain are embedded in the membrane, alongside the nonpolar fatty-acid chain of the other lipids. Through the interaction with the phospholipid fatty-acid chains, cholesterol increases membrane packing, which both alters membrane fluidity[16] and maintains membrane integrity so that animal cells do not need to build cell walls (like plants and most bacteria). The membrane remains stable and durable without being rigid, allowing animal cells to change shape and animals to move.
The structure of the tetracyclic ring of cholesterol contributes to the fluidity of the cell membrane, as the molecule is in a trans conformation making all but the side chain of cholesterol rigid and planar.[17] In this structural role, cholesterol also reduces the permeability of the plasma membrane to neutral solutes,[18] hydrogen ions, and sodium ions.[19]
Gates
Within the cell membrane, cholesterol also functions in intracellular transport, cell signaling and nerve conduction. Cholesterol is essential for the structure and function of invaginated caveolae and clathrin-coated pits, including caveola-dependent and clathrin-dependent endocytosis. The role of cholesterol in endocytosis of these types can be investigated by using methyl beta cyclodextrin (MβCD) to remove cholesterol from the plasma membrane.
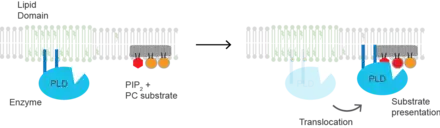
Substrate presentation
Cholesterol regulates the biological process of substrate presentation and the enzymes that use substrate presentation as a mechanism of their activation. (PLD2) is a well-defined example of an enzyme activated by substrate presentation.[20] The enzyme is palmitoylated causing the enzyme to traffic to cholesterol dependent lipid domains sometimes called "lipid rafts". The substrate of phospholipase D is phosphatidylcholine (PC) which is unsaturated and is of low abundance in lipid rafts. PC localizes to the disordered region of the cell along with the polyunsaturated lipid phosphatidylinositol 4,5-bisphosphate (PIP2). PLD2 has a PIP2 binding domain. When PIP2 concentration in the membrane increases, PLD2 leaves the cholesterol dependent domains and binds to PIP2 where it then gains access to its substrate PC and commences catalysis based on substrate presentation.
Signaling
Cholesterol is also implicated in cell signaling processes, assisting in the formation of lipid rafts in the plasma membrane, which brings receptor proteins in close proximity with high concentrations of second messenger molecules.[21] In multiple layers, cholesterol and phospholipids, both electrical insulators, can facilitate speed of transmission of electrical impulses along nerve tissue. For many neuron fibers, a myelin sheath, rich in cholesterol since it is derived from compacted layers of Schwann cell membrane, provides insulation for more efficient conduction of impulses.[22] Demyelination (loss of some of these Schwann cells) is believed to be part of the basis for multiple sclerosis.
Cholesterol binds to and affects the gating of a number of ion channels such as the nicotinic acetylcholine receptor, GABAA receptor, and the inward-rectifier potassium channel.[23] Cholesterol also activates the estrogen-related receptor alpha (ERRα), and may be the endogenous ligand for the receptor.[24][25] The constitutively active nature of the receptor may be explained by the fact that cholesterol is ubiquitous in the body.[25] Inhibition of ERRα signaling by reduction of cholesterol production has been identified as a key mediator of the effects of statins and bisphosphonates on bone, muscle, and macrophages.[24][25] On the basis of these findings, it has been suggested that the ERRα should be de-orphanized and classified as a receptor for cholesterol.[24][25]
Chemical precursor
Within cells, cholesterol is also a precursor molecule for several biochemical pathways. For example, it is the precursor molecule for the synthesis of vitamin D in the calcium metabolism and all steroid hormones, including the adrenal gland hormones cortisol and aldosterone, as well as the sex hormones progesterone, estrogens, and testosterone, and their derivatives.[4][26]
Metabolism
Cholesterol is recycled in the body. The liver excretes cholesterol into biliary fluids, which are then stored in the gallbladder, which then excretes them in a non-esterified form (via bile) into the digestive tract. Typically, about 50% of the excreted cholesterol is reabsorbed by the small intestine back into the bloodstream.[27]
Biosynthesis and regulation
Biosynthesis
All animal cells manufacture cholesterol, for both membrane structure and other uses, with relative production rates varying by cell type and organ function. About 80% of total daily cholesterol production occurs in the liver and the intestines;[28] other sites of higher synthesis rates include the brain, the adrenal glands, and the reproductive organs.
Synthesis within the body starts with the mevalonate pathway where two molecules of acetyl CoA condense to form acetoacetyl-CoA. This is followed by a second condensation between acetyl CoA and acetoacetyl-CoA to form 3-hydroxy-3-methylglutaryl CoA (HMG-CoA).[29]
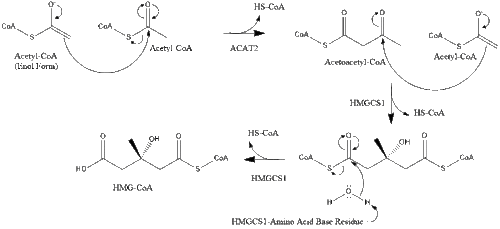
This molecule is then reduced to mevalonate by the enzyme HMG-CoA reductase. Production of mevalonate is the rate-limiting and irreversible step in cholesterol synthesis and is the site of action for statins (a class of cholesterol-lowering drugs).
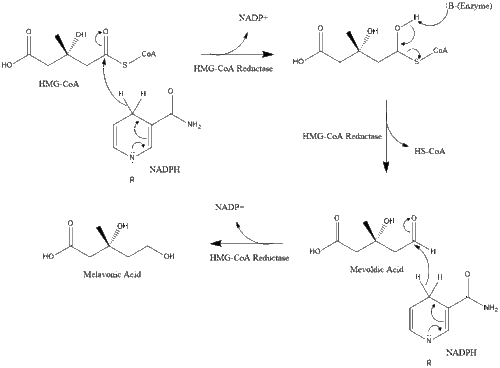
Mevalonate is finally converted to isopentenyl pyrophosphate (IPP) through two phosphorylation steps and one decarboxylation step that requires ATP.
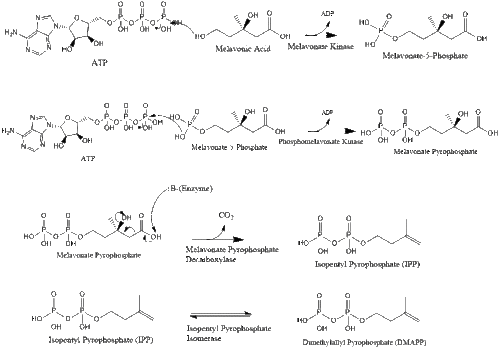
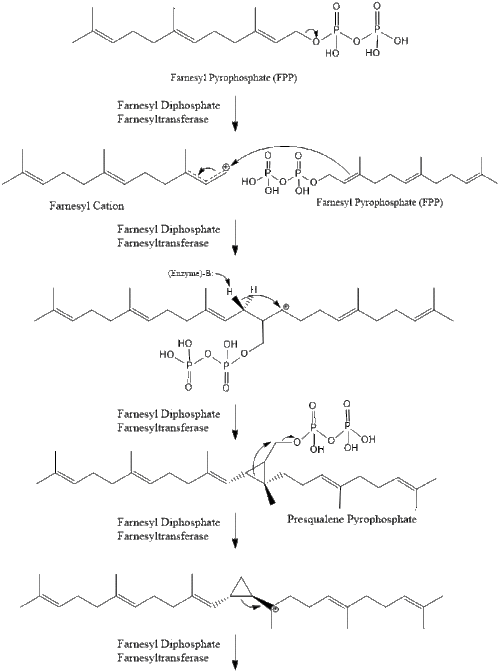
Three molecules of isopentenyl pyrophosphate condense to form farnesyl pyrophosphate through the action of geranyl transferase.
_reaction.svg.png.webp)
Two molecules of farnesyl pyrophosphate then condense to form squalene by the action of squalene synthase in the endoplasmic reticulum.[29]
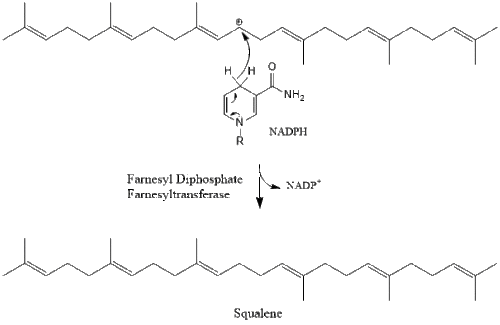
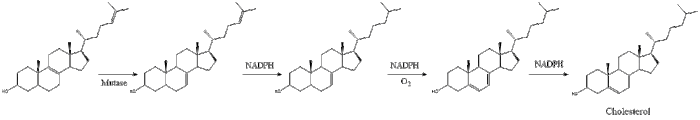
Oxidosqualene cyclase then cyclizes squalene to form lanosterol.
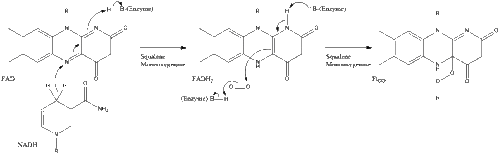
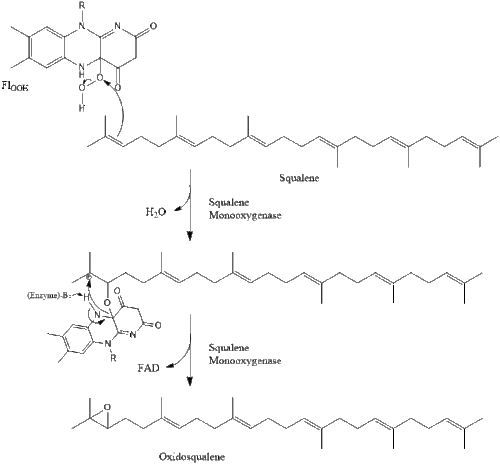
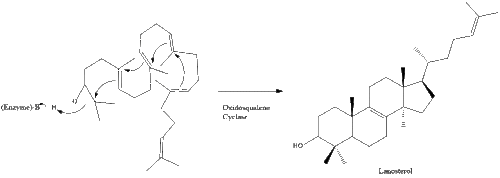
Finally, lanosterol is converted to cholesterol via either of two pathways, the Block pathway, or the Kandutsch-Russell pathway.[30][31][32][33][34] The final 19 steps to cholesterol contain NADPH and oxygen to help oxidize methyl groups for removal of carbons, mutases to move alkene groups, and NADH to help reduce ketones.
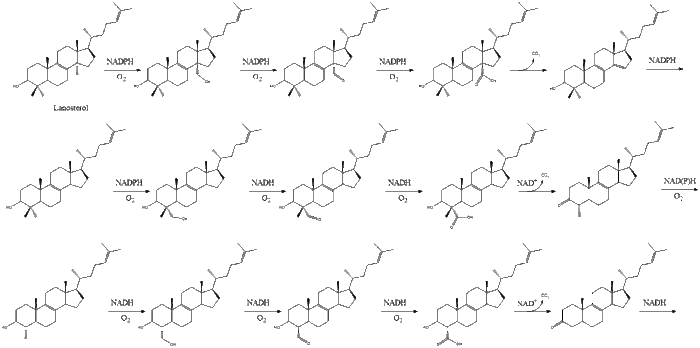
Konrad Bloch and Feodor Lynen shared the Nobel Prize in Physiology or Medicine in 1964 for their discoveries concerning some of the mechanisms and methods of regulation of cholesterol and fatty acid metabolism.[35]
Regulation of cholesterol synthesis
Biosynthesis of cholesterol is directly regulated by the cholesterol levels present, though the homeostatic mechanisms involved are only partly understood. A higher intake from food leads to a net decrease in endogenous production, whereas lower intake from food has the opposite effect. The main regulatory mechanism is the sensing of intracellular cholesterol in the endoplasmic reticulum by the protein SREBP (sterol regulatory element-binding protein 1 and 2).[36] In the presence of cholesterol, SREBP is bound to two other proteins: SCAP (SREBP cleavage-activating protein) and INSIG-1. When cholesterol levels fall, INSIG-1 dissociates from the SREBP-SCAP complex, which allows the complex to migrate to the Golgi apparatus. Here SREBP is cleaved by S1P and S2P (site-1 protease and site-2 protease), two enzymes that are activated by SCAP when cholesterol levels are low.
The cleaved SREBP then migrates to the nucleus, and acts as a transcription factor to bind to the sterol regulatory element (SRE), which stimulates the transcription of many genes. Among these are the low-density lipoprotein (LDL) receptor and HMG-CoA reductase. The LDL receptor scavenges circulating LDL from the bloodstream, whereas HMG-CoA reductase leads to an increase of endogenous production of cholesterol.[37] A large part of this signaling pathway was clarified by Dr. Michael S. Brown and Dr. Joseph L. Goldstein in the 1970s. In 1985, they received the Nobel Prize in Physiology or Medicine for their work. Their subsequent work shows how the SREBP pathway regulates expression of many genes that control lipid formation and metabolism and body fuel allocation.
Cholesterol synthesis can also be turned off when cholesterol levels are high. HMG-CoA reductase contains both a cytosolic domain (responsible for its catalytic function) and a membrane domain. The membrane domain senses signals for its degradation. Increasing concentrations of cholesterol (and other sterols) cause a change in this domain's oligomerization state, which makes it more susceptible to destruction by the proteosome. This enzyme's activity can also be reduced by phosphorylation by an AMP-activated protein kinase. Because this kinase is activated by AMP, which is produced when ATP is hydrolyzed, it follows that cholesterol synthesis is halted when ATP levels are low.[38]
Plasma transport and regulation of absorption
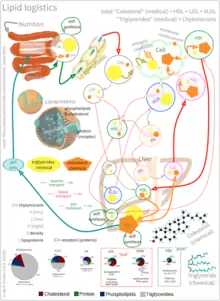
As an isolated molecule, cholesterol is only minimally soluble in water, or hydrophilic. Because of this, it dissolves in blood at exceedingly small concentrations. To be transported effectively, cholesterol is instead packaged within lipoproteins, complex discoidal particles with exterior amphiphilic proteins and lipids, whose outward-facing surfaces are water-soluble and inward-facing surfaces are lipid-soluble. This allows it to travel through the blood via emulsification. Unbound cholesterol, being amphipathic, is transported in the monolayer surface of the lipoprotein particle along with phospholipids and proteins. Cholesterol esters bound to fatty acid, on the other hand, are transported within the fatty hydrophilic core of the lipoprotein, along with triglyceride.[39]
There are several types of lipoproteins in the blood. In order of increasing density, they are chylomicrons, very-low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), low-density lipoprotein (LDL), and high-density lipoprotein (HDL). Lower protein/lipid ratios make for less dense lipoproteins. Cholesterol within different lipoproteins is identical, although some is carried as its native "free" alcohol form (the cholesterol-OH group facing the water surrounding the particles), while others as fatty acyl esters, known also as cholesterol esters, within the particles.[39]
Lipoprotein particles are organized by complex apolipoproteins, typically 80–100 different proteins per particle, which can be recognized and bound by specific receptors on cell membranes, directing their lipid payload into specific cells and tissues currently ingesting these fat transport particles. These surface receptors serve as unique molecular signatures, which then help determine fat distribution delivery throughout the body.[39]
Chylomicrons, the least dense cholesterol transport molecules, contain apolipoprotein B-48, apolipoprotein C, and apolipoprotein E (the principal cholesterol carrier in the brain[40]) in their shells. Chylomicrons carry fats from the intestine to muscle and other tissues in need of fatty acids for energy or fat production. Unused cholesterol remains in more cholesterol-rich chylomicron remnants, and taken up from here to the bloodstream by the liver.[39]
VLDL molecules are produced by the liver from triacylglycerol and cholesterol which was not used in the synthesis of bile acids. These molecules contain apolipoprotein B100 and apolipoprotein E in their shells, and can be degraded by lipoprotein lipase on the artery wall to IDL. This arterial wall cleavage allows absorption of triacylglycerol and increases concentration of circulating cholesterol. IDL molecules are then consumed in two processes: half is metabolized by HTGL and taken up by the LDL receptor on the liver cell surfaces, while the other half continues to lose triacylglycerols in the bloodstream until they become cholesterol laden LDL particles.[39]
LDL particles are the major blood cholesterol carriers. Each one contains approximately 1,500 molecules of cholesterol ester. LDL molecule shells contain just one molecule of apolipoprotein B100, recognized by LDL receptors in peripheral tissues. Upon binding of apolipoprotein B100, many LDL receptors concentrate in clathrin-coated pits. Both LDL and its receptor form vesicles within a cell via endocytosis. These vesicles then fuse with a lysosome, where the lysosomal acid lipase enzyme hydrolyzes the cholesterol esters. The cholesterol can then be used for membrane biosynthesis or esterified and stored within the cell, so as to not interfere with the cell membranes.[39]
LDL receptors are used up during cholesterol absorption, and its synthesis is regulated by SREBP, the same protein that controls the synthesis of cholesterol de novo, according to its presence inside the cell. A cell with abundant cholesterol will have its LDL receptor synthesis blocked, to prevent new cholesterol in LDL molecules from being taken up. Conversely, LDL receptor synthesis proceeds when a cell is deficient in cholesterol.[39]
When this process becomes unregulated, LDL molecules without receptors begin to appear in the blood. These LDL molecules are oxidized and taken up by macrophages, which become engorged and form foam cells. These foam cells often become trapped in the walls of blood vessels and contribute to atherosclerotic plaque formation. Differences in cholesterol homeostasis affect the development of early atherosclerosis (carotid intima-media thickness).[41] These plaques are the main causes of heart attacks, strokes, and other serious medical problems, leading to the association of so-called LDL cholesterol (actually a lipoprotein) with "bad" cholesterol.[38]
HDL particles are thought to transport cholesterol back to the liver, either for excretion or for other tissues that synthesize hormones, in a process known as reverse cholesterol transport (RCT).[42] Large numbers of HDL particles correlates with better health outcomes,[43] whereas low numbers of HDL particles is associated with atheromatous disease progression in the arteries.
Metabolism, recycling and excretion
Cholesterol is susceptible to oxidation and easily forms oxygenated derivatives called oxysterols. Three different mechanisms can form these: autoxidation, secondary oxidation to lipid peroxidation, and cholesterol-metabolizing enzyme oxidation. A great interest in oxysterols arose when they were shown to exert inhibitory actions on cholesterol biosynthesis.[44] This finding became known as the “oxysterol hypothesis”. Additional roles for oxysterols in human physiology include their participation in bile acid biosynthesis, function as transport forms of cholesterol, and regulation of gene transcription.[45]
In biochemical experiments radiolabelled forms of cholesterol, such as tritiated-cholesterol are used. These derivatives undergo degradation upon storage and it is essential to purify cholesterol prior to use. Cholesterol can be purified using small Sephadex LH-20 columns.[46]
Cholesterol is oxidized by the liver into a variety of bile acids.[47] These, in turn, are conjugated with glycine, taurine, glucuronic acid, or sulfate. A mixture of conjugated and nonconjugated bile acids, along with cholesterol itself, is excreted from the liver into the bile. Approximately 95% of the bile acids are reabsorbed from the intestines, and the remainder are lost in the feces.[48] The excretion and reabsorption of bile acids forms the basis of the enterohepatic circulation, which is essential for the digestion and absorption of dietary fats. Under certain circumstances, when more concentrated, as in the gallbladder, cholesterol crystallises and is the major constituent of most gallstones (lecithin and bilirubin gallstones also occur, but less frequently).[49] Every day, up to 1 g of cholesterol enters the colon. This cholesterol originates from the diet, bile, and desquamated intestinal cells, and can be metabolized by the colonic bacteria. Cholesterol is converted mainly into coprostanol, a nonabsorbable sterol that is excreted in the feces.
Although cholesterol is a steroid generally associated with mammals, the human pathogen Mycobacterium tuberculosis is able to completely degrade this molecule and contains a large number of genes that are regulated by its presence.[50] Many of these cholesterol-regulated genes are homologues of fatty acid β-oxidation genes, but have evolved in such a way as to bind large steroid substrates like cholesterol.[51][52]
Dietary sources
Animal fats are complex mixtures of triglycerides, with lesser amounts of both the phospholipids and cholesterol molecules from which all animal (and human) cell membranes are constructed. Since all animal cells manufacture cholesterol, all animal-based foods contain cholesterol in varying amounts.[53] Major dietary sources of cholesterol include red meat, egg yolks and whole eggs, liver, kidney, giblets, fish oil, and butter.[54] Human breast milk also contains significant quantities of cholesterol.[55]
Plant cells synthesize cholesterol as a precursor for other compounds, such as phytosterols and steroidal glycoalkaloids, with cholesterol remaining in plant foods only in minor amounts or absent.[54][56] Some plant foods, such as avocado, flax seeds and peanuts, contain phytosterols, which compete with cholesterol for absorption in the intestines, reduce the absorption of both dietary and bile cholesterol.[57] A typical diet contributes on the order of 0.2 gram of phytosterols, which is not enough to have a significant impact on blocking cholesterol absorption. Phytosterols intake can be supplemented through the use of phytosterol-containing functional foods or dietary supplements that are recognized as having potential to reduce levels of LDL-cholesterol.[58]
Medical guidelines and recommendations
In 2016, the United States Department of Agriculture Dietary Guidelines Advisory Committee recommended that Americans eat as little dietary cholesterol as possible.[59] Most foods that are rich in cholesterol are also high in saturated fat and thereby may increase the risk of cardiovascular disease.[60]
Some supplemental guidelines have recommended doses of phytosterols in the 1.6–3.0 grams per day range (Health Canada, EFSA, ATP III, FDA). A recent meta-analysis demonstrating a 12% reduction in LDL-cholesterol at a mean dose of 2.1 grams per day.[61] However, the benefits of a diet supplemented with phytosterols have also been questioned.[62]
Clinical significance
Hypercholesterolemia
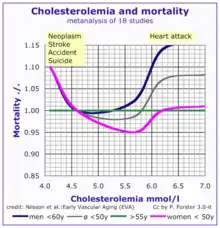
According to the lipid hypothesis, elevated levels of cholesterol in the blood lead to atherosclerosis which may increase the risk of heart attack, stroke, and peripheral artery disease. Since higher blood LDL – especially higher LDL concentrations and smaller LDL particle size – contributes to this process more than the cholesterol content of the HDL particles,[63] LDL particles are often termed "bad cholesterol". High concentrations of functional HDL, which can remove cholesterol from cells and atheromas, offer protection and are commonly referred to as "good cholesterol". These balances are mostly genetically determined, but can be changed by body composition, medications, diet,[64] and other factors.[65] A 2007 study demonstrated that blood total cholesterol levels have an exponential effect on cardiovascular and total mortality, with the association more pronounced in younger subjects. Because cardiovascular disease is relatively rare in the younger population, the impact of high cholesterol on health is larger in older people.[66]
Elevated levels of the lipoprotein fractions, LDL, IDL and VLDL, rather than the total cholesterol level, correlate with the extent and progress of atherosclerosis.[67] Conversely, the total cholesterol can be within normal limits, yet be made up primarily of small LDL and small HDL particles, under which conditions atheroma growth rates are high. A post hoc analysis of the IDEAL and the EPIC prospective studies found an association between high levels of HDL cholesterol (adjusted for apolipoprotein A-I and apolipoprotein B) and increased risk of cardiovascular disease, casting doubt on the cardioprotective role of "good cholesterol".[68][69]
One in 250 adults can have a genetic mutation for the LDL cholesterol receptor that causes them to have familial hypercholerolemia. Inherited high cholesterol can also include genetic mutations in the PCSK9 gene and the gene for apolipoprotein B.[70]
Elevated cholesterol levels are treated with a strict diet consisting of low saturated fat, trans fat-free, low cholesterol foods,[71][72] often followed by one of various hypolipidemic agents, such as statins, fibrates, cholesterol absorption inhibitors, nicotinic acid derivatives or bile acid sequestrants.[73] There are several international guidelines on the treatment of hypercholesterolaemia.[74]
Human trials using HMG-CoA reductase inhibitors, known as statins, have repeatedly confirmed that changing lipoprotein transport patterns from unhealthy to healthier patterns significantly lowers cardiovascular disease event rates, even for people with cholesterol values currently considered low for adults.[75] Studies have shown that reducing LDL cholesterol levels by about 38.7 mg/dL with the use of statins can reduce cardiovascular disease and stroke risk by about 21%.[76] Studies have also found that statins reduce atheroma progression.[77] As a result, people with a history of cardiovascular disease may derive benefit from statins irrespective of their cholesterol levels (total cholesterol below 5.0 mmol/L [193 mg/dL]),[78] and in men without cardiovascular disease, there is benefit from lowering abnormally high cholesterol levels ("primary prevention").[79] Primary prevention in women was originally practiced only by extension of the findings in studies on men,[80] since, in women, none of the large statin trials conducted prior to 2007 demonstrated a significant reduction in overall mortality or in cardiovascular endpoints.[81] Meta-analyses have demonstrated significant reductions in all-cause and cardiovascular mortality, without significant heterogeneity by sex.[82]
| Level | Interpretation | |
|---|---|---|
| mg/dL | mmol/L | |
| < 200 | < 5.2 | Desirable level (lower risk) |
| 200–240 | 5.2–6.2 | Borderline high risk |
| > 240 | > 6.2 | High risk |
The 1987 report of National Cholesterol Education Program, Adult Treatment Panels suggests the total blood cholesterol level should be: < 200 mg/dL normal blood cholesterol, 200–239 mg/dL borderline-high, > 240 mg/dL high cholesterol.[83] The American Heart Association provides a similar set of guidelines for total (fasting) blood cholesterol levels and risk for heart disease:[84] Statins are effective in lowering LDL cholesterol and widely used for primary prevention in people at high risk of cardiovascular disease, as well as in secondary prevention for those who have developed cardiovascular disease.[85]
More current testing methods determine LDL ("bad") and HDL ("good") cholesterol separately, allowing cholesterol analysis to be more nuanced. The desirable LDL level is considered to be less than 130 mg/dL (2.6 mmol/L),[86] although a newer upper limit of 70 mg/dL (1.8 mmol/L) can be considered in higher-risk individuals based on some of the above-mentioned trials. A ratio of total cholesterol to HDL—another useful measure—of far less than 5:1 is thought to be healthier.

Total cholesterol is defined as the sum of HDL, LDL, and VLDL. Usually, only the total, HDL, and triglycerides are measured. For cost reasons, the VLDL is usually estimated as one-fifth of the triglycerides and the LDL is estimated using the Friedewald formula (or a variant): estimated LDL = [total cholesterol] − [total HDL] − [estimated VLDL]. VLDL can be calculated by dividing total triglycerides by five. Direct LDL measures are used when triglycerides exceed 400 mg/dL. The estimated VLDL and LDL have more error when triglycerides are above 400 mg/dL.[87]
In the Framingham Heart Study, in subjects over 50 years of age, they found an 11% increase overall and 14% increase in cardiovascular disease mortality per 1 mg/dL per year drop in total cholesterol levels. The researchers attributed this phenomenon to the fact that people with severe chronic diseases or cancer tend to have below-normal cholesterol levels.[88] This explanation is not supported by the Vorarlberg Health Monitoring and Promotion Programme, in which men of all ages and women over 50 with very low cholesterol were likely to die of cancer, liver diseases, and mental diseases. This result indicates the low-cholesterol effect occurs even among younger respondents, contradicting the previous assessment among cohorts of older people that this is a proxy or marker for frailty occurring with age.[89]
Although there is a link between cholesterol and atherosclerosis as discussed above, a 2014 review concluded there is insufficient evidence to support the recommendation of high consumption of polyunsaturated fatty acids and low consumption of total saturated fats for cardiovascular health.[90] A 2016 review concluded that HDL cholesterol was inversely linked to mortality in people over age 60 years, and there was either no link between LDL and mortality, or that lower LDL was linked to a higher mortality risk, especially in older adults.[91]
Hypocholesterolemia
Abnormally low levels of cholesterol are termed hypocholesterolemia. Research into the causes of this state is relatively limited, but some studies suggest a link with depression, cancer, and cerebral hemorrhage. In general, the low cholesterol levels seem to be a consequence, rather than a cause, of an underlying illness.[66] A genetic defect in cholesterol synthesis causes Smith–Lemli–Opitz syndrome, which is often associated with low plasma cholesterol levels. Hyperthyroidism, or any other endocrine disturbance which causes upregulation of the LDL receptor, may result in hypocholesterolemia.[92]
Cholesterol testing
The American Heart Association recommends testing cholesterol every 4–6 years for people aged 20 years or older.[93] A separate set of American Heart Association guidelines issued in 2013 indicates that patients taking statin medications should have their cholesterol tested 4–12 weeks after their first dose and then every 3–12 months thereafter.[94]
A blood sample after 12-hour fasting is taken by a doctor, or a home cholesterol-monitoring device is used to measure a lipid profile, an approach used to estimate a person's lipoproteins, the vastly more important issue because lipoproteins have always been concordant with outcomes though the lipid profile is commonly discordant LDL Particle Number and Risk of Future Cardiovascular Disease in the Framingham Offspring Study.
The lipid profile measures: (a) total cholesterol, (b) cholesterol associated with HDL (i.e. Higher Density {than water} Lipids-transported-within-proteins) particles ("which can regress arterial disease"), (c) triglycerides and (d) (by a calculation and assumptions) cholesterol carried by LDL (i.e. Lower Density {than water} Lipids-transported-within-proteins) particles ("which drive arterial disease").
It is recommended to test cholesterol at least every five years if a person has total cholesterol of 5.2 mmol/L or more (200+ mg/dL), or if a man over age 45 or a woman over age 50 has HDL-C values less than 1 mmol/L (40 mg/dL), or there are other drivers heart disease and stroke. Additional drivers of heart disease include diabetes mellitus, hypertension (or use of anti-hypertensive medication), low HDL level, family history of coronary artery disease (CAD) and hypercholesterolemia, and cigarette smoking.[95]
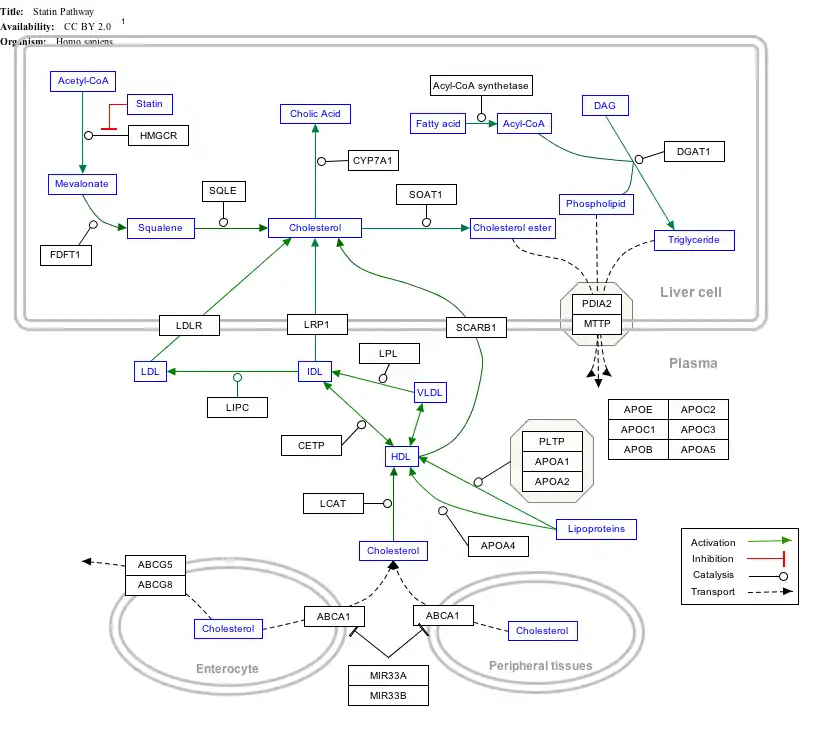
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- The interactive pathway map can be edited at WikiPathways: "Statin_Pathway_WP430".
Cholesteric liquid crystals
Some cholesterol derivatives (among other simple cholesteric lipids) are known to generate the liquid crystalline "cholesteric phase". The cholesteric phase is, in fact, a chiral nematic phase, and it changes colour when its temperature changes. This makes cholesterol derivatives useful for indicating temperature in liquid-crystal display thermometers and in temperature-sensitive paints.
Stereoisomers
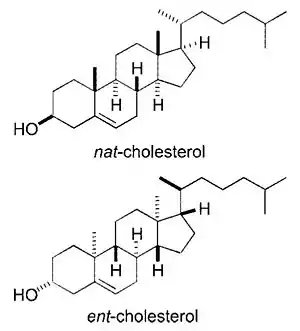
Cholesterol has 256 stereoisomers that arise from its 8 stereocenters, although only two of the stereoisomers are of biochemical significance (nat-cholesterol and ent-cholesterol, for natural and enantiomer, respectively),[96][97] and only one occurs naturally (nat-cholesterol).
See also
- Arcus senilis "Cholesterol ring" in the eyes
- Cardiovascular disease
- Cholesterol embolism
- Cholesterol total synthesis
- Familial hypercholesterolemia
- Hypercholesterolemia "High Cholesterol"
- Hypocholesterolemia "Low Cholesterol"
- Janus-faced molecule
- List of cholesterol in foods
- Niemann–Pick disease Type C
- Oxycholesterol
- Remnant cholesterol
Additional images
 Cholesterol units conversion
Cholesterol units conversion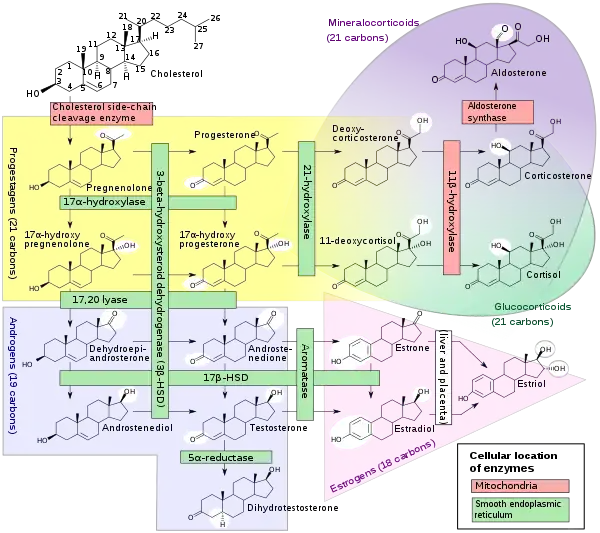 Steroidogenesis, using cholesterol as building material
Steroidogenesis, using cholesterol as building material Space-filling model of the Cholesterol molecule
Space-filling model of the Cholesterol molecule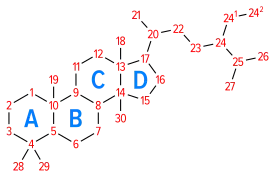 Numbering of the steroid nuclei
Numbering of the steroid nuclei
References
- "Cholesterol, 57-88-5". PubChem, National Library of Medicine, US National Institutes of Health. 9 November 2019. Retrieved 14 November 2019.
- "Safety (MSDS) data for cholesterol". Archived from the original on 12 July 2007. Retrieved 20 October 2007.
- Cholesterol at the US National Library of Medicine Medical Subject Headings (MeSH)
- Hanukoglu I (December 1992). "Steroidogenic enzymes: structure, function, and role in regulation of steroid hormone biosynthesis". The Journal of Steroid Biochemistry and Molecular Biology. 43 (8): 779–804. doi:10.1016/0960-0760(92)90307-5. PMID 22217824. S2CID 112729.
- Razin S, Tully JG (May 1970). "Cholesterol requirement of mycoplasmas". Journal of Bacteriology. 102 (2): 306–10. doi:10.1128/JB.102.2.306-310.1970. PMC 247552. PMID 4911537.
- Chevreul (1816) "Recherches chimiques sur les corps gras, et particulièrement sur leurs combinaisons avec les alcalis. Sixième mémoire. Examen des graisses d'homme, de mouton, de boeuf, de jaguar et d'oie" (Chemical researches on fatty substances, and particularly on their combinations o filippos ine kapios with alkalis. Sixth memoir. Study of human, sheep, beef, jaguar and goose fat), Annales de Chimie et de Physique, 2 : 339–372. From page 346 : "Je nommerai cholesterine, de χολη, bile, et στερεος, solide, la substance cristallisée des calculs biliares humains, ... " (I will name cholesterine — from χολη (bile) and στερεος (solid) — the crystalized substance from human gallstones ... )
- Olson RE (February 1998). "Discovery of the lipoproteins, their role in fat transport and their significance as risk factors". The Journal of Nutrition. 128 (2 Suppl): 439S–443S. doi:10.1093/jn/128.2.439S. PMID 9478044.
- "National Health and Nutrition Examination Survey" (PDF). United States Center for Disease Control. Retrieved 28 January 2012.
- Lecerf JM, de Lorgeril M (July 2011). "Dietary cholesterol: from physiology to cardiovascular risk". The British Journal of Nutrition. 106 (1): 6–14. doi:10.1017/S0007114511000237. PMID 21385506.
- Soliman GA (June 2018). "Dietary Cholesterol and the Lack of Evidence in Cardiovascular Disease". Nutrients. 10 (6): 780. doi:10.3390/nu10060780. PMC 6024687. PMID 29914176.
- Dubois C, Armand M, Mekki N, Portugal H, Pauli AM, Bernard PM, et al. (November 1994). "Effects of increasing amounts of dietary cholesterol on postprandial lipemia and lipoproteins in human subjects". Journal of Lipid Research. 35 (11): 1993–2007. PMID 7868978.
- Behrman EJ, Gopalan V (2005). Scovell WM (ed.). "Cholesterol and Plants". Journal of Chemical Education. 82 (12): 1791. Bibcode:2005JChEd..82.1791B. doi:10.1021/ed082p1791.
- John S, Sorokin AV, Thompson PD (February 2007). "Phytosterols and vascular disease". Current Opinion in Lipidology. 18 (1): 35–40. doi:10.1097/MOL.0b013e328011e9e3. PMID 17218830. S2CID 29213889.
- Jesch ED, Carr TP (June 2017). "Food Ingredients That Inhibit Cholesterol Absorption". Preventive Nutrition and Food Science. 22 (2): 67–80. doi:10.3746/pnf.2017.22.2.67. PMC 5503415. PMID 28702423.
- Agren JJ, Tvrzicka E, Nenonen MT, Helve T, Hänninen O (February 2001). "Divergent changes in serum sterols during a strict uncooked vegan diet in patients with rheumatoid arthritis". The British Journal of Nutrition. 85 (2): 137–9. doi:10.1079/BJN2000234. PMID 11242480.
- Sadava D, Hillis DM, Heller HC, Berenbaum MR (2011). Life: The Science of Biology 9th Edition. San Francisco: Freeman. pp. 105–114. ISBN 978-1-4292-4646-0.
- Ohvo-Rekilä H, Ramstedt B, Leppimäki P, Slotte JP (January 2002). "Cholesterol interactions with phospholipids in membranes". Progress in Lipid Research. 41 (1): 66–97. doi:10.1016/S0163-7827(01)00020-0. PMID 11694269.
- Yeagle PL (October 1991). "Modulation of membrane function by cholesterol". Biochimie. 73 (10): 1303–10. doi:10.1016/0300-9084(91)90093-G. PMID 1664240.
- Haines TH (July 2001). "Do sterols reduce proton and sodium leaks through lipid bilayers?". Progress in Lipid Research. 40 (4): 299–324. doi:10.1016/S0163-7827(01)00009-1. PMID 11412894.
- Petersen EN, Chung HW, Nayebosadri A, Hansen SB (December 2016). "Kinetic disruption of lipid rafts is a mechanosensor for phospholipase D". Nature Communications. 7: 13873. Bibcode:2016NatCo...713873P. doi:10.1038/ncomms13873. PMC 5171650. PMID 27976674.
- Incardona JP, Eaton S (April 2000). "Cholesterol in signal transduction". Current Opinion in Cell Biology. 12 (2): 193–203. doi:10.1016/S0955-0674(99)00076-9. PMID 10712926.
- Pawlina W, Ross MW (2006). Histology: a text and atlas: with correlated cell and molecular biology. Philadelphia: Lippincott Wiliams & Wilkins. p. 230. ISBN 978-0-7817-5056-1.
- Levitan I, Singh DK, Rosenhouse-Dantsker A (2014). "Cholesterol binding to ion channels". Frontiers in Physiology. 5: 65. doi:10.3389/fphys.2014.00065. PMC 3935357. PMID 24616704.
- Wei W, Schwaid AG, Wang X, Wang X, Chen S, Chu Q, et al. (March 2016). "Ligand Activation of ERRα by Cholesterol Mediates Statin and Bisphosphonate Effects". Cell Metabolism. 23 (3): 479–91. doi:10.1016/j.cmet.2015.12.010. PMC 4785078. PMID 26777690.
- Nuclear Receptors in Development and Disease. Elsevier Science. 17 May 2017. pp. 88–. ISBN 978-0-12-802196-5.
- Payne AH, Hales DB (December 2004). "Overview of steroidogenic enzymes in the pathway from cholesterol to active steroid hormones". Endocrine Reviews. 25 (6): 947–70. doi:10.1210/er.2003-0030. PMID 15583024.
- Cohn JS, Kamili A, Wat E, Chung RW, Tandy S (February 2010). "Dietary phospholipids and intestinal cholesterol absorption". Nutrients. 2 (2): 116–27. doi:10.3390/nu2020116. PMC 3257636. PMID 22254012.
- "How it's made: Cholesterol production in your body". Harvard Health Publishing. Retrieved 18 October 2018.
- "Biosynthesis and Regulation of Cholesterol (with Animation)". PharmaXChange.info. 17 September 2013.
- "Cholesterol metabolism (includes both Bloch and Kandutsch-Russell pathways) (Mus musculus) - WikiPathways". www.wikipathways.org. Retrieved 2 February 2021.
- Singh, P; Saxena, R; Srinivas, G; Pande, G; Chattopadhyay, A (2013). "Cholesterol biosynthesis and homeostasis in regulation of the cell cycle". PloS one. 8 (3): e58833. doi:10.1371/journal.pone.0058833. PMID 23554937.
- "Kandutsch-Russell pathway". pubchem.ncbi.nlm.nih.gov. Retrieved 2 February 2021.
- Berg J (2002). Biochemistry. New York: WH Freeman. ISBN 978-0-7167-3051-4.
- Rhodes CM, Stryer L, Tasker R (1995). Biochemistry (4th ed.). San Francisco: W.H. Freeman. pp. 280, 703. ISBN 978-0-7167-2009-6.
- "The Nobel Prize in Physiology or Medicine, 1964". =Nobel Prize, Nobel Media.
- Espenshade PJ, Hughes AL (2007). "Regulation of sterol synthesis in eukaryotes". Annual Review of Genetics. 41: 401–27. doi:10.1146/annurev.genet.41.110306.130315. PMID 17666007.
- Brown MS, Goldstein JL (May 1997). "The SREBP pathway: regulation of cholesterol metabolism by proteolysis of a membrane-bound transcription factor". Cell. 89 (3): 331–40. doi:10.1016/S0092-8674(00)80213-5. PMID 9150132. S2CID 17882616.
- Tymoczko JL, Berg T, Stryer L, Berg JM (2002). Biochemistry. San Francisco: W.H. Freeman. pp. 726–727. ISBN 978-0-7167-4955-4.
- Patton KT, Thibodeau GA (2010). Anatomy and Physiology (7 ed.). Mosby/Elsevier. ISBN 978-9996057762. Retrieved 20 March 2019.
- Mahley RW (July 2016). "Apolipoprotein E: from cardiovascular disease to neurodegenerative disorders". Journal of Molecular Medicine. 94 (7): 739–46. doi:10.1007/s00109-016-1427-y. PMC 4921111. PMID 27277824.
- Weingärtner O, Pinsdorf T, Rogacev KS, Blömer L, Grenner Y, Gräber S, et al. (October 2010). Federici M (ed.). "The relationships of markers of cholesterol homeostasis with carotid intima-media thickness". PLOS ONE. 5 (10): e13467. Bibcode:2010PLoSO...513467W. doi:10.1371/journal.pone.0013467. PMC 2956704. PMID 20976107.
- Lewis GF, Rader DJ (June 2005). "New insights into the regulation of HDL metabolism and reverse cholesterol transport". Circulation Research. 96 (12): 1221–32. doi:10.1161/01.RES.0000170946.56981.5c. PMID 15976321.
- Gordon DJ, Probstfield JL, Garrison RJ, Neaton JD, Castelli WP, Knoke JD, et al. (January 1989). "High-density lipoprotein cholesterol and cardiovascular disease. Four prospective American studies". Circulation. 79 (1): 8–15. doi:10.1161/01.CIR.79.1.8. PMID 2642759.
- Kandutsch AA, Chen HW, Heiniger HJ (August 1978). "Biological activity of some oxygenated sterols". Science. 201 (4355): 498–501. Bibcode:1978Sci...201..498K. doi:10.1126/science.663671. PMID 663671.
- Russell DW (December 2000). "Oxysterol biosynthetic enzymes". Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids. 1529 (1–3): 126–35. doi:10.1016/S1388-1981(00)00142-6. PMID 11111082.
- Hanukoglu I, Jefcoate CR (1980). "Pregnenolone separation from cholesterol using Sephadex LH-20 mini-columns". Journal of Chromatography A. 190 (1): 256–262. doi:10.1016/S0021-9673(00)85545-4.
- Javitt NB (December 1994). "Bile acid synthesis from cholesterol: regulatory and auxiliary pathways". FASEB Journal. 8 (15): 1308–11. doi:10.1096/fasebj.8.15.8001744. PMID 8001744. S2CID 20302590.
- Wolkoff AW, Cohen DE (February 2003). "Bile acid regulation of hepatic physiology: I. Hepatocyte transport of bile acids". American Journal of Physiology. Gastrointestinal and Liver Physiology. 284 (2): G175-9. doi:10.1152/ajpgi.00409.2002. PMID 12529265.
- Marschall HU, Einarsson C (June 2007). "Gallstone disease". Journal of Internal Medicine. 261 (6): 529–42. doi:10.1111/j.1365-2796.2007.01783.x. PMID 17547709. S2CID 8609639.
- Wipperman MF, Sampson NS, Thomas ST (2014). "Pathogen roid rage: cholesterol utilization by Mycobacterium tuberculosis". Critical Reviews in Biochemistry and Molecular Biology. 49 (4): 269–93. doi:10.3109/10409238.2014.895700. PMC 4255906. PMID 24611808.
- Thomas ST, Sampson NS (April 2013). "Mycobacterium tuberculosis utilizes a unique heterotetrameric structure for dehydrogenation of the cholesterol side chain". Biochemistry. 52 (17): 2895–904. doi:10.1021/bi4002979. PMC 3726044. PMID 23560677.
- Wipperman MF, Yang M, Thomas ST, Sampson NS (October 2013). "Shrinking the FadE proteome of Mycobacterium tuberculosis: insights into cholesterol metabolism through identification of an α2β2 heterotetrameric acyl coenzyme A dehydrogenase family". Journal of Bacteriology. 195 (19): 4331–41. doi:10.1128/JB.00502-13. PMC 3807453. PMID 23836861.
- William W C (2003). Lipid analysis: isolation, separation, identification, and structural analysis of lipids. Ayr, Scotland: Oily Press. ISBN 978-0-9531949-5-7.
- "Cholesterol content in foods, rank order per 100 g; In: USDA Food Composition Databases". United States Department of Agriculture. 2019. Retrieved 4 March 2019.
- Jensen RG, Hagerty MM, McMahon KE (June 1978). "Lipids of human milk and infant formulas: a review". The American Journal of Clinical Nutrition. 31 (6): 990–1016. doi:10.1093/ajcn/31.6.990. PMID 352132.
- Sonawane PD, Pollier J, Panda S, Szymanski J, Massalha H, Yona M, et al. (December 2016). "Plant cholesterol biosynthetic pathway overlaps with phytosterol metabolism". Nature Plants. 3 (1): 16205. doi:10.1038/nplants.2016.205. PMID 28005066. S2CID 5518449.
- De Smet E, Mensink RP, Plat J (July 2012). "Effects of plant sterols and stanols on intestinal cholesterol metabolism: suggested mechanisms from past to present". Molecular Nutrition & Food Research. 56 (7): 1058–72. doi:10.1002/mnfr.201100722. PMID 22623436.
- European Food Safety Authority, Journal (2010). "Scientific opinion on the substantiation of health claims related to plant sterols and plant stanols and maintenance of normal blood cholesterol concentrations".
- "2015–2020 Dietary Guidelines: Answers to Your Questions. What are "eating patterns" and why does the 2015–2020 Dietary Guidelines focus on them?". ChooseMyPlate.gov, US Department of Agriculture. January 2016. Retrieved 17 February 2017.
- Soliman GA (June 2018). "Dietary Cholesterol and the Lack of Evidence in Cardiovascular Disease". Nutrients. 10 (6): 780. doi:10.3390/nu10060780. PMID 29914176.
- Ras RT, Geleijnse JM, Trautwein EA (July 2014). "LDL-cholesterol-lowering effect of plant sterols and stanols across different dose ranges: a meta-analysis of randomised controlled studies". The British Journal of Nutrition. 112 (2): 214–9. doi:10.1017/S0007114514000750. PMC 4071994. PMID 24780090.
- Weingärtner O, Böhm M, Laufs U (February 2009). "Controversial role of plant sterol esters in the management of hypercholesterolaemia". European Heart Journal. 30 (4): 404–9. doi:10.1093/eurheartj/ehn580. PMC 2642922. PMID 19158117.
- Brunzell JD, Davidson M, Furberg CD, Goldberg RB, Howard BV, Stein JH, Witztum JL (April 2008). "Lipoprotein management in patients with cardiometabolic risk: consensus statement from the American Diabetes Association and the American College of Cardiology Foundation". Diabetes Care. 31 (4): 811–22. doi:10.2337/dc08-9018. PMID 18375431.
- Department of Health (UK), NHS Choices, "More evidence for Mediterranean diet". 8 March 2011. Access date: Nov 11, 2015
- Durrington P (August 2003). "Dyslipidaemia". Lancet. 362 (9385): 717–31. doi:10.1016/S0140-6736(03)14234-1. PMID 12957096. S2CID 208792416.
- Lewington S, Whitlock G, Clarke R, Sherliker P, Emberson J, Halsey J, et al. (December 2007). "Blood cholesterol and vascular mortality by age, sex, and blood pressure: a meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths". Lancet. 370 (9602): 1829–39. doi:10.1016/S0140-6736(07)61778-4. PMID 18061058. S2CID 54293528.
- "Detection, Evaluation and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) Final Report" (PDF). National Institutes of Health. National Heart, Lung and Blood Institute. 1 September 2002. Retrieved 27 October 2008.
- van der Steeg WA, Holme I, Boekholdt SM, Larsen ML, Lindahl C, Stroes ES, et al. (February 2008). "High-density lipoprotein cholesterol, high-density lipoprotein particle size, and apolipoprotein A-I: significance for cardiovascular risk: the IDEAL and EPIC-Norfolk studies". Journal of the American College of Cardiology. 51 (6): 634–42. doi:10.1016/j.jacc.2007.09.060. PMID 18261682.
- Robinson JG, Wang S, Jacobson TA (November 2012). "Meta-analysis of comparison of effectiveness of lowering apolipoprotein B versus low-density lipoprotein cholesterol and nonhigh-density lipoprotein cholesterol for cardiovascular risk reduction in randomized trials". The American Journal of Cardiology. 110 (10): 1468–76. doi:10.1016/j.amjcard.2012.07.007. PMID 22906895.
- "Familial Hypercholesterolemia (FH)". www.heart.org. Retrieved 2 August 2019.
- "How Can I Lower High Cholesterol" (PDF). American Heart Association. Archived from the original (PDF) on 21 December 2015. Retrieved 3 April 2011.
- "Diseases and Conditions. High cholesterol: Olive oil, Foods with added plant sterols or stanols, Other changes to your diet". Mayo Clinic. 2012. Retrieved 11 November 2015.
- National Institute for Health and Clinical Excellence. Clinical guideline 67: Lipid modification. London, 2008.
- Mannu GS, Zaman MJ, Gupta A, Rehman HU, Myint PK (October 2012). "Update on guidelines for management of hypercholesterolemia". Expert Review of Cardiovascular Therapy. 10 (10): 1239–49. doi:10.1586/erc.12.94. PMID 23190064. S2CID 5451203.
- Kizer JR, Madias C, Wilner B, Vaughan CJ, Mushlin AI, Trushin P, et al. (May 2010). "Relation of different measures of low-density lipoprotein cholesterol to risk of coronary artery disease and death in a meta-regression analysis of large-scale trials of statin therapy". The American Journal of Cardiology. 105 (9): 1289–96. doi:10.1016/j.amjcard.2009.12.051. PMC 2917836. PMID 20403481.
- Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, et al. (June 2019). "2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines". Circulation. 139 (25): e1082–e1143. doi:10.1161/CIR.0000000000000625. PMC 7403606. PMID 30586774.
- Nicholls SJ (August 2008). "Rosuvastatin and progression of atherosclerosis". Expert Review of Cardiovascular Therapy. 6 (7): 925–33. doi:10.1586/14779072.6.7.925. PMID 18666843. S2CID 46419583.
- Heart Protection Study Collaborative Group (July 2002). "MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial". Lancet. 360 (9326): 7–22. doi:10.1016/S0140-6736(02)09327-3. PMID 12114036. S2CID 35836642.
- Shepherd J, Cobbe SM, Ford I, Isles CG, Lorimer AR, MacFarlane PW, et al. (November 1995). "Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group". The New England Journal of Medicine. 333 (20): 1301–7. doi:10.1056/NEJM199511163332001. PMID 7566020.
- Grundy SM (May 2007). "Should women be offered cholesterol lowering drugs to prevent cardiovascular disease? Yes". BMJ. 334 (7601): 982. doi:10.1136/bmj.39202.399942.AD. PMC 1867899. PMID 17494017.
- Kendrick M (May 2007). "Should women be offered cholesterol lowering drugs to prevent cardiovascular disease? No". BMJ. 334 (7601): 983. doi:10.1136/bmj.39202.397488.AD. PMC 1867901. PMID 17494018.
- Brugts JJ, Yetgin T, Hoeks SE, Gotto AM, Shepherd J, Westendorp RG, et al. (June 2009). "The benefits of statins in people without established cardiovascular disease but with cardiovascular risk factors: meta-analysis of randomised controlled trials". BMJ. 338: b2376. doi:10.1136/bmj.b2376. PMC 2714690. PMID 19567909.
- "Report of the National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. The Expert Panel". Archives of Internal Medicine. 148 (1): 36–69. January 1988. doi:10.1001/archinte.148.1.36. PMID 3422148.
- "Cholesterol". American Heart Association. 30 April 2017. Retrieved 22 March 2019.
- Alenghat FJ, Davis AM (February 2019). "Management of Blood Cholesterol". JAMA. 321 (8): 800–801. doi:10.1001/jama.2019.0015. PMC 6679800. PMID 30715135.
- "About cholesterol". American Heart Association.
- Warnick GR, Knopp RH, Fitzpatrick V, Branson L (January 1990). "Estimating low-density lipoprotein cholesterol by the Friedewald equation is adequate for classifying patients on the basis of nationally recommended cutpoints". Clinical Chemistry. 36 (1): 15–9. doi:10.1093/clinchem/36.1.15. PMID 2297909.
- Anderson KM, Castelli WP, Levy D (April 1987). "Cholesterol and mortality. 30 years of follow-up from the Framingham study". JAMA. 257 (16): 2176–80. doi:10.1001/jama.257.16.2176. PMID 3560398.
- Ulmer H, Kelleher C, Diem G, Concin H (2004). "Why Eve is not Adam: prospective follow-up in 149650 women and men of cholesterol and other risk factors related to cardiovascular and all-cause mortality". Journal of Women's Health. 13 (1): 41–53. doi:10.1089/154099904322836447. PMID 15006277.
- Chowdhury R, Warnakula S, Kunutsor S, Crowe F, Ward HA, Johnson L, et al. (March 2014). "Association of dietary, circulating, and supplement fatty acids with coronary risk: a systematic review and meta-analysis". Annals of Internal Medicine. 160 (6): 398–406. doi:10.7326/M13-1788. PMID 24723079.
- Ravnskov U, Diamond DM, Hama R, Hamazaki T, Hammarskjöld B, Hynes N, et al. (June 2016). "Lack of an association or an inverse association between low-density-lipoprotein cholesterol and mortality in the elderly: a systematic review". BMJ Open. 6 (6): e010401. doi:10.1136/bmjopen-2015-010401. PMC 4908872. PMID 27292972.
- Rizos CV, Elisaf MS, Liberopoulos EN (24 February 2011). "Effects of thyroid dysfunction on lipid profile". The Open Cardiovascular Medicine Journal. 5 (1): 76–84. doi:10.2174/1874192401105010076. PMC 3109527. PMID 21660244.
- "How To Get Your Cholesterol Tested". American Heart Association. Retrieved 10 July 2013.
- Stone NJ, Robinson J, Goff DC (2013). "Getting a grasp of the Guidelines". American College of Cardiology. Archived from the original on 7 July 2014. Retrieved 2 April 2014.
- "Implications of Recent Clinical Trials for the ATP III Guidelines". National Heart, Lungs and Blood Institute. Archived from the original on 2 February 2014. Retrieved 2014-01-27.
- Westover EJ, Covey DF, Brockman HL, Brown RE, Pike LJ (December 2003). "Cholesterol depletion results in site-specific increases in epidermal growth factor receptor phosphorylation due to membrane level effects. Studies with cholesterol enantiomers". The Journal of Biological Chemistry. 278 (51): 51125–33. doi:10.1074/jbc.M304332200. PMC 2593805. PMID 14530278.
- Kristiana I, Luu W, Stevenson J, Cartland S, Jessup W, Belani JD, et al. (September 2012). "Cholesterol through the looking glass: ability of its enantiomer also to elicit homeostatic responses". The Journal of Biological Chemistry. 287 (40): 33897–904. doi:10.1074/jbc.M112.360537. PMC 3460484. PMID 22869373.
| Wikimedia Commons has media related to Cholesterol. |