Metrizamide
Metrizamide is a non-ionic iodine-based radiocontrast agent. A density gradient medium for the centrifugation of biological particles.
 | |
| Clinical data | |
|---|---|
| Trade names | Amipaque |
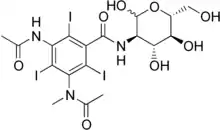
| Other names | 3-aAcetamido-2,4,6-triiodo-5-(N-methylacetamido)-N-[(3R,4R,5S,6R)-2,4,5-trihydroxy-6-(hydroxymethyl)oxan-3-yl]benzamide |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.045.872 |
| Chemical and physical data | |
| Formula | C18H22I3N3O8 |
| Molar mass | 789.100 g·mol−1 |
| | |
Historically metrizamide replaced iofendylate (trade names: Pantopaque, Myodil) as the contrast agent of choice for myelography (an X-ray study of the spine now largely replaced by MRI). The radio opacity characteristics are such that finer detail is displayed with metrizamide, as well as the advantage of reabsorption from spinal fluid and excretion from the body – since unlike Pantopaque, metrizamide is a water-soluble substance.
Both agents are administered by lumbar puncture (also referred to as a spinal tap or cisternal puncture), at the cervicocranial junction. The human patient is rolled from the lateral decubitus (lying on the side) to prone. Ankles are strapped to the end of a hard X-ray, CT, or MRI table. To obtain images of the cervical region the patient is then carefully tilted in the Trendelenberg position (head down) so the contrast agent (particularly Pantopaque) can enter the neck region. Care is given to prevent spilling dye into the posterior cranial fossa (back of the head) or to enter the cranial cavity in general. This speaks to the inability to remove the heavier or more viscous Pantopaque without aspirating it with spinal fluid (CSF) through a lumbar puncture needle in the low back (sticking up vertically) or back of the neck. Removal of Pantopauqe is necessary since it is non water-soluble. With metrizamide the issue is that if entering the cranial cavity and high dose exposure to the blood brain barrier, side effects are more likely encountered. Issues related to absorption into the general circulation are also encountered.
Side effects
- Headache
- Nausea
- Vomiting
- Mass effect, herniation, and major central nervous system complications including spinal cord compression
- Seizures
- Muscle rigidity and dystonic reaction
Further reading
- Ozdoba C, Gralla J, Rieke A, Binggeli R, Schroth G (2011). "Myelography in the Age of MRI: Why We Do It, and How We Do It". Radiology Research and Practice. 2011: 329017. doi:10.1155/2011/329017. PMC 3197073. PMID 22091378.
- Killebrew K, Whaley RA, Hayward JN, Scatliff JH (February 1983). "Complications of metrizamide myelography". Archives of Neurology. 40 (2): 78–80. doi:10.1001/archneur.1983.04050020040007. PMID 6824454.
- Boyd WR, Gardiner GA (September 1977). "Metrizamide myelography". AJR. American Journal of Roentgenology. 129 (3): 481–4. doi:10.2214/ajr.129.3.481. PMID 409203.
External links
- "Metrizamide". The Free Dictionary.
- "Metrizamide". Drug Bank.
- "Metrizamide". PubChem.
- "Metrizamide". Drugs.com.
