Molybdenum(III) bromide
Molybdenum(III) bromide is the inorganic compound with the formula MoBr3. It is a black solid that is insoluble in most solvents but dissolves in donor solvents such as pyridine.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Molybdenum(III) bromide | |
| Other names
Molybdenum tribromide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.033.259 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| MoBr3 | |
| Molar mass | 335.70 g/mol |
| Appearance | dark green to black solid |
| Density | 4.89 g/cm3 |
| Melting point | 500 °C (932 °F; 773 K) (decomposes) |
| insoluble | |
| Solubility | soluble in pyridine |
| +525.0·10−6 cm3/mol | |
| Related compounds | |
Other anions |
Molybdenum(III) chloride Molybdenum(III) iodide |
Related compounds |
Molybdenum(II) bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
Molybdenum(III) bromide is produced by the reaction of elemental molybdenum and bromine at 350 °C (662 °F).[1]
It can also be prepared from the reduction of molybdenum(IV) bromide with molybdenum metal, hydrogen gas, or a hydrocarbon.[2]
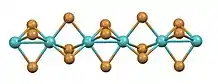
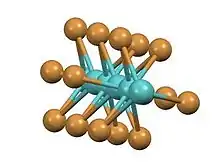
It has a structure consisting of infinite chains of face-sharing octahedra with alternatingly short and long Mo-Mo contacts. The same structure is adopted by the tribromides of ruthenium and technetium.[3][4] In contrast, in the high temperature phase of titanium(III) iodide, the Ti---Ti separation is invariant.
References
- F. Hein, S. Herzog "Molybdenum(III) Bromide" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1407.
- Perry, Dale L. (2011). Handbook of Inorganic Compounds (2nd ed.). Boca Raton: Taylor & Francis. p. 279. ISBN 978-1-4398-1461-1.
- Dietrich Babel: Die Verfeinerung der MoBr3-Struktur (Refinement of the MoBr3-Structure) In: Journal of Solid State Chemistry. 1972, volume 4, S. 410–416, doi:10.1016/0022-4596(72)90156-9.
- Order-Disorder Transformation in RuBr3 and MoBr3: A two-Dimensional Ising Model" Merlino, S.; Labella, L.; Marchetti, F.; Toscani, S. Chemistry of Materials 2004, volume 16, p3895-p3903
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.