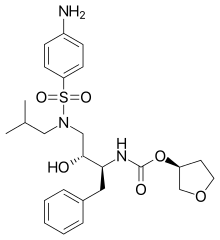
Amprenavir
Amprenavir (original brand name Agenerase, GlaxoSmithKline) is a protease inhibitor used to treat HIV infection. It was approved by the Food and Drug Administration on April 15, 1999, for twice-a-day dosing instead of needing to be taken every eight hours. The convenient dosing came at a price, as the dose required is 1,200 mg, delivered in 8 (eight) very large 150 mg gel capsules or 24 (twenty-four) 50 mg gel capsules twice daily.[1]
 | |
| Clinical data | |
|---|---|
| Trade names | Agenerase |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699051 |
| License data |
|
| Routes of administration | Oral (capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 90% |
| Metabolism | Hepatic |
| Elimination half-life | 7.1–10.6 hours |
| Excretion | <3% renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.262.589 |
| Chemical and physical data | |
| Formula | C25H35N3O6S |
| Molar mass | 505.63 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
It was patented in 1992 and approved for medical use in 1999.[2] Production of amprenavir was discontinued by the manufacturer on December 31, 2004; a prodrug version (fosamprenavir), is available.
Background
Research aimed at development of renin inhibitors as potential antihypertensive agents had led to the discovery of compounds that blocked the action of this peptide cleaving enzyme. The amino acid sequence cleaved by renin was found to be fortuitously the same as that required to produce the HIV peptide coat. Structure–activity studies on renin inhibitors proved to be of great value for developing HIV protease inhibitors. Incorporation of an amino alcohol moiety proved crucial to inhibitory activity for many of these agents. This unit is closely related to the one found in the statine, an unusual amino acid that forms part of the pepstatin, a fermentation product that inhibits protease enzymes.
References
- "Agenerase (amprenavir) Capsules. Full Prescribing Information. Section Dosage and Administration" (PDF). US Food and Drug Administration. GlaxoSmithKline and Vertex Pharmaceuticals Inc. Retrieved 29 November 2015.
- Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 509. ISBN 9783527607495.
- Shen CH, Wang YF, Kovalevsky AY, Harrison RW, Weber IT (September 2010). "Amprenavir complexes with HIV-1 protease and its drug-resistant mutants altering hydrophobic clusters". The FEBS Journal. 277 (18): 3699–714. doi:10.1111/j.1742-4658.2010.07771.x. PMC 2975871. PMID 20695887.
External links
- Amprenavir bound to proteins in the PDB