Cenicriviroc
Cenicriviroc (INN,[1] code names TAK-652, TBR-652) is an experimental drug candidate for the treatment of HIV infection[2] and in combination with Tropifexor for non-alcoholic steatohepatitis.[3] It is being developed by Takeda and Tobira Therapeutics.
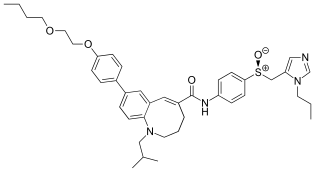 | |
| Names | |
|---|---|
| IUPAC name
(S,E)-8-(4-(2-Butoxyethoxy)phenyl)-1-isobutyl-N-(4-(((1-propyl-1H-imidazol-5-yl)methyl)sulfinyl)phenyl)-1,2,3,4-tetrahydrobenzo[b]azocine-5-carboxamide | |
| Other names
TAK-652; TBR-652 | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C41H52N4O4S | |
| Molar mass | 696.95 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Cenicriviroc is an inhibitor of CCR2 and CCR5 receptors,[4] allowing it to function as an entry inhibitor which prevents the virus from entering into a human cell. Inhibition of CCR2 may have an anti-inflammatory effect.
A double-blind, randomized, placebo-controlled clinical study to assess the antiviral activity, safety, and tolerability of cenicriviroc was conducted in 2010. HIV-infected patients taking cenicriviroc had significant reductions in viral load, with the effect persisting up to two weeks after discontinuation of treatment.[5] Additional Phase II clinical trials are underway.[6]
Phase IIb data presented at the 20th Conference on Retroviruses and Opportunistic Infections (CROI) in March 2013 showed similar viral suppression rates of 76% for patients taking 100 mg cenicriviroc, 73% with 200 mg cenicriviroc, and 71% with efavirenz. Non-response rates were higher with cenicriviroc, however, largely due to greater drop-out of patients. A new tablet formulation with lower pill burden may improve adherence. Looking at immune and inflammatory biomarkers, levels of MCP-1 increased and soluble CD14 decreased in the cenicriviroc arms.[7]
References
- "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 65" (PDF). World Health Organization. 2011. pp. 53–4. Retrieved 25 November 2016.
- Klibanov, OM; Williams, SH; Iler, CA (August 2010). "Cenicriviroc, an Orally Active CCR5 Antagonist for the Potential Treatment of HIV Infection". Current Opinion in Investigational Drugs. 11 (8): 940–50. PMID 20721836.
- https://clinicaltrials.gov/ct2/show/NCT03517540
- Baba, M; Takashima, K; Miyake, H; Kanzaki, N; Teshima, K; Wang, X; Shiraishi, M; Iizawa, Y (26 October 2005). "TAK-652 Inhibits CCR5-Mediated Human Immunodeficiency Virus Type 1 Infection In Vitro and Has Favorable Pharmacokinetics in Humans". Antimicrobial Agents and Chemotherapy. 49 (11): 4584–91. doi:10.1128/AAC.49.11.4584-4591.2005. PMC 1280155. PMID 16251299.
- Reviriego, C (July 2011). "Chemokine CCR2/CCR5 Receptor Antagonist Anti-HIV Agent". Drugs of the Future. 36 (7): 511–7. doi:10.1358/dof.2011.036.07.1622066.
- "Tobira Therapeutics Initiates Phase 2b Trial of Cenicriviroc". The Body. July 5, 2011.
- CROI 2013: CCR5/CCR2 Inhibitor Cenicriviroc Has Both Anti-HIV and Anti-inflammatory Effects. Highleyman, Liz. HIVandHepatitis.com. 7 March 2013.