Cervical cancer
Cervical cancer is a cancer arising from the cervix.[2] It is due to the abnormal growth of cells that have the ability to invade or spread to other parts of the body.[12] Early on, typically no symptoms are seen.[2] Later symptoms may include abnormal vaginal bleeding, pelvic pain or pain during sexual intercourse.[2] While bleeding after sex may not be serious, it may also indicate the presence of cervical cancer.[13]
| Cervical cancer | |
|---|---|
 | |
| Location of cervical cancer and an example of normal and abnormal cells | |
| Pronunciation | |
| Specialty | Oncology |
| Symptoms | Early: none[2] Later: vaginal bleeding, pelvic pain, pain during sexual intercourse[2] |
| Usual onset | Over 10 to 20 years[3] |
| Types | Squamous cell carcinoma, adenocarcinoma, others[4] |
| Causes | Human papillomavirus infection (HPV)[5][6] |
| Risk factors | Smoking, weak immune system, birth control pills, starting sex at a young age, many sexual partners or a partner with many sexual partners[2][4][7] |
| Diagnostic method | Cervical screening followed by a biopsy[2] |
| Prevention | Regular cervical screening, HPV vaccines, sexual intercourse with condoms[8][9], sexual abstinence |
| Treatment | Surgery, chemotherapy, radiation therapy, immunotherapy[2] |
| Prognosis | Five-year survival rate: 68% (US) 46% (India)[10] |
| Frequency | 570,000 new cases (2018)[11] |
| Deaths | 311,000 (2018)[11] |
Human papillomavirus infection (HPV) causes more than 90% of cases;[5][6] most people who have had HPV infections, however, do not develop cervical cancer.[3][14] Other risk factors include smoking, a weak immune system, birth control pills, starting sex at a young age, and having many sexual partners, but these are less important.[2][4] Cervical cancer typically develops from precancerous changes over 10 to 20 years.[3] About 90% of cervical cancer cases are squamous cell carcinomas, 10% are adenocarcinoma, and a small number are other types.[4] Diagnosis is typically by cervical screening followed by a biopsy.[2] Medical imaging is then done to determine whether or not the cancer has spread.[2]
HPV vaccines protect against two to seven high-risk strains of this family of viruses and may prevent up to 90% of cervical cancers.[9][15][16] As a risk of cancer still exists, guidelines recommend continuing regular Pap tests.[9] Other methods of prevention include having few or no sexual partners and the use of condoms.[8] Cervical cancer screening using the Pap test or acetic acid can identify precancerous changes, which when treated, can prevent the development of cancer.[17] Treatment may consist of some combination of surgery, chemotherapy, and radiation therapy.[2] Five-year survival rates in the United States are 68%.[18] Outcomes, however, depend very much on how early the cancer is detected.[4]
Worldwide, cervical cancer is both the fourth-most common cause of cancer and the fourth-most common cause of death from cancer in women.[3] In 2012, an estimated 528,000 cases of cervical cancer occurred, with 266,000 deaths.[3] This is about 8% of the total cases and total deaths from cancer.[19] About 70% of cervical cancers and 90% of deaths occur in developing countries.[3][20] In low-income countries, it is one of the most common causes of cancer death.[17] In developed countries, the widespread use of cervical screening programs has dramatically reduced rates of cervical cancer.[21] In medical research, the most famous immortalized cell line, known as HeLa, was developed from cervical cancer cells of a woman named Henrietta Lacks.[22]
Signs and symptoms
The early stages of cervical cancer may be completely free of symptoms.[5][21] Vaginal bleeding, contact bleeding (one most common form being bleeding after sexual intercourse), or (rarely) a vaginal mass may indicate the presence of malignancy. Also, moderate pain during sexual intercourse and vaginal discharge are symptoms of cervical cancer.[23] In advanced disease, metastases may be present in the abdomen, lungs, or elsewhere.[24]
Symptoms of advanced cervical cancer may include: loss of appetite, weight loss, fatigue, pelvic pain, back pain, leg pain, swollen legs, heavy vaginal bleeding, bone fractures, and (rarely) leakage of urine or feces from the vagina.[25] Bleeding after douching or after a pelvic exam is a common symptom of cervical cancer.[26]
Causes
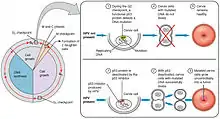
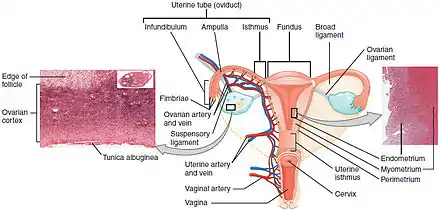
Infection with some types of HPV is the greatest risk factor for cervical cancer, followed by smoking.[27] HIV infection is also a risk factor.[27] Not all of the causes of cervical cancer are known, however, and several other contributing factors have been implicated.[28][29]
Human papillomavirus
HPV types 16 and 18 are the cause of 75% of cervical cancer cases globally, while 31 and 45 are the causes of another 10%.[30]
Women who have sex with men who have many other sexual partners or women who have many sexual partners have a greater risk.[31][32]
Of the 150-200 types of HPV known,[33][34] 15 are classified as high-risk types (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 68, 73, and 82), three as probable high-risk (26, 53, and 66), and 12 as low-risk (6, 11, 40, 42, 43, 44, 54, 61, 70, 72, 81, and CP6108).[35]
Genital warts, which are a form of benign tumor of epithelial cells, are also caused by various strains of HPV. However, these serotypes are usually not related to cervical cancer. Having multiple strains at the same time is common, including those that can cause cervical cancer along with those that cause warts. Infection with HPV is generally believed to be required for cervical cancer to occur.[36]
Smoking
Cigarette smoking, both active and passive, increases the risk of cervical cancer. Among HPV-infected women, current and former smokers have roughly two to three times the incidence of invasive cancer. Passive smoking is also associated with increased risk, but to a lesser extent.[37]
Smoking has also been linked to the development of cervical cancer.[38][39][27] Smoking can increase the risk in women a few different ways, which can be by direct and indirect methods of inducing cervical cancer.[38][27][40] A direct way of contracting this cancer is a smoker has a higher chance of cervical intraepithelial neoplasia (CIN3) occurring, which has the potential of forming cervical cancer.[38] When CIN3 lesions lead to cancer, most of them have the assistance of the HPV virus, but that is not always the case, which is why it can be considered a direct link to cervical cancer.[40] Heavy smoking and long-term smoking seem to have more of a risk of getting the CIN3 lesions than lighter smoking or not smoking at all.[41] Although smoking has been linked to cervical cancer, it aids in the development of HPV, which is the leading cause of this type of cancer.[27] Also, not only does it aid in the development of HPV, but also if the woman is already HPV-positive, she is at an even greater likelihood of contracting cervical cancer.[41]
Oral contraceptives
Long-term use of oral contraceptives is associated with increased risk of cervical cancer in women who have had HPV. Women who have used oral contraceptives for 5 to 9 years have about three times the incidence of invasive cancer, and those who used them for 10 years or longer have about four times the risk.[37]
Multiple pregnancies
Having many pregnancies is associated with an increased risk of cervical cancer. Among HPV-infected women, those who have had seven or more full-term pregnancies have around four times the risk of cancer compared with women with no pregnancies, and two to three times the risk of women who have had one or two full-term pregnancies.[37]
Diagnosis
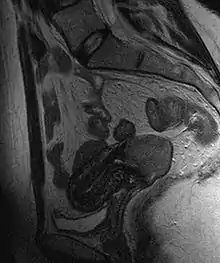
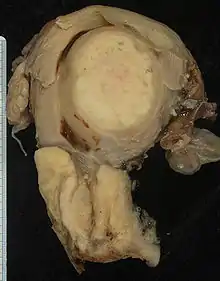
Biopsy
The Pap test can be used as a screening test, but produces a false negative in up to 50% of cases of cervical cancer.[42][43] Other concerns is the cost of doing Pap tests, which make them unaffordable in many areas of the world.[44]
Confirmation of the diagnosis of cervical cancer or precancer requires a biopsy of the cervix. This is often done through colposcopy, a magnified visual inspection of the cervix aided by using a dilute acetic acid (e.g. vinegar) solution to highlight abnormal cells on the surface of the cervix,[5] with visual contrast provided by staining the normal tissues a mahogany brown with Lugol's iodine.[45] Medical devices used for biopsy of the cervix include punch forceps. Colposcopic impression, the estimate of disease severity based on the visual inspection, forms part of the diagnosis. Further diagnostic and treatment procedures are loop electrical excision procedure and cervical conization, in which the inner lining of the cervix is removed to be examined pathologically. These are carried out if the biopsy confirms severe cervical intraepithelial neoplasia.
Often before the biopsy, the doctor asks for medical imaging to rule out other causes of woman's symptoms. Imaging modalities such as ultrasound, CT scan, and MRI have been used to look for alternating disease, spread of the tumor, and effect on adjacent structures. Typically, they appear as heterogeneous mass on the cervix.[46]
Interventions such as playing music during the procedure and viewing the procedure on a monitor can reduce the anxiety associated with the examination.[47]
Precancerous lesions
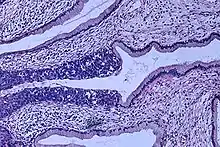
Cervical intraepithelial neoplasia, the potential precursor to cervical cancer, is often diagnosed on examination of cervical biopsies by a pathologist. For premalignant dysplastic changes, cervical intraepithelial neoplasia grading is used.
The naming and histologic classification of cervical carcinoma precursor lesions has changed many times over the 20th century. The World Health Organization classification[48][49] system was descriptive of the lesions, naming them mild, moderate, or severe dysplasia or carcinoma in situ (CIS). The term cervical intraepithelial neoplasia (CIN) was developed to place emphasis on the spectrum of abnormality in these lesions, and to help standardize treatment.[49] It classifies mild dysplasia as CIN1, moderate dysplasia as CIN2, and severe dysplasia and CIS as CIN3. More recently, CIN2 and CIN3 have been combined into CIN2/3. These results are what a pathologist might report from a biopsy.
These should not be confused with the Bethesda system terms for Pap test (cytopathology) results. Among the Bethesda results: Low-grade squamous intraepithelial lesion (LSIL) and high-grade squamous intraepithelial lesion (HSIL). An LSIL Pap may correspond to CIN1, and HSIL may correspond to CIN2 and CIN3,[49] but they are results of different tests, and the Pap test results need not match the histologic findings.
Cancer subtypes

Histologic subtypes of invasive cervical carcinoma include:[50][51] Though squamous cell carcinoma is the cervical cancer with the most incidence, the incidence of adenocarcinoma of the cervix has been increasing in recent decades.[5] Endocervical adenocarcinoma represents 20–25% of the histological types of cervical carcinoma. Gastric-type mucinous adenocarcinoma of the cervix is a rare type of cancer with aggressive behavior. This type of malignancy is not related to high-risk human papillomavirus (HPV).[52]
- Squamous cell carcinoma (about 80–85%[53][54])
- adenocarcinoma (about 15% of cervical cancers in the UK[48])
- Adenosquamous carcinoma
- Small cell carcinoma
- Neuroendocrine tumour
- Glassy cell carcinoma
- Villoglandular adenocarcinoma
Noncarcinoma malignancies which can rarely occur in the cervix include melanoma and lymphoma. The International Federation of Gynecology and Obstetrics (FIGO) stage does not incorporate lymph node involvement in contrast to the TNM staging for most other cancers. For cases treated surgically, information obtained from the pathologist can be used in assigning a separate pathologic stage, but is not to replace the original clinical stage.
Staging
Cervical cancer is staged by the FIGO system, which is based on clinical examination, rather than surgical findings. It allows only these diagnostic tests to be used in determining the stage: palpation, inspection, colposcopy, endocervical curettage, hysteroscopy, cystoscopy, proctoscopy, intravenous urography, and X-ray examination of the lungs and skeleton, and cervical conization.
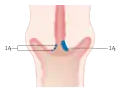 Stage 1A cervical cancer
Stage 1A cervical cancer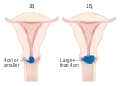 Stage 1B cervical cancer
Stage 1B cervical cancer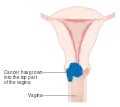 Stage 2A cervical cancer
Stage 2A cervical cancer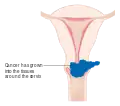 Stage 2B cervical cancer
Stage 2B cervical cancer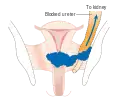 Stage 3B cervical cancer
Stage 3B cervical cancer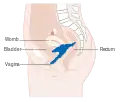 Stage 4A cervical cancer
Stage 4A cervical cancer Stage 4B cervical cancer
Stage 4B cervical cancer
Prevention
Screening

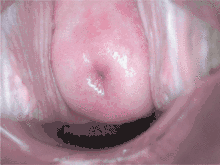

Checking cervical cells with the Papanicolaou test (Pap test) for cervical pre-cancer has dramatically reduced the number of cases of, and mortality from, cervical cancer.[21] Liquid-based cytology may reduced the number of inadequate samples.[55][56][57] Pap test screening every three to five years with appropriate follow-up can reduce cervical cancer incidence up to 80%.[58] Abnormal results may suggest the presence of precancerous changes, allowing examination and possible preventive treatment, known as colposcopy. The treatment of low-grade lesions may adversely affect subsequent fertility and pregnancy.[37] Personal invitations encouraging women to get screened are effective at increasing the likelihood they will do so. Educational materials also help increase the likelihood women will go for screening, but they are not as effective as invitations.[59]
According to the 2010 European guidelines, the age at which to start screening ranges between 20 and 30 years of age, but preferentially not before age 25 or 30 years, and depends on burden of the disease in the population and the available resources.[58]
In the United States, screening is recommended to begin at age 21, regardless of age at which a woman began having sex or other risk factors.[60] Pap tests should be done every three years between the ages of 21 and 65.[60] In women over the age of 65, screening may be discontinued if no abnormal screening results were seen within the previous 10 years and no history of CIN2 or higher exists.[60][61][62] HPV vaccination status does not change screening rates.[61]
A number of recommended options exist for screening those 30 to 65.[63] This includes cervical cytology every 3 years, HPV testing every 5 years, or HPV testing together with cytology every 5 years.[63][61] Screening is not beneficial before age 25, as the rate of disease is low. Screening is not beneficial in women older than 60 years if they have a history of negative results.[37] The American Society of Clinical Oncology guideline has recommend for different levels of resource availability.[64]
Pap tests have not been as effective in developing countries.[65] This is in part because many of these countries have an impoverished health care infrastructure, too few trained and skilled professionals to obtain and interpret Pap tests, uninformed women who get lost to follow-up, and a lengthy turn-around time to get results.[65] Visual inspection with acetic acid and HPV DNA testing have been tried, though with mixed success.[65]
Barrier protection
Barrier protection or spermicidal gel use during sexual intercourse decreases, but does not eliminate risk of transmitting the infection,[37] though condoms may protect against genital warts.[66] They also provide protection against other sexually transmitted infections, such as HIV and Chlamydia, which are associated with greater risks of developing cervical cancer.
Vaccination
Three HPV vaccines (Gardasil, Gardasil 9, and Cervarix) reduce the risk of cancerous or precancerous changes of the cervix and perineum by about 93% and 62%, respectively.[67] The vaccines are between 92% and 100% effective against HPV 16 and 18 up to at least 8 years.[37]
HPV vaccines are typically given to age 9 to 26, as the vaccine is most effective if given before infection occurs. The duration of effectiveness and whether a booster will be needed is unknown. The high cost of this vaccine has been a cause for concern. Several countries have considered (or are considering) programs to fund HPV vaccination. The American Society of Clinical Oncology guideline has recommendations for different levels of resource availability.[64]
Since 2010, young women in Japan have been eligible to receive the cervical cancer vaccination for free.[68] In June 2013, the Japanese Ministry of Health, Labor and Welfare mandated that, before administering the vaccine, medical institutions must inform women that the ministry does not recommend it.[68] However, the vaccine is still available at no cost to Japanese women who choose to accept the vaccination.[68]
Nutrition
Vitamin A is associated with a lower risk[69] as are vitamin B12, vitamin C, vitamin E, and beta-Carotene.[70]
Treatment
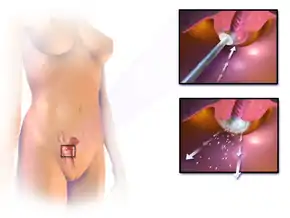
The treatment of cervical cancer varies worldwide, largely due to access to surgeons skilled in radical pelvic surgery, and the emergence of fertility-sparing therapy in developed nations. Less advanced stages of cervical cancer typically have treatment options that allow fertility to be maintained, if the patient desires.[71] Because cervical cancers are radiosensitive, radiation may be used in all stages where surgical options do not exist. Surgical intervention may have better outcomes than radiological approaches.[72] In addition, chemotherapy can be used to treat cervical cancer, and has been found to be more effective than radiation alone.[73] Evidence suggests chemoradiotherapy may increase overall survival and reduce the risk of disease recurrence compared to radiotherapy alone.[74] Peri-operative care approaches, such as 'fast-track surgery' or 'enhanced recovery programmes' may lower surgical stress and improve recovery after gynaecological cancer surgery.[75]
Microinvasive cancer (stage IA) may be treated by hysterectomy (removal of the whole uterus including part of the vagina).[76] For stage IA2, the lymph nodes are removed, as well. Alternatives include local surgical procedures such as a loop electrical excision procedure or cone biopsy.[77][78] A systematic review concluded that more evidence is needed to inform decisions about different surgical techniques for women with cervical cancer at stage IA2.[79]
If a cone biopsy does not produce clear margins[80] (findings on biopsy showing that the tumor is surrounded by cancer free tissue, suggesting all of the tumor is removed), one more possible treatment option for women who want to preserve their fertility is a trachelectomy.[81] This attempts to surgically remove the cancer while preserving the ovaries and uterus, providing for a more conservative operation than a hysterectomy. It is a viable option for those in stage I cervical cancer which has not spread; however, it is not yet considered a standard of care,[82] as few doctors are skilled in this procedure. Even the most experienced surgeon cannot promise that a trachelectomy can be performed until after surgical microscopic examination, as the extent of the spread of cancer is unknown. If the surgeon is not able to microscopically confirm clear margins of cervical tissue once the woman is under general anesthesia in the operating room, a hysterectomy may still be needed. This can only be done during the same operation if the woman has given prior consent. Due to the possible risk of cancer spread to the lymph nodes in stage 1B cancers and some stage 1A cancers, the surgeon may also need to remove some lymph nodes from around the uterus for pathologic evaluation.
A radical trachelectomy can be performed abdominally[83] or vaginally[84] and opinions are conflicting as to which is better.[85] A radical abdominal trachelectomy with lymphadenectomy usually only requires a two- to three-day hospital stay, and most women recover very quickly (about six weeks). Complications are uncommon, although women who are able to conceive after surgery are susceptible to preterm labor and possible late miscarriage.[86] A wait of at least one year is generally recommended before attempting to become pregnant after surgery.[87] Recurrence in the residual cervix is very rare if the cancer has been cleared with the trachelectomy.[82] Yet, women are recommended to practice vigilant prevention and follow-up care including Pap screenings/colposcopy, with biopsies of the remaining lower uterine segment as needed (every 3–4 months for at least 5 years) to monitor for any recurrence in addition to minimizing any new exposures to HPV through safe sex practices until one is actively trying to conceive.
Early stages (IB1 and IIA less than 4 cm) can be treated with radical hysterectomy with removal of the lymph nodes or radiation therapy. Radiation therapy is given as external beam radiotherapy to the pelvis and brachytherapy (internal radiation). Women treated with surgery who have high-risk features found on pathologic examination are given radiation therapy with or without chemotherapy to reduce the risk of relapse. A Cochrane review has found moderate-certainty evidence that radiation decreases the risk of disease progression in people with stage IB cervical cancer, when compared to no further treatment.[88] However, little evidence was found on its effects on overall survival.[88]

Larger early-stage tumors (IB2 and IIA more than 4 cm) may be treated with radiation therapy and cisplatin-based chemotherapy, hysterectomy (which then usually requires adjuvant radiation therapy), or cisplatin chemotherapy followed by hysterectomy. When cisplatin is present, it is thought to be the most active single agent in periodic diseases.[89] Such addition of platinum-based chemotherapy to chemoradiation seems not only to improve survival but also reduces risk of recurrence in women with early stage cervical cancer (IA2-IIA).[90] A Cochrane review found a lack of evidence on the benefits and harms of primary hysterectomy compared to primary chemoradiotherapy for cervical cancer in stage IB2.[91]
Advanced-stage tumors (IIB-IVA) are treated with radiation therapy and cisplatin-based chemotherapy. On June 15, 2006, the US Food and Drug Administration approved the use of a combination of two chemotherapy drugs, hycamtin and cisplatin, for women with late-stage (IVB) cervical cancer treatment.[92] Combination treatment has significant risk of neutropenia, anemia, and thrombocytopenia side effects.
There is insufficient evidence whether anticancer drugs after standard care help women with locally advanced cervical cancer to live longer.[93]
For surgery to be curative, the entire cancer must be removed with no cancer found at the margins of the removed tissue on examination under a microscope.[94] This procedure is known as exenteration.[94]
No evidence is available to suggest that any form of follow‐up approach is better or worse in terms of prolonging survival, improving quality of life or guiding the management of problems that can arise because of the treatment and that in the case of radiotherapy treatment worsen with time.[95] A 2019 review found no controlled trials regarding the efficacy and safety of interventions for vaginal bleeding in women with advanced cervical cancer.[96]
 Diagram showing the area removed with a posterior surgery
Diagram showing the area removed with a posterior surgery Diagram showing the area removed with a total operation
Diagram showing the area removed with a total operation Diagram showing the area removed with an anterior operation
Diagram showing the area removed with an anterior operation
Prognosis
Stage
Prognosis depends on the stage of the cancer. The chance of a survival rate is nearly 100% for women with microscopic forms of cervical cancer.[97] With treatment, the five-year relative survival rate for the earliest stage of invasive cervical cancer is 92%, and the overall (all stages combined) five-year survival rate is about 72%. These statistics may be improved when applied to women newly diagnosed, bearing in mind that these outcomes may be partly based on the state of treatment five years ago when the women studied were first diagnosed.[98]
With treatment, 80–90% of women with stage I cancer and 60–75% of those with stage II cancer are alive 5 years after diagnosis. Survival rates decrease to 30–40% for women with stage III cancer and 15% or fewer of those with stage IV cancer five years after diagnosis.[99] Recurrent cervical cancer detected at its earliest stages might be successfully treated with surgery, radiation, chemotherapy, or a combination of the three. About 35% of women with invasive cervical cancer have persistent or recurrent disease after treatment.[100]
By country
Five year survival in the United States for White women is 69% and for Black women is 57%.[101]
Regular screening has meant that precancerous changes and early-stage cervical cancers have been detected and treated early. Figures suggest that cervical screening is saving 5,000 lives each year in the UK by preventing cervical cancer.[102] About 1,000 women per year die of cervical cancer in the UK. All of the Nordic countries have cervical cancer-screening programs in place.[103] The Pap test was integrated into clinical practice in the Nordic countries in the 1960s.[103]
In Africa outcomes are often worse as diagnosis is frequently at a latter stage of disease.[104]
Epidemiology
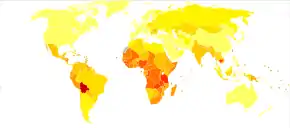
|
no data
<2.4
2.4-4.8
4.8-7.2
7.2-9.6
9.6-12
12-14.4
|
14.4-16.8
16.8-19.2
19.2-21.6
21.6–24
24–26.4
>26.4
|
Worldwide, cervical cancer is both the fourth-most common cause of cancer and deaths from cancer in women.[3] In 2018, 570,000 cases of cervical cancer were estimated to have occurred, with over 300,000 deaths.[106] It is the second-most common cause of female-specific cancer after breast cancer, accounting for around 8% of both total cancer cases and total cancer deaths in women.[19] About 80% of cervical cancers occur in developing countries.[107] It is the most frequently detected cancer during pregnancy, with an occurrence of 1.5 to 12 for every 100,000 pregnancies.[108]
Australia
Australia had 734 cases of cervical cancer in 2005. The number of women diagnosed with cervical cancer has dropped on average by 4.5% each year since organised screening began in 1991 (1991–2005).[109] Regular twice-yearly Pap tests can reduce the incidence of cervical cancer up to 90% in Australia, and save 1,200 Australian women from dying from the disease each year.[110] It is predicted that because of the success of the primary HPV testing programme there will be fewer than four new cases per 100 000 women annually by 2028.[111]
Canada
In Canada, an estimated 1,300 women will have been diagnosed with cervical cancer in 2008 and 380 will have died.[112]
India
In India, the number of people with cervical cancer is rising, but overall the age-adjusted rates are decreasing.[113] Usage of condoms in the female population has improved the survival of women with cancers of the cervix.[114]
European Union
In the European Union, about 34,000 new cases per year and over 16,000 deaths due to cervical cancer occurred in 2004.[58]
United Kingdom
Cervical cancer is the 12th-most common cancer in women in the UK (around 3,100 women were diagnosed with the disease in 2011), and accounts for 1% of cancer deaths (around 920 died in 2012).[115] With a 42% reduction from 1988 to 1997, the NHS-implemented screening programme has been highly successful, screening the highest-risk age group (25–49 years) every 3 years, and those ages 50–64 every 5 years.
United States
An estimated 13,170 new cervical cancers and 4,250 cervical cancer deaths will occur in the United States in 2019.[116] The median age at diagnosis is 50. The rates of new cases in the United States was 7.3 per 100,000 women, based on rates from 2012 to 2016. Cervical cancer deaths decreased by approximately 74% in the last 50 years, largely due to widespread Pap test screening.[117] The annual direct medical cost of cervical cancer prevention and treatment prior to introduction of the HPV vaccine was estimated at $6 billion.[117]
History
- 400 BCE — Hippocrates noted that cervical cancer was incurable.
- 1925 — Hinselmann invented the colposcope.
- 1928 — Papanicolaou developed the Papanicolaou technique.
- 1941 — Papanicolaou and Traut: Pap test screening began.
- 1946 — Aylesbury spatula was developed to scrape the cervix, collecting the sample for the Pap test.
- 1951 — First successful in-vitro cell line, HeLa, derived from biopsy of cervical cancer of Henrietta Lacks.
- 1976 — Harald zur Hausen and Gisam found HPV DNA in cervical cancer and genital warts; Hausen later won the Nobel Prize for his work.[118]
- 1988 — Bethesda System for reporting Pap results was developed.
- 2006 — First HPV vaccine was approved by the FDA.
- 2015 — HPV Vaccine shown to protect against infection at multiple body sites.[119]
- 2018 — Evidence for single-dose protection with HPV vaccine.[120]
Epidemiologists working in the early 20th century noted that cervical cancer behaved like a sexually transmitted disease. In summary:
- Cervical cancer was noted to be common in female sex workers.
- It was rare in nuns, except for those who had been sexually active before entering the convent (Rigoni in 1841).
- It was more common in the second wives of men whose first wives had died from cervical cancer.
- It was rare in Jewish women.[121]
- In 1935, Syverton and Berry discovered a relationship between RPV (Rabbit Papillomavirus) and skin cancer in rabbits (HPV is species-specific and therefore cannot be transmitted to rabbits).
These historical observations suggested that cervical cancer could be caused by a sexually transmitted agent. Initial research in the 1940s and 1950s attributed cervical cancer to smegma (e.g. Heins et al. 1958).[122] During the 1960s and 1970s it was suspected that infection with herpes simplex virus was the cause of the disease. In summary, HSV was seen as a likely cause because it is known to survive in the female reproductive tract, to be transmitted sexually in a way compatible with known risk factors, such as promiscuity and low socioeconomic status.[123] Herpes viruses were also implicated in other malignant diseases, including Burkitt's lymphoma, Nasopharyngeal carcinoma, Marek's disease and the Lucké renal adenocarcinoma. HSV was recovered from cervical tumour cells.
A description of human papillomavirus (HPV) by electron microscopy was given in 1949, and HPV-DNA was identified in 1963.[124] It was not until the 1980s that HPV was identified in cervical cancer tissue.[125] It has since been demonstrated that HPV is implicated in virtually all cervical cancers.[126] Specific viral subtypes implicated are HPV 16, 18, 31, 45 and others.
In work that was initiated in the mid 1980s, the HPV vaccine was developed, in parallel, by researchers at Georgetown University Medical Center, the University of Rochester, the University of Queensland in Australia, and the U.S. National Cancer Institute.[127] In 2006, the U.S. Food and Drug Administration (FDA) approved the first preventive HPV vaccine, marketed by Merck & Co. under the trade name Gardasil.
In November 2020, the World Health Organization, under backing from the World Health Assembly, set out a strategy to eliminate cervical cancer by 2050. The strategy involves vaccinating 90% of girls by the age of 15, screening 70% of women by the age of 35 and again by the age of 45, and treating 90% of women identified with cervical disease.[128]
Society and culture
Australia
In Australia, Aboriginal women are more than five times more likely to die from cervical cancer than non-Aboriginal women, suggesting that Aboriginal women are less likely to have regular Pap tests.[129] There are several factors that may limit indigenous women from engaging in regular cervical screening practices, including sensitivity in discussing the topic in Aboriginal communities, embarrassment, anxiety and fear about the procedure.[130] Difficulty in accessing screening services (for example, transport difficulties) and a lack of female GPs, trained Pap test providers and trained female Aboriginal Health Workers are also issues.[130]
The Australian Cervical Cancer Foundation (ACCF), founded in 2008, promotes 'women's health by eliminating cervical cancer and enabling treatment for women with cervical cancer and related health issues, in Australia and in developing countries.'[131] Ian Frazer, one of the developers of the Gardasil cervical cancer vaccine, is the scientific advisor to ACCF.[132] Janette Howard, the wife of the then-Prime Minister of Australia, John Howard, was diagnosed with cervical cancer in 1996, and first spoke publicly about the disease in 2006.[133]

United States
A 2007 survey of American women found 40% had heard of HPV infection and less than half of those knew it causes cervical cancer.[134] Over a longitudinal study from 1975 to 2000, it was found that people of lower socioeconomic census brackets had higher rates of late-stage cancer diagnosis and higher morbidity rates. After controlling for stage, there still existed differences in survival rates.[135]
References
- "CERVICAL | meaning in the Cambridge English Dictionary". dictionary.cambridge.org. Retrieved 5 October 2019.
- "Cervical Cancer Treatment (PDQ®)". NCI. 2014-03-14. Archived from the original on 5 July 2014. Retrieved 24 June 2014.
- World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 5.12. ISBN 978-9283204299.
- "Cervical Cancer Treatment (PDQ®)". National Cancer Institute. 2014-03-14. Archived from the original on 5 July 2014. Retrieved 25 June 2014.
- Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology (8th ed.). Saunders Elsevier. pp. 718–721. ISBN 978-1-4160-2973-1.
- Kufe, Donald (2009). Holland-Frei cancer medicine (8th ed.). New York: McGraw-Hill Medical. p. 1299. ISBN 9781607950141. Archived from the original on 2015-12-01.
- Bosch FX, de Sanjosé S (2007). "The epidemiology of human papillomavirus infection and cervical cancer". Disease Markers. 23 (4): 213–27. doi:10.1155/2007/914823. PMC 3850867. PMID 17627057.
- "Cervical Cancer Prevention (PDQ®)". National Cancer Institute. 2014-02-27. Archived from the original on 6 July 2014. Retrieved 25 June 2014.
- "Human Papillomavirus (HPV) Vaccines". National Cancer Institute. 2011-12-29. Archived from the original on 4 July 2014. Retrieved 25 June 2014.
- "Global Cancer Facts & Figures 3rd Edition" (PDF). 2015. p. 9. Archived (PDF) from the original on 2017-08-22. Retrieved 29 August 2017.
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (November 2018). "Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries". Ca. 68 (6): 394–424. doi:10.3322/caac.21492. PMID 30207593. S2CID 52188256.
- "Defining Cancer". National Cancer Institute. 2007-09-17. Archived from the original on 25 June 2014. Retrieved 10 June 2014.
- Tarney CM, Han J (2014). "Postcoital bleeding: a review on etiology, diagnosis, and management". Obstetrics and Gynecology International. 2014: 192087. doi:10.1155/2014/192087. PMC 4086375. PMID 25045355.
- Dunne EF, Park IU (December 2013). "HPV and HPV-associated diseases". Infectious Disease Clinics of North America. 27 (4): 765–78. doi:10.1016/j.idc.2013.09.001. PMID 24275269.
- "FDA approves Gardasil 9 for prevention of certain cancers caused by five additional types of HPV". U.S. Food and Drug Administration. 10 December 2014. Archived from the original on 10 January 2015. Retrieved 8 March 2015.
- Tran NP, Hung CF, Roden R, Wu TC (2014). Control of HPV infection and related cancer through vaccination. Recent Results in Cancer Research. 193. pp. 149–71. doi:10.1007/978-3-642-38965-8_9. ISBN 978-3-642-38964-1. PMID 24008298.
- World Health Organization (February 2014). "Fact sheet No. 297: Cancer". Archived from the original on 2014-02-13. Retrieved 2014-06-24.
- "SEER Stat Fact Sheets: Cervix Uteri Cancer". NCI. National Cancer Institute. November 10, 2014. Archived from the original on 6 July 2014. Retrieved 18 June 2014.
- World Cancer Report 2014. World Health Organization. 2014. pp. Chapter 1.1. ISBN 978-9283204299.
- "Cervical cancer prevention and control saves lives in the Republic of Korea". World Health Organization. Retrieved 1 November 2018.
- Canavan TP, Doshi NR (March 2000). "Cervical cancer". American Family Physician. 61 (5): 1369–76. PMID 10735343. Archived from the original on 2005-02-06.
- Jr, Charles E. Carraher (2014). Carraher's polymer chemistry (Ninth ed.). Boca Raton: Taylor & Francis. p. 385. ISBN 9781466552036. Archived from the original on 2015-10-22.
- "Cervical Cancer Symptoms, Signs, Causes, Stages & Treatment". medicinenet.com.
- Cheng X, Li H, Wu X (July 2016). "Advances in diagnosis and treatment of metastatic cervical cancer". Journal of Gynecologic Oncology. 27 (4): e43. doi:10.3802/jgo.2016.27.e43. PMC 4864519. PMID 27171673.
- Nanda, Rita (2006-06-09). "Cervical cancer". MedlinePlus Medical Encyclopedia. National Institutes of Health. Archived from the original on 2007-10-11. Retrieved 2007-12-02.
- "Cervical Cancer Prevention and Early Detection". Cancer. Archived from the original on 2015-07-10.
- Gadducci A, Barsotti C, Cosio S, Domenici L, Riccardo Genazzani A (August 2011). "Smoking habit, immune suppression, oral contraceptive use, and hormone replacement therapy use and cervical carcinogenesis: a review of the literature". Gynecological Endocrinology. 27 (8): 597–604. doi:10.3109/09513590.2011.558953. PMID 21438669. S2CID 25447563.
- Stuart Campbell; Ash Monga (2006). Gynaecology by Ten Teachers (18 ed.). Hodder Education. ISBN 978-0-340-81662-2.
- "Cervical Cancer Symptoms, Signs, Causes, Stages & Treatment". medicinenet.com.
- Dillman, Robert K.; Oldham, Robert O., eds. (2009). Principles of cancer biotherapy (5th ed.). Dordrecht: Springer. p. 149. ISBN 9789048122899. Archived from the original on 2015-10-29.
- "What Causes Cancer of the Cervix?". American Cancer Society. 2006-11-30. Archived from the original on 2007-10-13. Retrieved 2007-12-02.
- Marrazzo JM, Koutsky LA, Kiviat NB, Kuypers JM, Stine K (June 2001). "Papanicolaou test screening and prevalence of genital human papillomavirus among women who have sex with women". American Journal of Public Health. 91 (6): 947–52. doi:10.2105/AJPH.91.6.947. PMC 1446473. PMID 11392939.
- "HPV Type-Detect". Medical Diagnostic Laboratories. 2007-10-30. Archived from the original on 2007-09-27. Retrieved 2007-12-02.
- Gottlieb, Nicole (2002-04-24). "A Primer on HPV". Benchmarks. National Cancer Institute. Archived from the original on 2007-10-26. Retrieved 2007-12-02.
- Muñoz N, Bosch FX, de Sanjosé S, Herrero R, Castellsagué X, Shah KV, et al. (February 2003). "Epidemiologic classification of human papillomavirus types associated with cervical cancer" (PDF). The New England Journal of Medicine. 348 (6): 518–27. doi:10.1056/NEJMoa021641. hdl:2445/122831. PMID 12571259.
- Snijders PJ, Steenbergen RD, Heideman DA, Meijer CJ (January 2006). "HPV-mediated cervical carcinogenesis: concepts and clinical implications". The Journal of Pathology. 208 (2): 152–64. doi:10.1002/path.1866. PMID 16362994. S2CID 25400770.
- National Institutes of Health, National Cancer Institute: PDQ® Cervical Cancer Prevention Bethesda, MD: National Cancer Institute. Date last modified 05/01/2010. Accessed 06/04/2019.
- Luhn P, Walker J, Schiffman M, Zuna RE, Dunn ST, Gold MA, et al. (February 2013). "The role of co-factors in the progression from human papillomavirus infection to cervical cancer". Gynecologic Oncology. 128 (2): 265–70. doi:10.1016/j.ygyno.2012.11.003. PMC 4627848. PMID 23146688.
- Remschmidt C, Kaufmann AM, Hagemann I, Vartazarova E, Wichmann O, Deleré Y (March 2013). "Risk factors for cervical human papillomavirus infection and high-grade intraepithelial lesion in women aged 20 to 31 years in Germany". International Journal of Gynecological Cancer. 23 (3): 519–26. doi:10.1097/IGC.0b013e318285a4b2. PMID 23360813. S2CID 205679729.
- Agorastos T, Miliaras D, Lambropoulos AF, Chrisafi S, Kotsis A, Manthos A, Bontis J (July 2005). "Detection and typing of human papillomavirus DNA in uterine cervices with coexistent grade I and grade III intraepithelial neoplasia: biologic progression or independent lesions?". European Journal of Obstetrics, Gynecology, and Reproductive Biology. 121 (1): 99–103. doi:10.1016/j.ejogrb.2004.11.024. PMID 15949888.
- Jensen KE, Schmiedel S, Frederiksen K, Norrild B, Iftner T, Kjær SK (November 2012). "Risk for cervical intraepithelial neoplasia grade 3 or worse in relation to smoking among women with persistent human papillomavirus infection". Cancer Epidemiology, Biomarkers & Prevention. 21 (11): 1949–55. doi:10.1158/1055-9965.EPI-12-0663. PMC 3970163. PMID 23019238.
- Cecil Medicine: Expert Consult Premium Edition . ISBN 1437736084, 9781437736083. Page 1317.
- Berek and Hacker's Gynecologic Oncology. ISBN 0781795125, 9780781795128. Page 342
- Cronjé HS (February 2004). "Screening for cervical cancer in developing countries". International Journal of Gynaecology and Obstetrics. 84 (2): 101–8. doi:10.1016/j.ijgo.2003.09.009. PMID 14871510. S2CID 21356776.
- Sellors, JW (2003). "Chapter 4: An introduction to colposcopy: indications for colposcopy, instrumentation, principles and documentation of results". Colposcopy and treatment of cervical intraepithelial neoplasia: a beginners' manual. ISBN 978-92-832-0412-1.
- Pannu HK, Corl FM, Fishman EK (September–October 2001). "CT evaluation of cervical cancer: spectrum of disease". Radiographics. 21 (5): 1155–68. doi:10.1148/radiographics.21.5.g01se311155. PMID 11553823.
- Galaal K, Bryant A, Deane KH, Al-Khaduri M, Lopes AD (December 2011). "Interventions for reducing anxiety in women undergoing colposcopy". The Cochrane Database of Systematic Reviews (12): CD006013. doi:10.1002/14651858.cd006013.pub3. PMC 4161490. PMID 22161395.
- "Cancer Research UK website". Archived from the original on 2009-01-16. Retrieved 2009-01-03.
- DeMay, M (2007). Practical principles of cytopathology. Revised edition. Chicago, IL: American Society for Clinical Pathology Press. ISBN 978-0-89189-549-7.
- Garcia A, Hamid O, El-Khoueiry A (2006-07-06). "Cervical Cancer". eMedicine. WebMD. Archived from the original on 2007-12-09. Retrieved 2007-12-02.
- Dolinsky, Christopher (2006-07-17). "Cervical Cancer: The Basics". OncoLink. Abramson Cancer Center of the University of Pennsylvania. Archived from the original on 2008-01-18. Retrieved 2007-12-02.
- Mulita, Francesk; Iliopoulos, Fotios; Kehagias, Ioannis (2020). "A rare case of gastric-type mucinous endocervical adenocarcinoma in a 59-year-old woman". Menopausal Review. 19 (3): 147–150. doi:10.5114/pm.2020.99563. ISSN 1643-8876. PMC 7573334. PMID 33100952.
- "What Is Cervical Cancer?". American Cancer Society.
- "Cervical cancer – Types and grades". Cancer Research UK.
- Payne N, Chilcott J, McGoogan E (2000). "Liquid-based cytology in cervical screening: a rapid and systematic review". Health Technology Assessment. 4 (18): 1–73. doi:10.3310/hta4180. PMID 10932023. Archived from the original on 2011-06-11.
- Karnon J, Peters J, Platt J, Chilcott J, McGoogan E, Brewer N (May 2004). "Liquid-based cytology in cervical screening: an updated rapid and systematic review and economic analysis". Health Technology Assessment. 8 (20): iii, 1–78. doi:10.3310/hta8200. PMID 15147611.
- "Liquid Based Cytology (LBC): NHS Cervical Screening Programme". Archived from the original on 2011-01-08. Retrieved 2010-10-01.
- Arbyn M, Anttila A, Jordan J, Ronco G, Schenck U, Segnan N, et al. (March 2010). "European Guidelines for Quality Assurance in Cervical Cancer Screening. Second edition—summary document". Annals of Oncology. 21 (3): 448–58. doi:10.1093/annonc/mdp471. PMC 2826099. PMID 20176693.
- Everett T, Bryant A, Griffin MF, Martin-Hirsch PP, Forbes CA, Jepson RG (May 2011). Everett T (ed.). "Interventions targeted at women to encourage the uptake of cervical screening". The Cochrane Database of Systematic Reviews (5): CD002834. doi:10.1002/14651858.CD002834.pub2. PMC 4163962. PMID 21563135.
- "Cervical Cancer Screening Guidelines for Average-Risk Women" (PDF). cdc.gov. Archived (PDF) from the original on 1 February 2015. Retrieved 8 November 2014.
- Committee on Practice Bulletins—Gynecology (November 2012). "ACOG Practice Bulletin Number 131: Screening for cervical cancer". Obstetrics and Gynecology. 120 (5): 1222–38. doi:10.1097/AOG.0b013e318277c92a. PMID 23090560.
- Karjane N, Chelmow D (June 2013). "New cervical cancer screening guidelines, again". Obstetrics and Gynecology Clinics of North America. 40 (2): 211–23. doi:10.1016/j.ogc.2013.03.001. PMID 23732026.
- Curry SJ, Krist AH, Owens DK, Barry MJ, Caughey AB, Davidson KW, et al. (August 2018). "Screening for Cervical Cancer: US Preventive Services Task Force Recommendation Statement". JAMA. 320 (7): 674–686. doi:10.1001/jama.2018.10897. PMID 30140884.
- Arrossi S, Temin S, Garland S, Eckert LO, Bhatla N, Castellsagué X, et al. (October 2017). "Primary Prevention of Cervical Cancer: American Society of Clinical Oncology Resource-Stratified Guideline". Journal of Global Oncology. 3 (5): 611–634. doi:10.1200/JGO.2016.008151. PMC 5646902. PMID 29094100.
- World Health Organization (2014). Comprehensive cervical cancer control. A guide to essential practice - Second edition. ISBN 978-92-4-154895-3. Archived from the original on 2015-05-04.
- Manhart LE, Koutsky LA (November 2002). "Do condoms prevent genital HPV infection, external genital warts, or cervical neoplasia? A meta-analysis". Sexually Transmitted Diseases. 29 (11): 725–35. doi:10.1097/00007435-200211000-00018. PMID 12438912. S2CID 9869956.
- Medeiros LR, Rosa DD, da Rosa MI, Bozzetti MC, Zanini RR (October 2009). "Efficacy of human papillomavirus vaccines: a systematic quantitative review". International Journal of Gynecological Cancer. 19 (7): 1166–76. doi:10.1111/IGC.0b013e3181a3d100. PMID 19823051. S2CID 24695684.
- The Asahi Shimbun (15 June 2013). "Health ministry withdraws recommendation for cervical cancer vaccine". The Asahi Shimbun. Archived from the original on 19 June 2013.
- Zhang X, Dai B, Zhang B, Wang Z (February 2012). "Vitamin A and risk of cervical cancer: a meta-analysis". Gynecologic Oncology. 124 (2): 366–73. doi:10.1016/j.ygyno.2011.10.012. PMID 22005522.
- Myung SK, Ju W, Kim SC, Kim H (October 2011). "Vitamin or antioxidant intake (or serum level) and risk of cervical neoplasm: a meta-analysis". BJOG. 118 (11): 1285–91. doi:10.1111/j.1471-0528.2011.03032.x. hdl:2027.42/86903. PMID 21749626. S2CID 38761694.
- "Cervical Cancer Treatment Options | Treatment Choices by Stage". www.cancer.org. Retrieved 2020-09-12.
- Baalbergen A, Veenstra Y, Stalpers L (January 2013). "Primary surgery versus primary radiotherapy with or without chemotherapy for early adenocarcinoma of the uterine cervix". The Cochrane Database of Systematic Reviews (1): CD006248. doi:10.1002/14651858.CD006248.pub3. PMC 7387233. PMID 23440805.
- Einhorn N, Tropé C, Ridderheim M, Boman K, Sorbe B, Cavallin-Ståhl E (2003). "A systematic overview of radiation therapy effects in cervical cancer (cervix uteri)". Acta Oncologica. 42 (5–6): 546–56. doi:10.1080/02841860310014660. PMID 14596512.
- Chemoradiotherapy for Cervical Cancer Meta-analysis Collaboration (CCCMAC) (January 2010). "Reducing uncertainties about the effects of chemoradiotherapy for cervical cancer: individual patient data meta-analysis". The Cochrane Database of Systematic Reviews (1): CD008285. doi:10.1002/14651858.cd008285. PMC 7105912. PMID 20091664.
- Lu D, Wang X, Shi G (March 2015). "Perioperative enhanced recovery programmes for gynaecological cancer patients". The Cochrane Database of Systematic Reviews (3): CD008239. doi:10.1002/14651858.cd008239.pub4. PMC 6457837. PMID 25789452.
- van Nagell, J.R.; Greenwell, N.; Powell, D.F.; Donaldson, E.S.; Hanson, M.B.; Gay, E.C. (April 1983). "Microinvasive carcinoma of the cervix". American Journal of Obstetrics and Gynecology. 145 (8): 981–989. doi:10.1016/0002-9378(83)90852-9. ISSN 0002-9378. PMID 6837683.
- Erstad, Shannon (2007-01-12). "Cone biopsy (conization) for abnormal cervical cell changes". WebMD. Archived from the original on 2007-11-19. Retrieved 2007-12-02.
- Lin Y, Zhou J, Dai L, Cheng Y, Wang J (September 2017). "Vaginectomy and vaginoplasty for isolated vaginal recurrence 8 years after cervical cancer radical hysterectomy: A case report and literature review". The Journal of Obstetrics and Gynaecology Research. 43 (9): 1493–1497. doi:10.1111/jog.13375. PMID 28691384. S2CID 42161609.
- Kokka F, Bryant A, Brockbank E, Jeyarajah A (May 2014). "Surgical treatment of stage IA2 cervical cancer". The Cochrane Database of Systematic Reviews (5): CD010870. doi:10.1002/14651858.cd010870.pub2. PMC 6513277. PMID 24874726.
- Jones WB, Mercer GO, Lewis JL, Rubin SC, Hoskins WJ (October 1993). "Early invasive carcinoma of the cervix". Gynecologic Oncology. 51 (1): 26–32. doi:10.1006/gyno.1993.1241. PMID 8244170.
- Dolson, Laura (2001). "Trachelectomy". Archived from the original on 2007-09-27. Retrieved 2007-12-02.
- Burnett AF (February 2006). "Radical trachelectomy with laparoscopic lymphadenectomy: review of oncologic and obstetrical outcomes". Current Opinion in Obstetrics & Gynecology. 18 (1): 8–13. doi:10.1097/01.gco.0000192968.75190.dc. PMID 16493253. S2CID 22958941.
- Cibula D, Ungár L, Svárovský J, Zivný J, Freitag P (March 2005). "[Abdominal radical trachelectomy—technique and experience]". Ceska Gynekologie (in Czech). 70 (2): 117–22. PMID 15918265.
- Plante M, Renaud MC, Hoskins IA, Roy M (July 2005). "Vaginal radical trachelectomy: a valuable fertility-preserving option in the management of early-stage cervical cancer. A series of 50 pregnancies and review of the literature". Gynecologic Oncology. 98 (1): 3–10. doi:10.1016/j.ygyno.2005.04.014. PMID 15936061.
- Roy M, Plante M, Renaud MC, Têtu B (September 1996). "Vaginal radical hysterectomy versus abdominal radical hysterectomy in the treatment of early-stage cervical cancer". Gynecologic Oncology. 62 (3): 336–9. doi:10.1006/gyno.1996.0245. PMID 8812529.
- Dargent D, Martin X, Sacchetoni A, Mathevet P (April 2000). "Laparoscopic vaginal radical trachelectomy: a treatment to preserve the fertility of cervical carcinoma patients". Cancer. 88 (8): 1877–82. doi:10.1002/(SICI)1097-0142(20000415)88:8<1877::AID-CNCR17>3.0.CO;2-W. PMID 10760765.
- Schlaerth JB, Spirtos NM, Schlaerth AC (January 2003). "Radical trachelectomy and pelvic lymphadenectomy with uterine preservation in the treatment of cervical cancer". American Journal of Obstetrics and Gynecology. 188 (1): 29–34. doi:10.1067/mob.2003.124. PMID 12548192.
- Rogers L, Siu SS, Luesley D, Bryant A, Dickinson HO (May 2012). "Radiotherapy and chemoradiation after surgery for early cervical cancer". The Cochrane Database of Systematic Reviews (5): CD007583. doi:10.1002/14651858.cd007583.pub3. PMC 4171000. PMID 22592722.
- Waggoner SE (June 2003). "Cervical cancer". Lancet. 361 (9376): 2217–25. doi:10.1016/S0140-6736(03)13778-6. PMID 12842378. S2CID 24115541.
- Falcetta FS, Medeiros LR, Edelweiss MI, Pohlmann PR, Stein AT, Rosa DD (November 2016). "Adjuvant platinum-based chemotherapy for early stage cervical cancer". The Cochrane Database of Systematic Reviews. 11: CD005342. doi:10.1002/14651858.CD005342.pub4. PMC 4164460. PMID 27873308.
- Nama V, Angelopoulos G, Twigg J, Murdoch JB, Bailey J, Lawrie TA (October 2018). "Type II or type III radical hysterectomy compared to chemoradiotherapy as a primary intervention for stage IB2 cervical cancer". The Cochrane Database of Systematic Reviews. 10: CD011478. doi:10.1002/14651858.cd011478.pub2. PMC 6516889. PMID 30311942.
- "FDA Approves First Drug Treatment for Late-Stage Cervical Cancer". U.S. Food and Drug Administration. 2006-06-15. Archived from the original on 2007-10-10. Retrieved 2007-12-02.
- Tangjitgamol S, Katanyoo K, Laopaiboon M, Lumbiganon P, Manusirivithaya S, Supawattanabodee B (December 2014). "Adjuvant chemotherapy after concurrent chemoradiation for locally advanced cervical cancer". The Cochrane Database of Systematic Reviews (12): CD010401. doi:10.1002/14651858.cd010401.pub2. PMC 6402532. PMID 25470408.
- Sardain H, Lavoue V, Redpath M, Bertheuil N, Foucher F, Levêque J (August 2015). "Curative pelvic exenteration for recurrent cervical carcinoma in the era of concurrent chemotherapy and radiation therapy. A systematic review" (PDF). European Journal of Surgical Oncology. 41 (8): 975–85. doi:10.1016/j.ejso.2015.03.235. PMID 25922209.
- Lanceley A, Fiander A, McCormack M, Bryant A (November 2013). Cochrane Gynaecological, Neuro-oncology and Orphan Cancer Group (ed.). "Follow-up protocols for women with cervical cancer after primary treatment". The Cochrane Database of Systematic Reviews (11): CD008767. doi:10.1002/14651858.CD008767.pub2. PMID 24277645.
- Eleje GU, Eke AC, Igberase GO, Igwegbe AO, Eleje LI (March 2019). "Palliative interventions for controlling vaginal bleeding in advanced cervical cancer". The Cochrane Database of Systematic Reviews. 3: CD011000. doi:10.1002/14651858.cd011000.pub3. PMC 6423555. PMID 30888060.
- "Cervical Cancer". Encyclopedia of Women's Health.
- "What Are the Key Statistics About Cervical Cancer?". American Cancer Society. 2006-08-04. Archived from the original on 2007-10-30. Retrieved 2007-12-02.
- "Cervical Cancer". Cervical Cancer: Cancers of the Female Reproductive System: Merck Manual Home Edition. Merck Manual Home Edition. Archived from the original on 2006-12-16. Retrieved 2007-03-24.
- "Cervical Cancer". Cervical Cancer: Pathology, Symptoms and Signs, Diagnosis, Prognosis and Treatment. Armenian Health Network, Health.am. Archived from the original on 2007-02-07.
- "Cervical Cancer: Statistics | Cancer.Net". Cancer.Net. 2012-06-25. Archived from the original on 2017-08-27. Retrieved 2017-08-27.
- "Cervical cancer statistics and prognosis". Cancer Research UK. Archived from the original on 2007-05-20. Retrieved 2007-03-24.
- Nygård M (June 2011). "Screening for cervical cancer: when theory meets reality". BMC Cancer. 11: 240. doi:10.1186/1471-2407-11-240. PMC 3146446. PMID 21668947.
- Muliira RS, Salas AS, O'Brien B (2016). "Quality of Life among Female Cancer Survivors in Africa: An Integrative Literature Review". Asia-Pacific Journal of Oncology Nursing. 4 (1): 6–17. doi:10.4103/2347-5625.199078. PMC 5297234. PMID 28217724.
- "WHO Disease and injury country estimates". World Health Organization. 2009. Archived from the original on 2009-11-11. Retrieved Nov 11, 2009.
- "WHO Fact Sheet on HPV and Cervical Cancer". World Health Organization. 2018-01-24. Retrieved 2019-06-04.
- Kent A (Winter 2010). "HPV Vaccination and Testing". Reviews in Obstetrics & Gynecology. 3 (1): 33–4. PMC 2876324. PMID 20508781.
- Cordeiro CN, Gemignani ML (March 2017). "Gynecologic Malignancies in Pregnancy: Balancing Fetal Risks With Oncologic Safety". Obstetrical & Gynecological Survey. 72 (3): 184–193. doi:10.1097/OGX.0000000000000407. PMC 5358514. PMID 28304416.
- "Incidence and mortality rates". Archived from the original on 2009-09-12.
- "Archived copy". Archived from the original on 2011-03-14. Retrieved 2011-03-07.CS1 maint: archived copy as title (link)
- Hall, Michaela; et al. (2 October 2018). "The projected timeframe until cervical cancer elimination in Australia: a modelling study". Lancet. Retrieved 8 November 2018.
- MacDonald N, Stanbrook MB, Hébert PC (September 2008). "Human papillomavirus vaccine risk and reality". CMAJ (in French). 179 (6): 503, 505. doi:10.1503/cmaj.081238. PMC 2527393. PMID 18762616.
- National Cancer Registry Programme under Indian Council of Medical Research Reports
- Krishnatreya M, Kataki AC, Sharma JD, Nandy P, Gogoi G (2015). "Association of educational levels with survival in Indian patients with cancer of the uterine cervix". Asian Pacific Journal of Cancer Prevention. 16 (8): 3121–3. doi:10.7314/apjcp.2015.16.8.3121. PMID 25921107.
- "Cervical cancer statistics". Cancer Research UK. Archived from the original on 7 October 2014. Retrieved 27 October 2014.
- "Cancer Stat Facts: Cervical Cancer". National Cancer Institute SEER Program. Retrieved 2019-06-04.
- Armstrong EP (April 2010). "Prophylaxis of cervical cancer and related cervical disease: a review of the cost-effectiveness of vaccination against oncogenic HPV types". Journal of Managed Care Pharmacy. 16 (3): 217–30. doi:10.18553/jmcp.2010.16.3.217. PMID 20331326. S2CID 14373353.
- zur Hausen H (May 2002). "Papillomaviruses and cancer: from basic studies to clinical application". Nature Reviews. Cancer. 2 (5): 342–50. doi:10.1038/nrc798. PMID 12044010. S2CID 4991177.
- Beachler DC, Kreimer AR, Schiffman M, Herrero R, Wacholder S, Rodriguez AC, et al. (January 2016). "Multisite HPV16/18 Vaccine Efficacy Against Cervical, Anal, and Oral HPV Infection". Journal of the National Cancer Institute. 108 (1): djv302. doi:10.1093/jnci/djv302. PMC 4862406. PMID 26467666.
- Kreimer AR, Herrero R, Sampson JN, Porras C, Lowy DR, Schiller JT, et al. (August 2018). "Evidence for single-dose protection by the bivalent HPV vaccine-Review of the Costa Rica HPV vaccine trial and future research studies". Vaccine. 36 (32 Pt A): 4774–4782. doi:10.1016/j.vaccine.2017.12.078. PMC 6054558. PMID 29366703.
- Menczer J (February 2003). "The low incidence of cervical cancer in Jewish women: has the puzzle finally been solved?". The Israel Medical Association Journal. 5 (2): 120–3. PMID 12674663. Archived from the original on 8 December 2015. Retrieved 28 November 2015.
- Heins HC, Dennis EJ, Pratthomas HR (October 1958). "The possible role of smegma in carcinoma of the cervix". American Journal of Obstetrics and Gynecology. 76 (4): 726–33, discussion 733–5. doi:10.1016/0002-9378(58)90004-8. PMID 13583012.
- Alexander ER (June 1973). "Possible etiologies of cancer of the cervix other than herpesvirus". Cancer Research. 33 (6): 1485–90. PMID 4352386.
- Rosenblatt A, Guidi HG (2009-08-06). Human Papillomavirus: A Practical Guide for Urologists. Springer Science & Business Media. ISBN 9783540709749.
- Dürst M, Gissmann L, Ikenberg H, zur Hausen H (June 1983). "A papillomavirus DNA from a cervical carcinoma and its prevalence in cancer biopsy samples from different geographic regions". Proceedings of the National Academy of Sciences of the United States of America. 80 (12): 3812–5. Bibcode:1983PNAS...80.3812D. doi:10.1073/pnas.80.12.3812. PMC 394142. PMID 6304740..
- Lowy DR, Schiller JT (May 2006). "Prophylactic human papillomavirus vaccines". The Journal of Clinical Investigation. 116 (5): 1167–73. doi:10.1172/JCI28607. PMC 1451224. PMID 16670757.
- McNeil C (April 2006). "Who invented the VLP cervical cancer vaccines?". Journal of the National Cancer Institute. 98 (7): 433. doi:10.1093/jnci/djj144. PMID 16595773.
- "WHO rolls out plan to rid world of cervical cancer, saving millions of lives". UN News. 2020-11-16. Retrieved 2020-11-27.
- Cancer Institute NSW (2013). "Information about cervical screening for Aboriginal women". NSW Government. Archived from the original on 11 April 2013.
- Mary-Anne Romano (17 October 2011). "Aboriginal cervical cancer rates parallel health inequity". Science Network Western Australia. Archived from the original on 14 May 2013.
- Australian Cervical Cancer Foundation. "Vision and Mission". Australian Cervical Cancer Foundation. Archived from the original on 12 May 2013.
- Australian Cervical Cancer Foundation. "Our People". Australian Cervical Cancer Foundation. Archived from the original on 12 May 2013.
- Gillian Bradford (16 October 2006). "Janette Howard speaks on her battle with cervical cancer". Australian Broadcasting Corporation. Archived from the original on 3 November 2012.
- Tiro JA, Meissner HI, Kobrin S, Chollette V (February 2007). "What do women in the U.S. know about human papillomavirus and cervical cancer?". Cancer Epidemiology, Biomarkers & Prevention. 16 (2): 288–94. doi:10.1158/1055-9965.EPI-06-0756. PMID 17267388.
- Singh GK, Miller BA, Hankey BF, Edwards BK (September 2004). "Persistent area socioeconomic disparities in U.S. incidence of cervical cancer, mortality, stage, and survival, 1975–2000". Cancer. 101 (5): 1051–7. doi:10.1002/cncr.20467. PMID 15329915. S2CID 26033629.
Further reading
- Arbyn M, Castellsagué X, de Sanjosé S, Bruni L, Saraiya M, Bray F, Ferlay J (December 2011). "Worldwide burden of cervical cancer in 2008". Annals of Oncology. 22 (12): 2675–86. doi:10.1093/annonc/mdr015. PMID 21471563.
- Chuang LT, Temin S, Camacho R, Dueñas-Gonzalez A, Feldman S, Gultekin M, et al. (October 2016). "Management and Care of Women With Invasive Cervical Cancer: American Society of Clinical Oncology Resource-Stratified Clinical Practice Guideline". Journal of Global Oncology. 2 (5): 311–340. doi:10.1200/JGO.2016.003954. PMC 5493265. PMID 28717717.
- Peto J, Gilham C, Fletcher O, Matthews FE (2004). "The cervical cancer epidemic that screening has prevented in the UK". Lancet. 364 (9430): 249–56. doi:10.1016/S0140-6736(04)16674-9. PMID 15262102. S2CID 11059712.
- Pimenta JM, Galindo C, Jenkins D, Taylor SM (November 2013). "Estimate of the global burden of cervical adenocarcinoma and potential impact of prophylactic human papillomavirus vaccination". BMC Cancer. 13 (1): 553. doi:10.1186/1471-2407-13-553. PMC 3871005. PMID 24261839.
- Bhatla N, Aoki D, Sharma DN, Sankaranarayanan R (October 2018). "Cancer of the cervix uteri". International Journal of Gynaecology and Obstetrics. 143 Suppl 2: 22–36. doi:10.1002/ijgo.12611. PMID 30306584.
External links
| Classification | |
|---|---|
| External resources |
| Wikimedia Commons has media related to Cervical cancer. |