Laminin, beta 1
Laminin subunit beta-1 is a protein that in humans is encoded by the LAMB1 gene.[5][6][7][8]
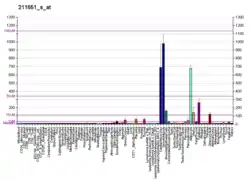
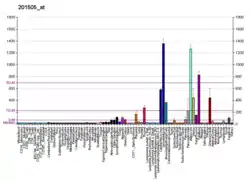
Laminins, a family of extracellular matrix glycoproteins, are the major noncollagenous constituent of basement membranes. They have been implicated in a wide variety of biological processes including cell adhesion, differentiation, migration, signaling, neurite outgrowth and metastasis. Laminins are composed of 3 non identical chains: laminin alpha, beta and gamma (formerly A, B1, and B2, respectively) and they form a cruciform structure consisting of 3 short arms, each formed by a different chain, and a long arm composed of all 3 chains. Each laminin chain is a multidomain protein encoded by a distinct gene. Several isoforms of each chain have been described. Different alpha, beta and gamma chain isomers combine to give rise to different heterotrimeric laminin isoforms, which are designated by Arabic numerals in the order of their discovery, i.e. alpha1beta1gamma1 heterotrimer is laminin 1. The biological functions of the different chains and trimer molecules are largely unknown, but some of the chains have been shown to differ with respect to their tissue distribution, presumably reflecting diverse functions in vivo. This gene encodes the beta chain isoform laminin, beta 1. The beta 1 chain has 7 structurally distinct domains, which it shares with other beta chain isomers. The C-terminal helical region containing domains I and II are separated by domain alpha, domains III and V contain several EGF-like repeats, and domains IV and VI have a globular conformation. Laminin, beta 1 is expressed in most tissues that produce basement membranes, and is one of the 3 chains constituting laminin 1, the first laminin isolated from Engelbreth-Holm-Swarm (EHS) tumor. A sequence in the beta 1 chain that is involved in cell attachment, chemotaxis, and binding to the laminin receptor was identified and shown to have the capacity to inhibit metastasis.[8]
5′-UTR of Laminin-B1 harbors IRES (internal ribosome entry site) between −293 and −1 upstream of the start codon. IRES are involved in cancer malignancy.[9]
References
- GRCh38: Ensembl release 89: ENSG00000091136 - Ensembl, May 2017
- GRCm38: Ensembl release 89: ENSMUSG00000002900 - Ensembl, May 2017
- "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- Ikonen J, Pikkarainen T, Savolainen ER, Tryggvason K (Feb 1989). "A Hpa I polymorphism in the human laminin B1 chain gene on 7q22". Nucleic Acids Res. 17 (1): 473. doi:10.1093/nar/17.1.473. PMC 331592. PMID 2563160.
- Roche KB, Moore JW, Surana RB, Wilson BE (May 1989). "Aortic root dilatation associated with partial trisomy 7(q31.2----qter)". Pediatr Cardiol. 10 (1): 53–5. doi:10.1007/BF02328637. PMID 2704655. S2CID 22352528.
- Bonneau D, Huret JL, Godeau G, Couet D, Putterman M, Tanzer J, Babin P, Larregue M (Sep 1991). "Recurrent ctb(7)(q31.3) and possible laminin involvement in a neonatal cutis laxa with a Marfan phenotype". Hum Genet. 87 (3): 317–9. doi:10.1007/bf00200911. PMID 1864606. S2CID 32445071.
- "Entrez Gene: LAMB1 laminin, beta 1".
- Petz M, Them N, Huber H, Beug H, Mikulits W (January 2012). "La enhances IRES-mediated translation of laminin B1 during malignant epithelial to mesenchymal transition". Nucleic Acids Research. 40 (1): 290–302. doi:10.1093/nar/gkr717. PMID 21896617.
Further reading
- Ljubimova JY, Fujita M, Khazenzon NM, et al. (2006). "Changes in laminin isoforms associated with brain tumor invasion and angiogenesis". Front. Biosci. 11 (1): 81–8. doi:10.2741/1781. PMC 3506377. PMID 16146715.
- Vuolteenaho R, Chow LT, Tryggvason K (1990). "Structure of the human laminin B1 chain gene". J. Biol. Chem. 265 (26): 15611–6. PMID 1975589.
- Sasaki M, Kato S, Kohno K, et al. (1987). "Sequence of the cDNA encoding the laminin B1 chain reveals a multidomain protein containing cysteine-rich repeats". Proc. Natl. Acad. Sci. U.S.A. 84 (4): 935–9. doi:10.1073/pnas.84.4.935. PMC 304334. PMID 3493487.
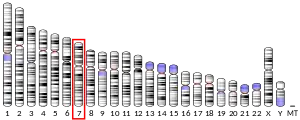
- Pikkarainen T, Eddy R, Fukushima Y, et al. (1987). "Human laminin B1 chain. A multidomain protein with gene (LAMB1) locus in the q22 region of chromosome 7". J. Biol. Chem. 262 (22): 10454–62. PMID 3611077.
- Jaye M, Modi WS, Ricca GA, et al. (1987). "Isolation of a cDNA clone for the human laminin-B1 chain and its gene localization". Am. J. Hum. Genet. 41 (4): 605–15. PMC 1684322. PMID 3661559.
- Burgeson RE, Chiquet M, Deutzmann R, et al. (1994). "A new nomenclature for the laminins". Matrix Biol. 14 (3): 209–11. doi:10.1016/0945-053X(94)90184-8. PMID 7921537.
- Lohi J, Leivo I, Franssila K, Virtanen I (1997). "Changes in the distribution of integrins and their basement membrane ligands during development of human thyroid follicular epithelium". Histochem. J. 29 (4): 337–45. doi:10.1023/A:1026482700109. PMID 9184849. S2CID 25517944.
- Taylor J, Muntoni F, Robb S, et al. (1997). "Early onset autosomal dominant myopathy with rigidity of the spine: a possible role for laminin beta 1?". Neuromuscul. Disord. 7 (4): 211–6. doi:10.1016/S0960-8966(97)00461-6. PMID 9196901. S2CID 11981067.
- Sewry CA, D'Alessandro M, Wilson LA, et al. (1997). "Expression of laminin chains in skin in merosin-deficient congenital muscular dystrophy". Neuropediatrics. 28 (4): 217–22. doi:10.1055/s-2007-973703. PMID 9309712.
- Mahida YR, Beltinger J, Makh S, et al. (1998). "Adult human colonic subepithelial myofibroblasts express extracellular matrix proteins and cyclooxygenase-1 and -2". Am. J. Physiol. 273 (6 Pt 1): G1341–8. doi:10.1152/ajpgi.1997.273.6.G1341. PMID 9435560.
- O'Grady P, Thai TC, Saito H (1998). "The laminin-nidogen complex is a ligand for a specific splice isoform of the transmembrane protein tyrosine phosphatase LAR". J. Cell Biol. 141 (7): 1675–84. doi:10.1083/jcb.141.7.1675. PMC 2133008. PMID 9647658.
- Koch M, Olson PF, Albus A, et al. (1999). "Characterization and expression of the laminin gamma3 chain: a novel, non-basement membrane-associated, laminin chain". J. Cell Biol. 145 (3): 605–18. doi:10.1083/jcb.145.3.605. PMC 2185082. PMID 10225960.
- Merlini L, Villanova M, Sabatelli P, et al. (1999). "Decreased expression of laminin beta 1 in chromosome 21-linked Bethlem myopathy". Neuromuscul. Disord. 9 (5): 326–9. doi:10.1016/S0960-8966(99)00022-X. PMID 10407855. S2CID 53256492.
- Kikkawa Y, Sanzen N, Fujiwara H, et al. (2000). "Integrin binding specificity of laminin-10/11: laminin-10/11 are recognized by alpha 3 beta 1, alpha 6 beta 1 and alpha 6 beta 4 integrins". J. Cell Sci. 113. ( Pt 5): 869–76. PMID 10671376.
- Hirosaki T, Mizushima H, Tsubota Y, et al. (2000). "Structural requirement of carboxyl-terminal globular domains of laminin alpha 3 chain for promotion of rapid cell adhesion and migration by laminin-5". J. Biol. Chem. 275 (29): 22495–502. doi:10.1074/jbc.M001326200. PMID 10801807.
- Champliaud MF, Virtanen I, Tiger CF, et al. (2000). "Posttranslational modifications and beta/gamma chain associations of human laminin alpha1 and laminin alpha5 chains: purification of laminin-3 from placenta". Exp. Cell Res. 259 (2): 326–35. doi:10.1006/excr.2000.4980. PMID 10964500.
- Pedraza C, Geberhiwot T, Ingerpuu S, et al. (2000). "Monocytic cells synthesize, adhere to, and migrate on laminin-8 (alpha 4 beta 1 gamma 1)". J. Immunol. 165 (10): 5831–8. doi:10.4049/jimmunol.165.10.5831. PMID 11067943.