Lauric acid
Lauric acid or systematically, dodecanoic acid, is a saturated fatty acid with a 12-carbon atom chain, thus having many properties of medium-chain fatty acids, is a bright white, powdery solid with a faint odor of bay oil or soap. The salts and esters of lauric acid are known as laurates.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Dodecanoic acid | |
| Other names
n-Dodecanoic acid, Dodecylic acid, Dodecoic acid,
Laurostearic acid, Vulvic acid, 1-Undecanecarboxylic acid, Duodecylic acid, C12:0 (Lipid numbers) | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.075 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H24O2 | |
| Molar mass | 200.322 g·mol−1 |
| Appearance | White powder |
| Odor | Slight odor of bay oil |
| Density | 1.007 g/cm3 (24 °C)[1] 0.8744 g/cm3 (41.5 °C)[2] 0.8679 g/cm3 (50 °C)[3] |
| Melting point | 43.8 °C (110.8 °F; 316.9 K)[3] |
| Boiling point | 297.9 °C (568.2 °F; 571.0 K) 282.5 °C (540.5 °F; 555.6 K) at 512 mmHg[1] 225.1 °C (437.2 °F; 498.2 K) at 100 mmHg[3][4] |
| 37 mg/L (0 °C) 55 mg/L (20 °C) 63 mg/L (30 °C) 72 mg/L (45 °C) 83 mg/L (100 °C)[5] | |
| Solubility | Soluble in alcohols, diethyl ether, phenyls, haloalkanes, acetates[5] |
| Solubility in methanol | 12.7 g/100 g (0 °C) 120 g/100 g (20 °C) 2250 g/100 g (40 °C)[5] |
| Solubility in acetone | 8.95 g/100 g (0 °C) 60.5 g/100 g (20 °C) 1590 g/100 g (40 °C)[5] |
| Solubility in ethyl acetate | 9.4 g/100 g (0 °C) 52 g/100 g (20°C) 1250 g/100 g (40°C)[5] |
| Solubility in toluene | 15.3 g/100 g (0 °C) 97 g/100 g (20°C) 1410 g/100 g (40°C)[5] |
| log P | 4.6[6] |
| Vapor pressure | 2.13·10−6 kPa (25 °C)[6] 0.42 kPa (150 °C)[4] 6.67 kPa (210 °C)[7] |
| Acidity (pKa) | 5.3 (20 °C)[6] |
| Thermal conductivity | 0.442 W/m·K (solid)[2] 0.1921 W/m·K (72.5 °C) 0.1748 W/m·K (106 °C)[1] |
Refractive index (nD) |
1.423 (70 °C)[1] 1.4183 (82 °C)[3] |
| Viscosity | 6.88 cP (50 °C) 5.37 cP (60 °C)[2] |
| Structure | |
| Monoclinic (α-form)[8] Triclinic, aP228 (γ-form)[9] | |
| P21/a, No. 14 (α-form)[8] P1, No. 2 (γ-form)[9] | |
| 2/m (α-form)[8] 1 (γ-form)[9] | |
α = 90°, β = 129.22°, γ = 90° | |
| Thermochemistry | |
Heat capacity (C) |
404.28 J/mol·K[4] |
Std enthalpy of formation (ΔfH⦵298) |
−775.6 kJ/mol[6] |
Std enthalpy of combustion (ΔcH⦵298) |
7377 kJ/mol 7425.8 kJ/mol (292 K)[4] |
| Hazards | |
| GHS pictograms | 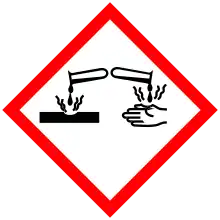 |
| GHS Signal word | Danger |
| H412[7] | |
| P273[7] | |
| NFPA 704 (fire diamond) | |
| Flash point | > 113 °C (235 °F; 386 K)[7] |
| Related compounds | |
Related compounds |
Glyceryl laurate |
| Related compounds | |
Related compounds |
Undecanoic acid Tridecanoic acid Dodecanol Dodecanal Sodium lauryl sulfate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Occurrence
Lauric acid, as a component of triglycerides, comprises about half of the fatty-acid content in coconut milk, coconut oil, laurel oil, and palm kernel oil (not to be confused with palm oil),[10][11] Otherwise, it is relatively uncommon. It is also found in human breast milk (6.2% of total fat), cow's milk (2.9%), and goat's milk (3.1%).[10]
In various plants
- The palm tree Attalea speciosa, a species popularly known in Brazil as babassu – 50% in babassu oil
- Attalea cohune, the cohune palm (also rain tree, American oil palm, corozo palm or manaca palm) – 46.5% in cohune oil
- Astrocaryum murumuru (Arecaceae) a palm native to the Amazon – 47.5% in "murumuru butter"
- Coconut oil 49%
- Pycnanthus kombo (African nutmeg)
- Virola surinamensis (wild nutmeg) 7.8–11.5%
- Peach palm seed 10.4%
- Betel nut 9%
- Date palm seed 0.56–5.4%
- Durio graveolens (a species of durian) 1.31%.[12]
- Macadamia nut 0.072–1.1%
- Plum 0.35–0.38%
- Watermelon seed 0.33%
- Viburnum opulus 0.24-0.33%[13]
- Citrullus lanatus (egusi melon)
- Pumpkin flower 205 ppm, pumpkin seed 472 ppm
In Insects
- Black soldier fly Hermetia illucens 30-50 mg/100 mg fat.[14]
Properties
Although 95% of medium-chain triglycerides are absorbed through the portal vein, only 25–30% of lauric acid is absorbed through it.[15]
Like many other fatty acids, lauric acid is inexpensive, has a long shelf-life, is nontoxic, and is safe to handle. It is used mainly for the production of soaps and cosmetics. For these purposes, lauric acid is reacted with sodium hydroxide to give sodium laurate, which is a soap. Most commonly, sodium laurate is obtained by saponification of various oils, such as coconut oil. These precursors give mixtures of sodium laurate and other soaps.[11]
Laboratory use
In the laboratory, lauric acid may be used to investigate the molar mass of an unknown substance via the freezing-point depression. The choice of lauric acid is convenient because the melting point of the pure compound is relatively high (43.8°C). Its cryoscopic constant is 3.9°C·kg/mol. By melting lauric acid with the unknown substance, allowing it to cool, and recording the temperature at which the mixture freezes, the molar mass of the unknown compound may be determined.[16]
Potential medicinal properties
Lauric acid increases total serum cholesterol more than many other fatty acids, but mostly high-density lipoprotein (HDL) (the "good" blood cholesterol). As a result, lauric acid has been characterized as having "a more favorable effect on total HDL cholesterol than any other fatty acid [examined], either saturated or unsaturated".[17] In general, a lower total/HDL serum cholesterol ratio correlates with a decrease in atherosclerotic risk.[18] Nonetheless, an extensive meta-analysis on foods affecting the total LDL/serum cholesterol ratio found in 2003 that the net effects of lauric acid on coronary artery disease outcomes remained uncertain.[19] A 2016 review of coconut oil (which is nearly half lauric acid) was similarly inconclusive about the effects on cardiovascular disease risk.[15]
References
- G., Chuah T.; D., Rozanna; A., Salmiah; Y., Thomas Choong S.; M., Sa'ari (2006). "Fatty acids used as phase change materials (PCMs) for thermal energy storage in building material applications" (PDF). University Putra Malaysia. Archived from the original (PDF) on 2014-11-03. Retrieved 2014-06-22.
- Mezaki, Reiji; Mochizuki, Masafumi; Ogawa, Kohei (2000). Engineering data on mixing (1st ed.). Elsevier Science B.V. p. 278. ISBN 0-444-82802-8.
- Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- Dodecanoic acid in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD), http://webbook.nist.gov (retrieved 2014-06-14)
- Seidell, Atherton; Linke, William F. (1952). Solubilities of inorganic and organic compounds (3rd ed.). New York: D. Van Nostrand Company. pp. 742–743.
- CID 3893 from PubChem
- Sigma-Aldrich Co., Lauric acid. Retrieved on 2014-06-14.
- Vand, V.; Morley, W. M.; Lomer, T. R. (1951). "The crystal structure of lauric acid". Acta Crystallographica. 4 (4): 324–329. doi:10.1107/S0365110X51001069.
- Sydow, Erik von (1956). "On the structure of the crystal form A of lauric acid" (PDF). actachemscand.org. Acta Chemica Scandinavica. Retrieved 2014-06-14.
- Beare-Rogers, J.; Dieffenbacher, A.; Holm, J.V. (2001). "Lexicon of lipid nutrition (IUPAC Technical Report)". Pure and Applied Chemistry. 73 (4): 685–744. doi:10.1351/pac200173040685. S2CID 84492006.
- David J. Anneken, Sabine Both, Ralf Christoph, Georg Fieg, Udo Steinberner, Alfred Westfechtel "Fatty Acids" in Ullmann's Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a10_245.pub2
- Nasaruddin, Mohd hanif; Noor, Noor Qhairul Izzreen Mohd; Mamat, Hasmadi (2013). "Komposisi Proksimat dan Komponen Asid Lemak Durian Kuning (Durio graveolens) Sabah" [Proximate and Fatty Acid Composition of Sabah Yellow Durian (Durio graveolens)] (PDF). Sains Malaysiana (in Malay). 42 (9): 1283–1288. ISSN 0126-6039. OCLC 857479186. Retrieved 28 November 2017.
- Zarifikhosroshahi; Tugba Murathan; Kafkas; Okatan (2019). "Variation in volatile and fatty acid contents among Viburnum opulus L. Fruits growing different locations". Scientia Horticulturae. 264: 109160. doi:10.1016/j.scienta.2019.109160.
- Montevecchi, G.; Zanasi, L.; Masino, F.; Maistrello, L.; Antonelli, A. (2019). "Black soldier fly (Hermetia illucens L.): effect on the fat integrity using different approaches to the killing of the prepupae". Journal of Insects as Food and Feed. 6 (2): 121–131. doi:10.3920/JIFF2019.0002.
- Eyres L, Eyres MF, Chisholm A, Brown RC (2016). "Coconut oil consumption and cardiovascular risk factors in humans". Nutrition Reviews. 74 (4): 267–280. doi:10.1093/nutrit/nuw002. PMC 4892314. PMID 26946252.
- http://faculty.sites.uci.edu/chem1l/files/2015/04/Freezing-Point-Depression.pdf
- Mensink RP, Zock PL, Kester AD, Katan MB (May 2003). "Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials". American Journal of Clinical Nutrition. 77 (5): 1146–1155. doi:10.1093/ajcn/77.5.1146. ISSN 0002-9165. PMID 12716665.
- Thijssen, M.A. and R.P. Mensink. (2005). Fatty Acids and Atherosclerotic Risk. In Arnold von Eckardstein (Ed.) Atherosclerosis: Diet and Drugs. Springer. pp. 171–172. ISBN 978-3-540-22569-0.
- Effects of dietary fatty acids and carbohydrates on the ratio of serum total to HDL cholesterol and on serum lipids and apolipoproteins: a meta-analysis of 60 controlled trials
Further reading
- Berner, Louise A. (1993). Defining the Role of Milkfat in Balanced Diets. In John E. Kinsella (Ed.) Advances in Food and Nutrition Research – Volume 37. Academic Press. pp. 159–166. ISBN 978-0-12-016437-0.
- Kabara, Jon J. (1978). The Pharmacological Effect of Lipids. Champaign IL: American Oil Chemist's Society. ISBN 9991817697.
- Kabara, Jon J. (2008). Fats Are Good for You and Other Secrets – How Saturated Fat and Cholesterol Actually Benefit the Body. North Atlantic Books. ISBN 1-55643-690-4.

