Mercury sulfide
Mercury sulfide, mercuric sulfide, mercury sulphide, or mercury(II) sulfide is a chemical compound composed of the chemical elements mercury and sulfur. It is represented by the chemical formula HgS. It is virtually insoluble in water.[4]
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Mercury sulfide | |
| Other names | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.014.270 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| HgS | |
| Molar mass | 232.66 g/mol |
| Density | 8.10 g/cm3 |
| Melting point | 580 °C (1,076 °F; 853 K) decomposes |
| insoluble | |
| Band gap | 2.1 eV (direct, α-HgS) [1] |
| −55.4·10−6 cm3/mol | |
Refractive index (nD) |
w=2.905, e=3.256, bire=0.3510 (α-HgS) [2] |
| Thermochemistry | |
Std molar entropy (S |
78 J·mol−1·K−1[3] |
Std enthalpy of formation (ΔfH⦵298) |
−58 kJ·mol−1[3] |
| Hazards | |
| Safety data sheet | ICSC 0981 |
EU classification (DSD) (outdated) |
Very toxic (T+) Dangerous for the environment (N) |
| R-phrases (outdated) | R26/27/28, R33, R50/53 |
| S-phrases (outdated) | (S1/2), S13, S28, S45, S60, S61 |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions |
Mercury oxide Mercury selenide Mercury telluride |
Other cations |
Zinc sulfide Cadmium sulfide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
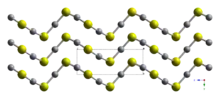
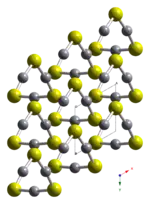
Crystal structure

HgS is dimorphic with two crystal forms:
- red cinnabar (α-HgS, trigonal, hP6, P3221), is the form in which mercury is most commonly found in nature.
- black, metacinnabar (β-HgS), is less common in nature and adopts the zinc blende (T2d-F43m) crystal structure.
Crystals of red, α-HgS, are optically active. This is caused by the Hg-S helices in the structure.[5]
Preparation and chemistry
β-HgS is precipitated as a black powder when H2S is bubbled through solutions of Hg(II) salts.[6] β-HgS is unreactive to all but concentrated acids.[4]
Mercury metal is produced from the cinnabar ore by roasting in air and condensing the vapour.[4]
Uses
When α-HgS is used as a red pigment, it is known as vermilion. The tendency of vermilion to darken has been ascribed to conversion from red α-HgS to black β-HgS. However β-HgS was not detected at excavations in Pompeii, where originally red walls darkened, and was attributed to the formation of Hg-Cl compounds (e.g., corderoite, calomel, and terlinguaite) and calcium sulfate, gypsum.[7]
As the mercury cell as used in the chlor-alkali industry (Castner–Kellner process) is being phased out over concerns over mercury emissions, the metallic mercury from these setups is converted into mercury sulfide for underground storage.
See also
- Mercury poisoning
- Mercury(I) sulfide (mercurous sulfide), Hg
2S
References
- L. I. Berger, Semiconductor Materials (1997) CRC Press ISBN 0-8493-8912-7
- Webminerals
- Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A22. ISBN 978-0-618-94690-7.
- Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 1406. ISBN 978-0-08-022057-4.
- A. M. Glazer, K. Stadnicka (1986). "On the origin of optical activity in crystal structures". J. Appl. Cryst. 19 (2): 108–122. doi:10.1107/S0021889886089823.
- Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York: Wiley-Interscience, ISBN 0-471-19957-5
- Cotte, M; Susini J; Metrich N; Moscato A; Gratziu C; Bertagnini A; Pagano M (2006). "Blackening of Pompeian Cinnabar Paintings: X-ray Microspectroscopy Analysis". Anal. Chem. 78 (21): 7484–7492. doi:10.1021/ac0612224. PMID 17073416.
