Selenium dichloride
Selenium dichloride is the inorganic compound with the formula SeCl2. It forms red-brown solutions in ethers. Selenium dichloride has been prepared by treating gray selenium with sulfuryl chloride. Adducts of selenium dichloride with thioethers and thioureas are well characterized. Related complexes of tellurium dichloride are known.
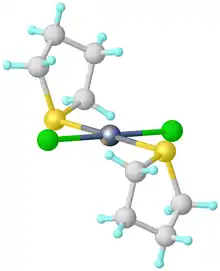
Structure of SeCl2(tetrahydrothiophene)2.[1]
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Cl2Se | |
| Molar mass | 149.87 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Solutions of selenium dichloride are unstable at room temperature, forming selenium monochloride after several minutes at room temperature:[2]
- 3 SeCl2 → Se2Cl2 + SeCl4
References
- Jolleys, A.; Levason, W.; Reid, G. (2013). "Thioether Coordination to Divalent Selenium Halide Acceptors – Synthesis, Properties and Structures". Dalton Transactions. 42 (8): 2963–2972. doi:10.1039/C2DT32665E. PMID 23250231.
- Maaninen, Arto; Chivers, Tristram; Parvez, Masood; Pietikäinen, Jarkko; Laitinen, Risto S. (1999). "Syntheses of THF Solutions of SeX2 (X = Cl, Br) and a New Route to Selenium Sulfides SenS8-n (n = 1-5): X-Ray Crystal Structures of SeCl2(tht)2 and SeCl2·tmtu". Inorganic Chemistry. 38: 4093–4097. doi:10.1021/ic981430h.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.