Anti-obesity medication
Anti-obesity medication or weight loss medications are pharmacological agents that reduce or control weight. These medications alter one of the fundamental processes of the human body, weight regulation, by altering either appetite, or absorption of calories.[1] The main treatment modalities for overweight and obese individuals remain dieting and physical exercise.
| Part of a series on |
| Human body weight |
|---|
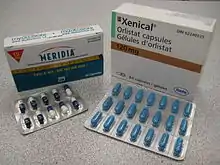
In the United States orlistat (Xenical) is currently approved by the FDA for long-term use.[2][3] It reduces intestinal fat absorption by inhibiting pancreatic lipase. Rimonabant (Acomplia), a second medication, works via a specific blockade of the endocannabinoid system. It has been developed from the knowledge that cannabis smokers often experience hunger, which is often referred to as "the munchies".[4] It had been approved in Europe for the treatment of obesity but has not received approval in the United States or Canada due to safety concerns.[5][6] The European Medicines Agency in October 2008 recommended the suspension of the sale of rimonabant as the risks seem to be greater than the benefits.[7] Sibutramine (Meridia), which acts in the brain to inhibit deactivation of the neurotransmitters, thereby decreasing appetite was withdrawn from the United States and Canadian markets in October 2010 due to cardiovascular concerns.[3][8]
Because of potential side effects, and limited evidence of small benefits in weight reduction especially in obese children and adolescents,[9] it is recommended that anti-obesity medications only be prescribed for obesity where it is hoped that the benefits of the treatment outweigh its risks.[10][11]
Mechanisms of action
Current and potential anti-obesity medications may operate through one or more of the following mechanisms:
- Catecholamine releasing agents such as amphetamine, phentermine, and related substituted amphetamines (e.g., bupropion) which act as appetite suppressants are the main tools used for the treatment of obesity.[12][13]
- Increase of the body's metabolism.
- Interference with the body's ability to absorb specific nutrients in food. For example, Orlistat (also known as Xenical and Alli) blocks fat breakdown and thereby prevents fat absorption. The OTC fiber supplements glucomannan and guar gum have been used for the purpose of inhibiting digestion and lowering caloric absorption
Anorectics are primarily intended to suppress the appetite, but most of the medications in this class also act as stimulants (e.g., amphetamine), and patients have abused medications "off label" to suppress appetite (e.g. digoxin).
History
The first described attempts at producing weight loss are those of Soranus of Ephesus, a Greek physician, in the second century AD. He prescribed elixirs of laxatives and purgatives, as well as heat, massage, and exercise. This remained the mainstay of treatment for well over a thousand years. It was not until the 1920s and 1930s that new treatments began to appear. Based on its effectiveness for hypothyroidism, thyroid hormone became a popular treatment for obesity in euthyroid people. It had a modest effect but produced the symptoms of hyperthyroidism as a side effect, such as palpitations and difficulty sleeping. 2,4-Dinitrophenol (DNP) was introduced in 1933; this worked by uncoupling the biological process of oxidative phosphorylation in mitochondria, causing them to produce heat instead of ATP. The most significant side effect was a sensation of warmth, frequently with sweating. Overdose, although rare, led to a rise in body temperature and, ultimately, fatal hyperthermia. By the end of 1938 DNP had fallen out of use because the FDA had become empowered to put pressure on manufacturers, who voluntarily withdrew it from the market.[14]
Amphetamines (marketed as Benzedrine) became popular for weight loss during the late 1930s. They worked primarily by suppressing appetite, and had other beneficial effects such as increased alertness. Use of amphetamines increased over the subsequent decades, including Obetrol and culminating in the "rainbow diet pill" regime.[15] This was a combination of multiple pills, all thought to help with weight loss, taken throughout the day. Typical regimens included stimulants, such as amphetamines, as well as thyroid hormone, diuretics, digitalis, laxatives, and often a barbiturate to suppress the side effects of the stimulants.[15] In 1967/1968 a number of deaths attributed to diet pills triggered a Senate investigation and the gradual implementation of greater restrictions on the market.[16] While rainbow diet pills were banned in the US in the late 1960s, they reappeared in South America and Europe in the 1980s.[15] Eventually rainbow diet pills were re-introduced into the US by the 2000s and led to additional adverse health effects.[17][18][19]
Meanwhile, phentermine had been FDA approved in 1959 and fenfluramine in 1973. The two were no more popular than other medications until in 1992 a researcher reported that when combined the two caused a 10% weight loss which was maintained for more than two years.[20] Fen-phen was born and rapidly became the most commonly prescribed diet medication. Dexfenfluramine (Redux) was developed in the mid-1990s as an alternative to fenfluramine with fewer side-effects, and received regulatory approval in 1996. However, this coincided with mounting evidence that the combination could cause valvular heart disease in up to 30% of those who had taken it, leading to withdrawal of Fen-phen and dexfenfluramine from the market in September 1997.[16]
Ephedra was removed from the US market in 2004 over concerns that it raises blood pressure and could lead to strokes and death.[21]
Medication
Some patients find that diet and exercise is not a viable option; for these patients, anti-obesity medications can be a last resort. Some prescription weight loss medications are stimulants, which are recommended only for short-term use, and thus are of limited usefulness for extremely obese patients, who may need to reduce weight over months or years.[22]
Orlistat
Orlistat (Xenical) reduces intestinal fat absorption by inhibiting the enzyme pancreatic lipase. Frequent oily bowel movements steatorrhea is a possible side effect of using Orlistat. But if fat in the diet is reduced, symptoms often improve. Originally available only by prescription, it was approved by the FDA for over-the-counter sale in February 2007.[23] On 26 May 2010, the U.S. Food and Drug Administration (FDA) has approved a revised label for Xenical to include new safety information about cases of severe liver injury that have been reported rarely with the use of this medication.[24] Of the 40 million users of Orlistat worldwide, 13 cases of severe liver damage have been reported.[25]
Cetilistat
Cetilistat is a medication designed to treat obesity. It acts in the same way as the older medication Orlistat by inhibiting pancreatic lipase, an enzyme that breaks down triglycerides in the intestine. Without this enzyme, triglycerides from the diet are prevented from being hydrolyzed into absorbable free fatty acids and are excreted undigested.[26]
A published phase 2 trial found cetilistat significantly reduced weight with and was better tolerated than orlistat.[27]
Lorcaserin
Lorcaserin (Belviq) was approved 28 June 2012 for obesity with other co-morbidities. The average weight loss by study participants was modest, but the most common side effects of the medication are considered benign.[28] It reduces appetite by activating a type of serotonin receptor known as the 5-HT2C receptor in a region of the brain called the hypothalamus, which is known to control appetite.[29] This drug has now been withdrawn from the market because a safety clinical trial shows an increased occurrence of cancer.[30]
Sibutramine
Sibutramine (Reductil or Meridia) is an anorectic or appetite suppressant, reducing the desire to eat. Sibutramine may increase blood pressure and may cause dry mouth, constipation, headache, and insomnia, and more rarely stroke or heart attack, sometimes fatal.
In the past, it was noted by the US that Meridia was a harmless medication for fighting obesity. The US District Court of the Northern District of Ohio rejected 113 cases complaining about the negative effects of the medication, stating that the clients lacked supporting facts and that the representatives involved were not qualified enough.[31]
Sibutramine has been withdrawn from the market in the United States,[32] the UK,[33] the EU,[34] Australia,[35] Canada,[36] Hong Kong[37] and Colombia.[38] Its risks (non-life-threatening myocardial infarction and stroke) have been shown to outweigh the benefits.
Rimonabant
Rimonabant (also known as SR141716; trade names Acomplia and Zimulti)[39] was an anorectic antiobesity medication that was first approved in Europe in 2006 but was withdrawn worldwide in 2008 due to serious psychiatric side effects; it was never approved in the United States.[40][41] Rimonabant is an inverse agonist for the cannabinoid receptor CB1 and was the first medication approved in that class.[42][43]
Metformin
In people with type 2 diabetes mellitus, the medication metformin (Glucophage) can reduce weight.[44] Metformin limits the amount of glucose that is produced by the liver as well as increases muscle consumption of glucose. It also helps in increasing the body's response to insulin.[45]
Exenatide/liraglutide/semaglutide
Exenatide (Byetta) is a long-acting analogue of the hormone GLP-1, which the intestines secrete in response to the presence of food. Among other effects, GLP-1 delays stomach emptying and promotes a feeling of fullness after eating. Some obese people are deficient in GLP-1, and dieting reduces GLP-1 further.[46] Byetta is currently available as a treatment for Diabetes mellitus type 2. Some, but not all, patients find that they lose substantial weight when taking Byetta. Drawbacks of Byetta include that it must be injected subcutaneously twice daily, and that it causes severe nausea in some patients, especially when therapy is initiated. Byetta is recommended only for patients with Type 2 Diabetes.[47]
Liraglutide (Saxenda) is another GLP-1 analogue for daily administration.
Semaglutide (Ozempic) is yet another GLP-1 analogue, more effective and administered once weekly.
Amylin/pramlinatide
An analogue of amylin (secreted by the Beta cells of the pancreas in a fixed ratio when insulin is released and activated) pramlintide, originally developed by Amylin Pharmaceuticals, now owned by AstraZeneca Pharmaceuticals, is currently available for treating diabetes and is in testing for treating obesity in non-diabetics.
Phentermine/topiramate
The combination of phentermine and topiramate, brand name Qsymia (formerly Qnexa) was approved by the U.S. FDA on 17 July 2012, as an obesity treatment complementary to a diet and exercise regimen.[48] The European Medicines Agency, by contrast, rejected the combination as a treatment for obesity, citing concerns about long-term effects on the heart and blood vessels, mental health and cognitive side-effects.
Naltrexone/bupropion
Naltrexone/bupropion is a combination medication used for weight loss in those that are either obese or overweight with some weight-related illnesses. It combines low doses of bupropion and naltrexone. Both medications have individually shown some evidence of effectiveness in weight loss, and the combination has been shown to have some synergistic effects on weight. In September 2014, a sustained release formulation of the medication was approved for marketing in the United States under the brand name Contrave.[49] The combination was approved for use in the European Union in March 2015, under the brand name Mysimba.[50]
Other medications
Other weight loss medications have also been associated with medical complications, such as fatal pulmonary hypertension and heart valve damage due to Redux and Fen-phen, and hemorrhagic stroke due phenylpropanolamine.[51][52] Many of these substances are related to amphetamine.
Tesofensine (NS2330) is a serotonin–noradrenaline–dopamine reuptake inhibitor from the phenyltropane family of medications, which is being developed for the treatment of obesity.[53] Tesofensine was originally developed by a Danish biotechnology company, NeuroSearch, who transferred the rights to Saniona in 2014.[54] Tesofensine has been evaluated in Phase 1 and Phase 2 human clinical studies with the aim of investigating treatment potential with regards to obesity.
Dietary supplements, foodstuffs, or programs for weight loss are heavily promoted through advertisements in print, on television, and on the internet. The US Food and Drug Administration recommends caution with use of these products,[55] since many of the claims of safety and effectiveness are unsubstantiated, and many of the studies purporting to demonstrate their effectiveness are funded by the manufactures and suffer a high degree of bias.[56] Individuals with anorexia nervosa or bulimia nervosa, and some athletes, try to control body weight with diet pills, laxatives, or diuretic medications, although the latter two generally have no impact on body fat and only cause short-lived weight-loss through dehydration.[57] Both diuretics and laxatives can cause electrolyte abnormalities which may cause cognitive, heart, and muscle problems, and can be fatal. Pyruvate, which is found in red apples, cheese, and red wine, is sometimes marketed as a weight loss supplement, but has not been thoroughly studied and its weight loss effect has not been demonstrated.[58]
Herbal and alternative medicine
Many products marketed as botanical weight loss supplements actually contain unapproved stimulants including analogues of amphetamine,[59] methamphetamine[60] and ephedra.[61] Some botanical supplements include high dosages of compounds found in plants with stimulant effects including yohimbine and higenamine.[62][63]
Canadian clinical practice guidelines state that there is insufficient evidence to recommend in favor of or against using herbal medicine, dietary supplements or homeopathy against obesity.[64]
Conjugated linoleic acid is claimed to help reduce obesity but it is ineffective for this use.[65]
The ECA Stack cannot be marketed in most developed countries but used to be marketed as a weight loss; it provided modest short term weight loss but evidence for the long term was lacking. Additionally there was a risk of adverse effects on the cardiovascular, mental, digestive, and nervous systems.[66]
Side effects
Some anti-obesity medications can have severe, even, lethal side effects, fen-phen being a famous example. Fen-phen was reported through the FDA to cause abnormal echocardiograms, heart valve problems, and rare valvular diseases.[67] One of, if not the first, to sound alarms was Sir Arthur MacNalty, Chief Medical Officer (United Kingdom). As early as the 1930s, he warned against the use of dinitrophenol as an anti-obesity medication and the injudicious and/or medically unsupervised use of thyroid hormone to achieve weight reduction.[68][69] The side effects are often associated with the medication's mechanism of action. In general, stimulants carry a risk of high blood pressure, faster heart rate, palpitations, closed-angle glaucoma, drug addiction, restlessness, agitation, and insomnia.
Another medication, orlistat, blocks absorption of dietary fats, and as a result may cause oily spotting bowel movements (steatorrhea), oily stools, stomach pain, and flatulence.[70] A similar medication designed for patients with Type 2 diabetes is Acarbose; which partially blocks absorption of carbohydrates in the small intestine, and produces similar side effects including stomach pain and flatulence.[71]
Research
Other classes of medications in development include lipase inhibitors, similar to orlistat. Another lipase inhibitor, called GT 389–255, was being developed by Peptimmune[72] (licensed from Genzyme). This was a novel combination of an inhibitor and a polymer designed to bind the undigested triglycerides therefore allowing increased fat expulsion without side effects such as oily stools that occur with orlistat. The development stalled as Phase 1 trials were conducted in 2004 and there was no further human clinical development afterward. In 2011, Peptimmune filed for Chapter 7 Liquidation.[73]
See also
References
- National Institute for Health and Clinical Excellence. Clinical guideline 43: Obesity: The prevention, identification, assessment and management of overweight and obesity in adults and children. London, 2006.
- "WIN – Publication – Prescription Medications for the Treatment of Obesity". National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). National Institutes of Health. Archived from the original on 13 January 2009. Retrieved 14 January 2009.
- "www.fda.gov".
- Wadman, Meredith (January 2006). "Rimonabant adds appetizing choice to slim obesity market". Nature Medicine. 12 (1): 27. doi:10.1038/nm0106-27. ISSN 1546-170X. PMID 16397550. S2CID 5298220.
- "Anti-obesity drug no magic bullet". Canadian Broadcasting Corporation. 2 January 2007. Retrieved 19 September 2008.
- "FDA Briefing Document NDA 21-888 Zimulti (rimonabant) Tablets, 20 mg Sanofi Aventis Advisory Committee" (PDF). Food and Drug Administration. 13 June 2007. Archived (PDF) from the original on 10 September 2008. Retrieved 19 September 2008.
- "The European Medicines Agency recommends suspension of the marketing authorisation of Acomplia". ema.europa.eu. Retrieved 30 August 2020.
- Government of Canada, Health Canada (23 October 2012). "Recalls and safety alerts". healthycanadians.gc.ca.
- Mead, E; Atkinson, G; Richter, B; Metzendorf, MI; Baur, L; Finer, N; Corpeleijn, E; O'Malley, C; Ells, LJ (29 November 2016). "Drug interventions for the treatment of obesity in children and adolescents". The Cochrane Database of Systematic Reviews. 11: CD012436. doi:10.1002/14651858.CD012436. PMC 6472619. PMID 27899001.
- Snow V, Barry P, Fitterman N, Qaseem A, Weiss K (2005). "Pharmacologic and surgical management of obesity in primary care: a clinical practice guideline from the American College of Physicians". Ann. Intern. Med. 142 (7): 525–31. doi:10.7326/0003-4819-142-7-200504050-00011. PMID 15809464.
- Cooke D, Bloom S (2006). "The obesity pipeline: current strategies in the development of anti-obesity drugs". Nature Reviews. Drug Discovery. 5 (11): 919–31. doi:10.1038/nrd2136. PMID 17080028. S2CID 28733429.
- "Evekeo Prescribing Information" (PDF). Arbor Pharmaceuticals LLC. April 2014. pp. 1–2. Retrieved 11 August 2015.
- Bray, G. A. (1993). "Use and Abuse of Appetite-Suppressant Drugs in the Treatment of Obesity". Annals of Internal Medicine. 119 (7, Pt. 2): 707–713. doi:10.7326/0003-4819-119-7_Part_2-199310011-00016. PMID 8363202. S2CID 39529451.
- Parascandola J (November 1974). "Dinitrophenol and bioenergetics: an historical perspective". Mol. Cell. Biochem. 5 (1–2): 69–77. doi:10.1007/BF01874175. PMID 4610359. S2CID 2656970.
- Cohen, Pieter A.; Goday, Alberto; Swann, John P. (September 2012). "The Return of Rainbow Diet Pills". American Journal of Public Health. 102 (9): 1676–1686. doi:10.2105/AJPH.2012.300655. PMC 3482033. PMID 22813089.
- Pool, Robert (2001). Fat: Fighting the Obesity Epidemic. Oxford, UK: Oxford University Press. ISBN 978-0-19-511853-7.
- Cohen, Pieter A. (19 December 2008). "Imported Fenproporex-based Diet Pills from Brazil: A Report of Two Cases". Journal of General Internal Medicine. 24 (3): 430–433. doi:10.1007/s11606-008-0878-4. PMC 2642570. PMID 19096898.
- Cohen, Pieter A.; McCormick, Danny; Casey, Carolyn; Dawson, Glen F.; Hacker, Karen A. (9 December 2007). "Imported Compounded Diet Pill Use Among Brazilian Women Immigrants in the United States". Journal of Immigrant and Minority Health. 11 (3): 229–236. doi:10.1007/s10903-007-9099-x. PMID 18066718. S2CID 8730835.
- Smith, Benjamin R.; Cohen, Pieter A. (May 2010). "Dependence on the Brazilian Diet Pill: A Case Report". American Journal on Addictions. 19 (3): 291–292. doi:10.1111/j.1521-0391.2010.00034.x. PMID 20525038.
- Weintraub M (May 1992). "Long-term weight control: The National Heart, Lung, and Blood Institute funded multimodal intervention study". Clin. Pharmacol. Ther. 51 (5): 581–5. doi:10.1038/clpt.1992.68. PMID 1445528. S2CID 221606104.
- Kolata, Gina (2007). Rethinking thin: The new science of weight loss – and the myths and realities of dieting. Picador. ISBN 978-0-312-42785-6.
- "Obesity Medication: Gastrointestinal Agents, Other, CNS Stimulants, Anorexiants, Glucagon-like Peptide-1 Agonists, Antidepressants, dopamine reuptake inhibitors; opioid antagonists". emedicine.medscape.com.
- "Press Announcements". Fda.gov. 8 (10): 1845. 1970. Bibcode:1970JPoSB...8.1845.. doi:10.1002/pol.1970.160081020.
- "FDA Drug Safety Communication: Completed safety review of Xenical/Alli (orlistat) and severe liver injury". fda.gov.
- Aronne, L.J.; Powell, A.G.; Apovian, C.M. (2011). "Emerging pharmacotherapy for obesity". Expert Opinion on Emerging Drugs. 16 (3): 587–96. doi:10.1517/14728214.2011.609168. PMID 21834735. S2CID 9253331.
- Yamada Y, Kato T, Ogino H, Ashina S, Kato K (2008). "Cetilistat (ATL-962), a novel pancreatic lipase inhibitor, ameliorates body weight gain and improves lipid profiles in rats". Hormone and Metabolic Research. 40 (8): 539–43. doi:10.1055/s-2008-1076699. PMID 18500680. S2CID 29076657.
- Kopelman, P.; Groot Gde, H.; Rissanen, A.; Rossner, S.; Toubro, S.; Palmer, R.; Hallam, R.; Bryson, A.; Hickling, R. I. (January 2010). "Weight loss, HbA1c reduction, and tolerability of cetilistat in a randomized, placebo-controlled phase 2 trial in obese diabetics: comparison with orlistat (Xenical)". Obesity. 18 (1): 108–15. doi:10.1038/oby.2009.155. PMID 19461584. S2CID 205526626.
- Bray, GA; Frühbeck, G; Ryan, DH; Wilding, JP (7 May 2016). "Management of obesity". Lancet. 387 (10031): 1947–56. doi:10.1016/S0140-6736(16)00271-3. PMID 26868660. S2CID 21805769.
- Shukla, AP; Kumar, RB; Aronne, LJ (2015). "Lorcaserin Hcl for the treatment of obesity". Expert Opinion on Pharmacotherapy. 16 (16): 2531–8. doi:10.1517/14656566.2015.1096345. PMID 26472579. S2CID 44520532.
- https://www.fda.gov/drugs/fda-drug-safety-podcasts/fda-requests-withdrawal-weight-loss-drug-belviq-belviq-xr-lorcaserin-market#:~:text=On%20February%2013%2C%202020%20FDA,an%20increased%20occurrence%20of%20cancer.
- Obesity, Fitness & Wellness Week (14 August 2004). "Legal Issues; Court dismisses claims against anti-obesity medication". Biotech Week: 11. ISSN 1535-2757.
- Rockoff, Jonathan D.; Dooren, Jennifer Corbett (8 October 2010). "Abbott Pulls Diet Drug Meridia Off US Shelves". The Wall Street Journal. Archived from the original on 11 October 2010. Retrieved 8 October 2010.
- "Top obesity drug sibutramine being suspended". BBC News. 22 January 2010. Archived from the original on 25 January 2010. Retrieved 22 January 2010.
- (in German) Sibutramin-Vertrieb in der Europäischen Union ausgesetzt Archived 19 July 2012 at Archive.today. Abbott Laboratories in Germany. Press Release 2010-01-21. Retrieved 2010-01-27
- "Sibutramine (brand name Reductil) Information – Australia". Abbott Laboratories. 2010. Archived from the original on 14 October 2010. Retrieved 8 October 2010.
- "MedEffect Canada - Advisories, Warnings and Recalls - Health Canada". hc-sc.gc.ca.
- "De-registration of pharmaceutical products containing sibutramine" (Press release). info.gov in Hong Kong. 2 November 2010. Retrieved 8 November 2010.
- "Top 10 Diet Pills That Work in 2014". TENMANIA. 2014. Retrieved 15 April 2014.
- "Rimonabant". AdisInsight. Retrieved 21 February 2017.
- Sam, AH; Salem, V; Ghatei, MA (2011). "Rimonabant: From RIO to Ban". Journal of Obesity. 2011: 432607. doi:10.1155/2011/432607. PMC 3136184. PMID 21773005.
- Moreira, FA; Crippa, JA (June 2009). "The psychiatric side-effects of rimonabant". Revista Brasileira de Psiquiatria (Sao Paulo, Brazil : 1999). 31 (2): 145–53. doi:10.1590/s1516-44462009000200012. PMID 19578688.
- Fong TM, Heymsfield SB (September 2009). "Cannabinoid-1 receptor inverse agonists: current understanding of mechanism of action and unanswered questions". Int J Obes (Lond). 33 (9): 947–55. doi:10.1038/ijo.2009.132. PMID 19597516.
- "European Approval Comes Early for Sanofi-Aventis' Acomplia". IHS. 23 June 2006.
- George A. Bray; Frank L. Greenway (1999). "Current and Potential Drugs for Treatment of Obesity: Table 19: Clinical trials with metformin for the treatment of obese diabetics". Endocrine Reviews. 20 (6): 805–87. doi:10.1210/er.20.6.805. PMID 10605627.
- "Diabetes Medications: Metformin" (PDF). Archived from the original (PDF) on 8 July 2015. Retrieved 21 October 2015.
- de Luis DA, Gonzalez Sagrado M, Conde R, Aller R, Izaola O (2007). "Decreased basal levels of glucagon-like peptide-1 after weight loss in obese subjects". Ann. Nutr. Metab. 51 (2): 134–8. doi:10.1159/000103273. PMID 17536190. S2CID 25707685.
- Sauer, N; Reining, F; Schulze Zur Wiesch, C; Burkhardt, T; Aberle, J (July 2015). "Off-label antiobesity treatment in patients without diabetes with GLP-1 agonists in clinical practice". Hormone and Metabolic Research. 47 (8): 560–4. doi:10.1055/s-0034-1387793. PMID 25230325.
- Salynn Boyles (17 July 2012). "FDA approves diet drug Qsymia : agency warns of increased risk for oral birth defects". WebMD., citing "FDA approves weight-management drug Qsymia" (Press release). U.S. Food and Drug Administration. 17 July 2012.
- "Contrave Extended-Release- naltrexone hydrochloride and bupropion hydrochloride tablet, extended release". DailyMed. 26 April 2019. Retrieved 5 August 2020.
- "Mysimba EPAR". European Medicines Agency (EMA). Retrieved 5 August 2020.
- Abenhaim L; Moride Y; Brenot F; et al. (August 1996). "Appetite-suppressant drugs and the risk of primary pulmonary hypertension. International Primary Pulmonary Hypertension Study Group". N. Engl. J. Med. 335 (9): 609–16. doi:10.1056/NEJM199608293350901. PMID 8692238.
- Alfred P. Fishman, MD (1999). "Aminorex to Fen/Phen: An Epidemic Foretold". Circulation. 99 (1): 156–161. doi:10.1161/01.CIR.99.1.156. PMID 9884392.
- Doggrell, SA (2009). "Tesofensine--a novel potent weight loss medicine. Evaluation of: Astrup A, Breum L, Jensen TJ, Kroustrup JP, Larsen TM. Effect of tesofensine on bodyweight loss, body composition, and quality of life in obese patients: a randomised, double-blind, placebo-controlled trial. Lancet 2008;372:1906-13" (PDF). Expert Opin Investig Drugs. 18 (7): 1043–6. doi:10.1517/13543780902967632. PMID 19548858. S2CID 207475155.
- "NeuroSearch A/S signs agreement to transfer Phase I-II projects NS2359 and NS2330 (Tesofensine)". newsclient.omxgroup.com.
- U.S. Food and Drug Administration: The Facts About Weight Loss Products and Programs Archived 11 August 2007 at the Wayback Machine
- "Prepared Statement of the Federal Trade Commission on the Marketing of Dietary Supplements" (Press release). Committee on Governmental Affairs, United States Senate. 8 October 2002. Archived from the original on 25 August 2006. Retrieved 7 August 2006.
- Martin M, Schlabach G, Shibinski K (1998). "The Use of Nonprescription Weight Loss Products Among Female Basketball, Softball, and Volleyball Athletes from NCAA Division I Institutions: Issues and Concerns". J Athl Train. 33 (1): 41–44. PMC 1320374. PMID 16558483.
- George A. Bray; Frank L. Greenway (1999). "Current and Potential Drugs for Treatment of Obesity: Postabsorptive modifiers of nutrient metabolism". Endocrine Reviews. 20 (6): 805–87. doi:10.1210/er.20.6.805. PMID 10605627.
- Cohen, Pieter A.; Bloszies, Clayton; Yee, Caleb; Gerona, Roy (March 2016). "An amphetamine isomer whose efficacy and safety in humans has never been studied, β-methylphenylethylamine (BMPEA), is found in multiple dietary supplements". Drug Testing and Analysis. 8 (3–4): 328–333. doi:10.1002/dta.1793. PMID 25847603. S2CID 205762285.
- Cohen, Pieter A.; Travis, John C.; Venhuis, Bastiaan J. (July 2014). "A methamphetamine analog ( -diethyl-phenylethylamine) identified in a mainstream dietary supplement". Drug Testing and Analysis. 6 (7–8): 805–807. doi:10.1002/dta.1578. PMID 24124092. S2CID 42232885.
- Cohen, Pieter A.; Avula, Bharathi; Venhuis, Bastiaan; Travis, John C.; Wang, Yan-Hong; Khan, Ikhlas A. (January 2017). "Pharmaceutical doses of the banned stimulant oxilofrine found in dietary supplements sold in the USA". Drug Testing and Analysis. 9 (1): 135–142. doi:10.1002/dta.1976. PMID 27062112. S2CID 9463413.
- Cohen, Pieter A.; Wang, Yan-Hong; Maller, Gregory; DeSouza, Renan; Khan, Ikhlas A. (March 2016). "Pharmaceutical quantities of yohimbine found in dietary supplements in the USA". Drug Testing and Analysis. 8 (3–4): 357–369. doi:10.1002/dta.1849. PMID 26391406.
- Cohen, Pieter A.; Travis, John C.; Keizers, Peter H. J.; Boyer, Frederick E.; Venhuis, Bastiaan J. (6 September 2018). "The stimulant higenamine in weight loss and sports supplements". Clinical Toxicology. 57 (2): 125–130. doi:10.1080/15563650.2018.1497171. PMID 30188222. S2CID 52165506.
- Lau DC, Douketis JD, Morrison KM, Hramiak IM, Sharma AM, Ur E (April 2007). "2006 Canadian clinical practice guidelines on the management and prevention of obesity in adults and children [summary]". CMAJ. 176 (8): S1–13. doi:10.1503/cmaj.061409. PMC 1839777. PMID 17420481.
- Onakpoya IJ, Posadzki PP, Watson LK, Davies LA, Ernst E (March 2012). "The efficacy of long-term conjugated linoleic acid (CLA) supplementation on body composition in overweight and obese individuals: a systematic review and meta-analysis of randomized clinical trials". Eur J Nutr (Systematic review). 51 (2): 127–34. doi:10.1007/s00394-011-0253-9. PMID 21990002. S2CID 39625058.
- Shekelle PG, Hardy ML, Morton SC, et al. (March 2003). "Efficacy and safety of ephedra and ephedrine for weight loss and athletic performance: a meta-analysis". JAMA. 289 (12): 1537–45. doi:10.1001/jama.289.12.1470. PMID 12672771.
- Bachorik, Lawrence. "FDA Announces Withdrawal Fenfluramine and Dexfenfluramine (Fen-Phen)". U.S. Food and Drug Administration. FDA. Retrieved 27 January 2014.
- , Launceston, Tasmania, Australia, Examiner, Friday, 21 January 1938, p 14, which states in postscript "However, the sex which for many years injured its health by tight lacing is not likely to be deterred from slimming by such considerations, The dictates of fashion will be paramount." Sir Arthur was particularly concerned with the neurological side effects of the then popular practice of dosing with thyroid extract to lose weight and, also, use of the then much vaunted weight loss medication dinitrophenol, which his report found killed as many patients as it reduced in girth, as well as, the compromise of the malnourished person's immune system and their consequent, often, inability to resist infectious diseases like the then endemic tuberculosis (archaic "epidemics of consumption").
- See, also, , Sidney Morning Herald, 17 Nov. 1937, p 10.
- Johnson, Kimball. "Alli: A Weight Loss Drug". WebMD. Retrieved 27 January 2014.
- "acarbose - oral, Precose". MedicineNet. Retrieved 27 January 2014.
- "Peptimmune homepage". peptimmune.com. Archived from the original on 23 March 2018. Retrieved 30 September 2019.
- McBride, Ryan. "Genzyme-Spinout Peptimmune Files for Chapter 7 Liquidation". Xconomy Boston. Retrieved 5 September 2011.
Further reading
Boss, Olivier; Karl G. Hofbauer (2004). Pharmacotherapy of obesity: options and alternatives. Boca Raton: CRC Press. ISBN 978-0-415-30321-7.
External links
 Media related to Anti-obesity medication at Wikimedia Commons
Media related to Anti-obesity medication at Wikimedia Commons- Prescription Medications for the Treatment of Obesity