Finerenone
Finerenone (INN, USAN) (developmental code name BAY-94-8862) is a nonsteroidal antimineralocorticoid that is in phase III clinical trials for the treatment of chronic kidney disease in people with type II diabetes as of October 2015. It has less relative affinity to other steroid hormone receptors than currently available antimineralocorticoids such as eplerenone and spironolactone, which should result in fewer adverse effects like gynaecomastia, impotence, and low libido.[1][2]
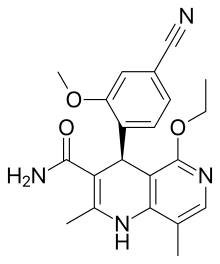 | |
| Clinical data | |
|---|---|
| Other names | BAY 94-8862 |
| Routes of administration | Oral |
| Drug class | Antimineralocorticoid |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.247.614 |
| Chemical and physical data | |
| Formula | C21H22N4O3 |
| Molar mass | 378.432 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Pharmacology
Finerenone blocks mineralocorticoid receptors, which makes it a potassium-sparing diuretic.
This table compares inhibitory (blocking) concentrations (IC50, unit: nM) of three antimineralocorticoids. Mineralocorticoid receptor inhibition is responsible for the desired action of the drugs, whereas inhibition of the other receptors potentially leads to side effects. Lower values mean stronger inhibition.[3]
| Spironolactone | Eplerenone | Finerenone | |
|---|---|---|---|
| Mineralocorticoid receptor | 24 | 990 | 18 |
| Glucocorticoid receptor | 2400 | 22,000 | >10,000 |
| Androgen receptor | 77 | 21,200 | >10,000 |
| Progesterone receptor | 740 | 31,200 | >10,000 |
The above-listed drugs have insignificant affinity for the estrogen receptor.
Finerenone acts as an antagonist to mineralocorticoid receptors harboring the S810L mutation, unlike other traditional MR inhibitors such as spironolactone and eplerenone that incidentally act as agonists.[4]
Chemistry
Unlike currently marketed antimineralocorticoids, finerenone is not a steroid but a dihydropyridine derivative.
Research
In the Phase II ARTS-DN study, finerenone dose-dependently reduced urine albumin to creatinine ratio in patients with diabetic kidney disease.[5] Based on these findings, finerenone is being studied in the large Phase III FIDELIO and FIGARO outcome studies designed to assess whether finerenone reduces risk of CKD progression and adverse cardiovascular events in patients with Chronic Kidney Disease and Type 2 Diabetes. These trials have enrolled more than 13,000 patients with primary completion of FIDELIO anticipated in 2020 and FIGARO IN 2021.[6][7]
References
- Ruilope LM, Tamargo J (April 2017). "Renin-angiotensin system blockade: Finerenone". Nephrologie & Therapeutique. 13 Suppl 1: S47–S53. doi:10.1016/j.nephro.2017.02.003. PMID 28577743.
- Pitt B, Anker SD, Böhm M, Gheorghiade M, Køber L, Krum H, et al. (February 2015). "Rationale and design of MinerAlocorticoid Receptor antagonist Tolerability Study-Heart Failure (ARTS-HF): a randomized study of finerenone vs. eplerenone in patients who have worsening chronic heart failure with diabetes and/or chronic kidney disease" (PDF). European Journal of Heart Failure. 17 (2): 224–32. doi:10.1002/ejhf.218. PMID 25678098. S2CID 205781715.
- Schubert-Zsilavecz M, Wurglics M (Fall 2015). "Finerenone". Neue Arzneimittel.
- Amazit L, Le Billan F, Kolkhof P, Lamribet K, Viengchareun S, Fay MR, et al. (September 2015). "Finerenone Impedes Aldosterone-dependent Nuclear Import of the Mineralocorticoid Receptor and Prevents Genomic Recruitment of Steroid Receptor Coactivator-1". The Journal of Biological Chemistry. 290 (36): 21876–89. doi:10.1074/jbc.M115.657957. PMC 4571943. PMID 26203193.
- Bakris GL, Agarwal R, Chan JC, Cooper ME, Gansevoort RT, Haller H, et al. (September 2015). "Effect of Finerenone on Albuminuria in Patients With Diabetic Nephropathy: A Randomized Clinical Trial". JAMA. 314 (9): 884–94. doi:10.1001/jama.2015.10081. PMID 26325557.
- Clinical trial number NCT02540993 for "Efficacy and Safety of Finerenone in Subjects With Type 2 Diabetes Mellitus and Diabetic Kidney Disease (FIDELIO-DKD)" at ClinicalTrials.gov
- Clinical trial number NCT02545049 for "Efficacy and Safety of Finerenone in Subjects With Type 2 Diabetes Mellitus and the Clinical Diagnosis of Diabetic Kidney Disease (FIGARO-DKD)" at ClinicalTrials.gov