Neuropathic pain
Neuropathic pain is pain caused by damage or disease affecting the somatosensory nervous system.[1] Neuropathic pain may be associated with abnormal sensations called dysesthesia or pain from normally non-painful stimuli (allodynia). It may have continuous and/or episodic (paroxysmal) components. The latter resemble stabbings or electric shocks. Common qualities include burning or coldness, "pins and needles" sensations, numbness and itching.
| Neuropathic pain | |
|---|---|
| Specialty | Neurology |
| Duration | Short to Long-Term |
| Causes | Damage to Nervous System such as Diabetes, Viruses, Injuries, Spinal Cord Injury, Stroke |
| Diagnostic method | Clinical History, Description of Pain |
| Differential diagnosis | Underlying cause of lesion may be related to multiple sclerosis, cancer, diabetes or may be sporadic |
| Medication | Anti-Convulsant/Anti-Depressant |
Up to 7%-8% of the European population is affected, and in 5% of persons it may be severe.[2][3] Neuropathic pain may result from disorders of the peripheral nervous system or the central nervous system (brain and spinal cord). Thus, neuropathic pain may be divided into peripheral neuropathic pain, central neuropathic pain, or mixed (peripheral and central) neuropathic pain. Neuropathic pain may occur in isolation or in combination with other forms of pain. Medical treatments focus on identifying the underlying cause and relieving pain. In cases of neuropathy, the pain may progress to insensitivity.
Diagnosis
Diagnosis of pain conditions relies on the character of the pain with a sharp stabbing character and the presence of particular features such as mechanical allodynia and cold allodynia. Neuropathic pain also tends to affect defined dermatomes and there may be limits to the area of pain. For neuropathic pain, clinicians look for an underlying lesion to the nervous system or an inciting cause consistent with the development of neuropathic pain. The obvious presence of an underlying feature or cause is not always detectable, and response to treatment may be used as a surrogate particularly in cases where diagnosis of the underlying lesion leaves the patient in pain for a prolonged period of time. MRI may be helpful in the identification of underlying lesions, reversible causes or serious underlying conditions such as primary presentation of a tumor or multiple sclerosis. Quantitative sensory testing (QST), a system of detailed analysis of the somatosensory system, is frequently used in research situations to identify neuropathic pain and a more detailed analysis of its components. It has been suggested by some authorities that QST may have a future role in the diagnosis of neuropathic pain and in particular the identification of neuropathic pain subtypes. Neuropathic pain can occur alone or in combination with other types of pain. The identification of neuropathic pain components is important as different classes of analgesic are required.[4] Difficulties in identifying subtypes of patients with chronic neuropathic pain underlie some of the difficulties in treatment including identifying suitable cohorts of patients for randomized clinical trials.[5]
Cause
Central neuropathic pain is found in spinal cord injury, multiple sclerosis,[6] and some strokes. Peripheral neuropathies are commonly caused by diabetes, metabolic disorders, herpes zoster infection, HIV-related neuropathies, nutritional deficiencies, toxins, remote manifestations of malignancies, immune mediated disorders and physical trauma to a nerve trunk.[7][8] Neuropathic pain is common in cancer as a direct result of cancer on peripheral nerves (e.g., compression by a tumor), or as a side effect of chemotherapy (chemotherapy-induced peripheral neuropathy),[9][10] radiation injury or surgery.
Comorbidities
Neuropathic pain has profound physiological effects on the brain which can manifest as psychological disorders. Rodent models where the social effects of chronic pain can be isolated from other factors suggest that induction of chronic pain can cause anxio-depressive symptoms and that particular circuits in the brain have a direct connection.[11][12] Depression and neuropathic pain may have a bidirectional relationship and relief of co-morbid depression may underlie some of the therapeutic efficacy of antidepressants in neuropathic pain. Neuropathic pain has important effects on social well-being that should not be ignored. Neuropathic pain sufferers may have difficulty working exhibiting higher levels of presenteeism, absenteeism and unemployment,[13] exhibit higher levels of substance misuse (which may be related to attempted self-medication),[14][15] and present difficulties with social interactions.[16] Moreover uncontrolled neuropathic pain is a significant risk factor for suicide.[17] Certain classes of neuropathic pain may cause serious adverse effects necessitating hospital admission, for instance trigeminal neuralgia can present as a severe crisis where the patient may have difficulty talking, eating and drinking.[18] As neuropathic pain may be comorbid with cancer, it can have important dose limiting effects on certain classes of chemotherapeutic.[19]
Treatments
Neuropathic pain can be very difficult to treat with only some 40-60% of people achieving partial relief.[20]
General approach
First line treatments are certain antidepressants (tricyclic antidepressants and serotonin–norepinephrine reuptake inhibitors), anticonvulsants (pregabalin and gabapentin).[21][22] Opioid analgesics are recognized as useful agents but are not recommended as first line treatments.[22] A broader range of treatments are used in specialist care. There are limited data and guidance for the long-term treatment of pain. Notably, strong evidence from randomized controlled trials is not available for all interventions.
Anticonvulsants
Pregabalin and gabapentin may reduce pain associated with diabetic neuropathy.[23][21][24][25] The anticonvulsants carbamazepine and oxcarbazepine are especially effective in trigeminal neuralgia. Carbamazepine is a voltage-gated sodium channel inhibitor, and reduces neuronal excitability by preventing depolarisation.[26] Carbamazepine is most commonly prescribed to treat trigeminal neuralgia due to clinical experience and early clinical trials showing strong efficacy. Gabapentin may reduce symptoms associated with neuropathic pain or fibromyalgia in some people.[21] There is no predictor test to determine if it will be effective for a particular person. A short trial period of gabapentin therapy is recommended, to determine the effectiveness for that person. 62% of people taking gabapentin may have at least one adverse event, however the incidence of serious adverse events was found to be low.[21] Although gabapentin and pregabalin possess low abuse potential, these drugs can cause physical dependence over the course of normal treatment, and certain patients may become psychologically dependent as well.
Meta analysis of randomized clinical trials suggests that Lamotrigine is not useful for the majority of patients although it may have use in treatment refractory cases.[27]
Antidepressants
Dual serotonin-norepinephrine reuptake inhibitors in particular duloxetine, as well as tricyclic antidepressants in particular amitriptyline, and nortriptyline are considered first-line medications for this condition.[22]
Opioids
Opioids, while commonly used in chronic neuropathic pain, are not a recommended first or second line treatment.[28] In the short and long term they are of unclear benefit, although clinical experience suggests that opioids like tramadol may be useful for treating sudden onset severe pain[29] In the intermediate term evidence of low quality supports utility.[29]
Several opioids, particularly levorphanol, methadone and ketobemidone, possess NMDA receptor antagonism in addition to their µ-opioid agonist properties. Methadone does so because it is a racemic mixture; only the l-isomer is a potent µ-opioid agonist. The d-isomer does not have opioid agonist action and acts as an NMDA receptor antagonist; d-methadone is analgesic in experimental models of chronic pain.[30]
There is little evidence to indicate that one strong opioid is more effective than another. Expert opinion leans toward the use of methadone for neuropathic pain, in part because of its NMDA antagonism. It is reasonable to base the choice of opioid on other factors.[31] It is unclear if fentanyl gives pain relief to people with neuropathic pain.[32] The potential pain relief benefits of strong opioids must be weighed against their significant addiction potential under normal clinical use and some authorities suggest that they should be reserved for cancer pain.[33] Importantly, recent observational studies suggest a pain-relief benefit in non-cancer related chronic pain of reducing or terminating long-term opioid therapy.[34][35]
Non-Pharmaceutical Interventions
Non-pharmaceutical treatments such as exercise, physical therapy and psychotherapy may be useful adjuncts to treatment.[36]
Botulinum toxin type A
Local intradermal injection of botulinum neurotoxin type A may be helpful in chronic focal painful neuropathies. However, it causes muscle paralysis which may impact quality of life.[37]
Cannabinoids
Evidence for the use of Cannabis based medicines is limited, low-moderate quality evidence suggests a benefit in pain intensity, sleep quality and psychological distress. The use of Cannabis has to be weighed against the negative psychotomimetic effects that may impact quality of life.[38][39] Therefore cannabis is not recommended as a mainstream treatment but may have a place for treatment refractory cases.
Neuromodulators
Neuromodulation is a field of science, medicine and bioengineering that encompasses both implantable and non-implantable technologies (electrical and chemical) for treatment purposes.[40]
Implanted devices are expensive and carry the risk of complications. Available studies have focused on conditions having a different prevalence than neuropathic pain patients in general. More research is needed to define the range of conditions that they might benefit.
Deep brain stimulation
The best long-term results with deep brain stimulation have been reported with targets in the periventricular/periaqueductal grey matter (79%), or the periventricular/periaqueductal grey matter plus thalamus and/or internal capsule (87%).[41] There is a significant complication rate, which increases over time.[42]
Motor cortex stimulation
Stimulation of the primary motor cortex through electrodes placed within the skull but outside the thick meningeal membrane (dura) has been used to treat pain. The level of stimulation is below that for motor stimulation. As compared with spinal stimulation, which is associated with noticeable tingling (paresthesia) at treatment levels, the only palpable effect is pain relief.[43][44]
Spinal cord stimulators implanted spinal pumps
Spinal cord stimulators use electrodes placed adjacent to but outside the spinal cord. The overall complication rate is one-third, most commonly due to lead migration or breakage but advancements in the past decade have driven complication rates much lower. Lack of pain relief occasionally prompts device removal.[45]
NMDA antagonism
The N-methyl-D-aspartate (NMDA) receptor seems to play a major role in neuropathic pain and in the development of opioid tolerance. Dextromethorphan is an NMDA antagonist at high doses. Experiments in both animals and humans have established that NMDA antagonists such as ketamine and dextromethorphan can alleviate neuropathic pain and reverse opioid tolerance.[46] Unfortunately, only a few NMDA antagonists are clinically available and their use is limited by a very short half life (ketamine), weak activity (memantine) or unacceptable side effects (dextromethorpan).
Intrathecal drug delivery
Intrathecal pumps deliver medication to the fluid filled (subarachnoid) space surrounding the spinal cord. Opioids alone or opioids with adjunctive medication (either a local anesthetic or clonidine) Rarely there are complications such as serious infection (meningitis), urinary retention, hormonal disturbance and intrathecal granuloma formation have been noted with intrathecal infusion, associated with the delivery method.
Conotoxins
Ziconotide is a voltage-gated calcium channel blocker which may be used in severe cases of ongoing neuropathic pain[47] it is delivered intrathecally.
Topical agents
In some forms of neuropathy the topical application of local anesthetics such as lidocaine may provide relief. A transdermal patch containing lidocaine is available commercially in some countries.
Repeated topical applications of capsaicin are followed by a prolonged period of reduced skin sensibility referred to as desensitization, or nociceptor inactivation. Capsaicin causes reversible degeneration of epidermal nerve fibers.[48] Notably the capsaicin used for the relief of neuropathic pain is a substantially higher concentration than capsaicin creams available over the counter, there is no evidence that over the counter capsaicin cream can improve neuropathic pain[49] and topical capsaicin can itself induce pain.[48]
Surgical Interventions
Orthopaedic interventions are frequently used to correct underlying pathology which may contribute to neuropathic pain. Many orthopaedic procedures have more limited evidence. Historically, neurosurgeons have attempted lesions of regions of the brain, spinal cord and peripheral nervous system. Whilst they cause some short term analgesia, these are considered to be universally ineffective.
Herbal products
There is no good evidence that herbal products (nutmeg or St John's wort) are useful for treating neuropathic pain.[50]
Dietary supplements
A 2007 review of studies found that injected (parenteral) administration of alpha lipoic acid (ALA) was found to reduce the various symptoms of peripheral diabetic neuropathy.[51] While some studies on orally administered ALA had suggested a reduction in both the positive symptoms of diabetic neuropathy (dysesthesia including stabbing and burning pain) as well as neuropathic deficits (paresthesia),[52] the meta-analysis showed "more conflicting data whether it improves sensory symptoms or just neuropathic deficits alone".[51] There is some limited evidence that ALA is also helpful in some other non-diabetic neuropathies.[53]
Benfotiamine is an oral prodrug of Vitamin B1 that has several placebo-controlled double-blind trials proving efficacy in treating neuropathy and various other diabetic comorbidities.[54][55]
History
The history of pain management can be traced back to ancient times. Galen also suggested nerve tissue as the transferring route of pain to the brain through the invisible psychic pneuma.[56] The idea of origination of pain from the nerve itself, without any exciting pathology in other organs is presented by medieval medical scholars such as Rhazes, Haly Abbas and Avicenna. They named this type of pain specifically as "vaja al asab" [nerve originated pain], described its numbness, tingling and needling quality, discussed its etiology and the differentiating characteristics.[57] The description of neuralgia was made by John Fothergill (1712-1780). In a medical article entitled “Clinical Lecture on Lead Neuropathy" published in 1924 the word "Neuropathy" was used for the first time by Gordon.[58]
Proposed mechanistic basis for neuropathic pain
The underlying pathophysiology of neuropathic pain remains a contested topic. The etiology and mechanism of pain are related to the cause of the pain. Certain forms of neuropathic pain are associated with lesions to the central nervous system such as thalamic pain associated with certain lesions (for instance strokes) to the thalamus[59] whereas other forms of pain have a peripheral inciting injury such as traumatic neuropathies. The inciting cause of neuropathy has important consequences for its mechanistic basis as different tissues and cells are involved. The mechanistic basis of neuropathic pain remains controversial as do the relative contributions of each pathway. Notably our understanding of these processes is largely driven by rodent models in part because studying these tissues in living adults is difficult.
Peripheral
With peripheral nervous system lesions, a number of processes may occur. Intact neurons may become unusually sensitive and develop spontaneous pathological activity and abnormal excitability.
During neuropathic pain, ectopic activity arises in the peripheral nociceptors and this appears to be due in part to changes in the ion channel expression at the level of the periphery. There may be an increase in the expression or activity of voltage gated sodium and calcium channels which will support action potential generation. There may also be a decrease in potassium channels which would normally oppose action potential generation. Each of these changes appears to support an increase in excitability, which may allow endogenous stimuli to cause spontaneous pain.[60]
Central
Central mechanisms of neuropathic pain involve a number of major pathways. Nociception is ordinarily transduced by a polysynaptic pathway through the spinal cord, and up the spinothalamic tract to the thalamus and then the cortex. Broadly speaking in neuropathic pain, neurons are hypersensitized, glia become activated and there is a loss of inhibitory tone.
Pain gates
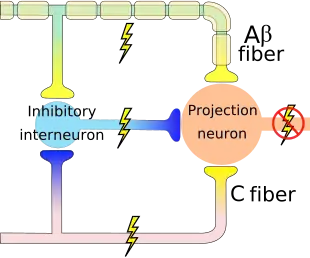
A major hypothesis in the theory of pain perception is the hypothesized pain gates predicted by Wall and Melzack's Gate Control Theory. The theory predicts that the activation of central pain inhibitory neurons by non-pain sensing neurons prevents the transmission of non-harmful stimuli to pain centers in the brain. A loss of inhibitory neurons, GAD65/67 expression (the enzymes that synthesise GABA; the predominant inhibitory transmitter in the adult brain), has been observed in some systems following peripheral neuropathy such as in rats, and mice.[61] Although, these observations remain controversial with some investigators unable to detect a change. The loss of inhibitory inputs may allow fibers to transmit messages via the spinothalamic tract thus causing pain in normally painless stimuli. This loss of inhibition may not be limited to the spinal cord and a loss of GABA has also been observed in chronic pain patients in the thalamus.[62]
Glia

During neuropathic pain, glia become "activated" leading to the release of proteins that modulate neural activity. The activation of glia remains an area of intense interest for researchers. Microglia, the brain and spinal cord resident immune cells, respond to extrinsic cues. The source of these cues may include neurons secreting chemokines such as CCL21 and surface immobilized chemokines such as CX3CL1. Other glia such as astrocytes and oligodendrocytes may also release these extrinsic cues for microglia and microglia themselves may produce proteins that amplify the response.[63] The effect of microglia on neurons that leads to the neurons being sensitized is controversial. Brain derived neurotrophic factor, prostaglandins, TNF and IL-1β may be produced by microglia and cause changes in neurons that lead to hyperexcitability.[64][65]
Central sensitization
Central sensitization is a potential component of neuropathic pain. It refers to a change in synaptic plasticity, efficacy, and intrinsic disinhibition that leads to an uncoupling of noxious inputs. In the sensitized neuron, outputs are no longer coupled to the intensity or duration and many inputs may be combined together.[66]
Circuit Potentiation
During high frequency stimulation synapses conveying nociceptive information may become hyper efficient in a process that is similar although not identical to long-term potentiation.[67] Molecules such as substance P may be involved in potentiation via neurokinin receptors. NMDA activation also triggers a change in the post synapse, it activates receptor kinases that increase receptor trafficking and post-translationally modify receptors causing changes in their excitability.[66]
Cellular
The phenomena described above are dependent on changes at the cellular and molecular levels. Altered expression of ion channels, changes in neurotransmitters and their receptors, as well as altered gene expression in response to neural input, are at play.[68] Neuropathic pain is associated with changes in sodium and calcium channel subunit expression resulting in functional changes. In chronic nerve injury, there is redistribution and alteration of subunit compositions of sodium and calcium channels resulting in spontaneous firing at ectopic sites along the sensory pathway.[8]
See also
References
- "Taxonomy". International Association for the Study of pain. Archived from the original on 13 January 2015. Retrieved 3 May 2015.
- Torrance N, Smith BH, Bennett MI, Lee AJ (April 2006). "The epidemiology of chronic pain of predominantly neuropathic origin. Results from a general population survey". J Pain. 7 (4): 281–9. doi:10.1016/j.jpain.2005.11.008. PMID 16618472.
- Bouhassira D, Lantéri-Minet M, Attal N, Laurent B, Touboul C (June 2008). "Prevalence of chronic pain with neuropathic characteristics in the general population". Pain. 136 (3): 380–7. doi:10.1016/j.pain.2007.08.013. PMID 17888574. S2CID 599627.
- Freynhagen, R.; Bennett, M. I. (2009-08-12). "Diagnosis and management of neuropathic pain". BMJ. 339: b3002. doi:10.1136/bmj.b3002. ISSN 0959-8138. PMID 19675082. S2CID 20690119.
- Themistocleous, Andreas C.; Crombez, Geert; Baskozos, Georgios; Bennett, David L. (September 2018). "Using stratified medicine to understand, diagnose and treat neuropathic pain". Pain. 159 (Suppl 1): S31–S42. doi:10.1097/j.pain.0000000000001301. ISSN 0304-3959. PMC 6130809. PMID 30113945.
- Foley P, Vesterinen H, Laird B, et al. (2013). "Prevalence and natural history of pain in adults with multiple sclerosis: Systematic review and meta-analysis". Pain. 154 (5): 632–42. doi:10.1016/j.pain.2012.12.002. PMID 23318126. S2CID 25807525.
- Portenoy RK (1989). "Painful polyneuropathy". Neurol Clin. 7 (2): 265–88. doi:10.1016/S0733-8619(18)30813-2. PMID 2566901.
- Vaillancourt PD, Langevin HM (1999). "Painful peripheral neuropathies". Med. Clin. North Am. 83 (3): 627–42, vi. doi:10.1016/S0025-7125(05)70127-9. PMID 10386118.
- Chemotherapy-induced Peripheral Neuropathy Fact Sheet, Retrieved on 29 December 2008
- Archived 2009-07-08 at the Wayback Machine Cancerbackup, Macmillan Cancer Support, Peripheral neuropathy, Retrieved on 29 December 2008
- Hooten, W. Michael (July 2016). "Chronic Pain and Mental Health Disorders: Shared Neural Mechanisms, Epidemiology, and Treatment". Mayo Clinic Proceedings. 91 (7): 955–970. doi:10.1016/j.mayocp.2016.04.029. ISSN 1942-5546. PMID 27344405.
- Bravo, Lidia; Llorca-Torralba, Meritxell; Suárez-Pereira, Irene; Berrocoso, Esther (2020-08-01). "Pain in neuropsychiatry: Insights from animal models". Neuroscience & Biobehavioral Reviews. 115: 96–115. doi:10.1016/j.neubiorev.2020.04.029. ISSN 0149-7634. PMID 32437745. S2CID 218580755.
- Schaefer, Caroline; Sadosky, Alesia; Mann, Rachael; Daniel, Shoshana; Parsons, Bruce; Tuchman, Michael; Anschel, Alan; Stacey, Brett R; Nalamachu, Srinivas; Nieshoff, Edward (2014-10-29). "Pain severity and the economic burden of neuropathic pain in the United States: BEAT Neuropathic Pain Observational Study". ClinicoEconomics and Outcomes Research. 6: 483–496. doi:10.2147/CEOR.S63323. ISSN 1178-6981. PMC 4218900. PMID 25378940.
- M, Egli; Gf, Koob; S, Edwards (November 2012). "Alcohol Dependence as a Chronic Pain Disorder". Neuroscience and Biobehavioral Reviews. 36 (10): 2179–92. doi:10.1016/j.neubiorev.2012.07.010. PMC 3612891. PMID 22975446.
- Alford, Daniel P.; German, Jacqueline S.; Samet, Jeffrey H.; Cheng, Debbie M.; Lloyd-Travaglini, Christine A.; Saitz, Richard (2016-05-01). "Primary Care Patients with Drug Use Report Chronic Pain and Self-Medicate with Alcohol and Other Drugs". Journal of General Internal Medicine. 31 (5): 486–491. doi:10.1007/s11606-016-3586-5. ISSN 1525-1497. PMC 4835374. PMID 26809204.
- Dueñas, María; Ojeda, Begoña; Salazar, Alejandro; Mico, Juan Antonio; Failde, Inmaculada (2016-06-28). "A review of chronic pain impact on patients, their social environment and the health care system". Journal of Pain Research. 9: 457–467. doi:10.2147/JPR.S105892. ISSN 1178-7090. PMC 4935027. PMID 27418853.
- Petrosky, Emiko; Harpaz, Rafael; Fowler, Katherine A.; Bohm, Michele K.; Helmick, Charles G.; Yuan, Keming; Betz, Carter J. (2018-09-11). "Chronic Pain Among Suicide Decedents, 2003 to 2014: Findings From the National Violent Death Reporting System". Annals of Internal Medicine. 169 (7): 448–455. doi:10.7326/M18-0830. ISSN 0003-4819. PMC 6913029. PMID 30208405.
- Bennetto, Luke; Patel, Nikunj K.; Fuller, Geraint (2007-01-27). "Trigeminal neuralgia and its management". BMJ (Clinical Research Ed.). 334 (7586): 201–205. doi:10.1136/bmj.39085.614792.BE. ISSN 1756-1833. PMC 1782012. PMID 17255614.
- Staff, Nathan P.; Grisold, Anna; Grisold, Wolfgang; Windebank, Anthony J. (June 2017). "Chemotherapy-Induced Peripheral Neuropathy: A Current Review". Annals of Neurology. 81 (6): 772–781. doi:10.1002/ana.24951. ISSN 0364-5134. PMC 5656281. PMID 28486769.
- Dworkin RH, O'Connor AB, Backonja M, et al. (2007). "Pharmacologic management of neuropathic pain: evidence-based recommendations". Pain. 132 (3): 237–51. doi:10.1016/j.pain.2007.08.033. PMID 17920770. S2CID 9333155.
- Wiffen, PJ; Derry, S; Bell, RF; Rice, AS; Tölle, TR; Phillips, T; Moore, RA (9 June 2017). "Gabapentin for chronic neuropathic pain in adults". The Cochrane Database of Systematic Reviews. 6: CD007938. doi:10.1002/14651858.CD007938.pub4. PMC 6452908. PMID 28597471.
- Dworkin, RH; O'Connor, AB; Audette, J; Baron, R; Gourlay, GK; Haanpää, ML; Kent, JL; Krane, EJ; Lebel, AA; Levy, RM; Mackey, SC; Mayer, J; Miaskowski, C; Raja, SN; Rice, AS; Schmader, KE; Stacey, B; Stanos, S; Treede, RD; Turk, DC; Walco, GA; Wells, CD (Mar 2010). "Recommendations for the pharmacological management of neuropathic pain: an overview and literature update". Mayo Clinic Proceedings. 85 (3 Suppl): S3-14. doi:10.4065/mcp.2009.0649. PMC 2844007. PMID 20194146.
- Moore, R. Andrew; Straube, Sebastian; Wiffen, Philip J.; Derry, Sheena; McQuay, Henry J. (2009-07-08). "Pregabalin for acute and chronic pain in adults". The Cochrane Database of Systematic Reviews (3): CD007076. doi:10.1002/14651858.CD007076.pub2. ISSN 1469-493X. PMC 4167351. PMID 19588419.
- "Pharmacological treatment for all neuropathic pain except trigeminal neuralgia". NICE Guidance CG173. 2013.
- "Neuropathic pain in adults: pharmacological management in non-specialist settings | Guidance and guidelines | NICE". www.nice.org.uk.
- Al-Quliti, Khalid W. (April 2015). "Update on neuropathic pain treatment for trigeminal neuralgia". Neurosciences. 20 (2): 107–114. doi:10.17712/nsj.2015.2.20140501. ISSN 1319-6138. PMC 4727618. PMID 25864062.
- Wiffen, PJ; Derry, S; Moore, RA (December 3, 2013). "Lamotrigine for chronic neuropathic pain and fibromyalgia in adults". The Cochrane Database of Systematic Reviews. 12 (12): CD006044. doi:10.1002/14651858.CD006044.pub4. PMC 6485508. PMID 24297457.
- Dowell, D; Haegerich, TM; Chou, R (18 March 2016). "CDC Guideline for Prescribing Opioids for Chronic Pain - United States, 2016". MMWR. Recommendations and Reports. 65 (1): 1–49. doi:10.15585/mmwr.rr6501e1. PMID 26987082.
Several guidelines agree that first- and second-line drugs for neuropathic pain include anticonvulsants (gabapentin or pregabalin), tricyclic antidepressants, and SNRIs
- McNicol, ED; Midbari, A; Eisenberg, E (29 August 2013). "Opioids for neuropathic pain". The Cochrane Database of Systematic Reviews (8): CD006146. doi:10.1002/14651858.CD006146.pub2. PMC 6353125. PMID 23986501.
- Davis AM, Inturrisi CE (1999). "d-Methadone blocks morphine tolerance and N-methyl-D-aspartate-induced hyperalgesia". J. Pharmacol. Exp. Ther. 289 (2): 1048–53. PMID 10215686.
- Bruera E, Palmer JL, Bosnjak S, et al. (2004). "Methadone versus morphine as a first-line strong opioid for cancer pain: a randomized, double-blind study". J. Clin. Oncol. 22 (1): 185–92. doi:10.1200/JCO.2004.03.172. PMID 14701781.
- Derry, Sheena; Stannard, Cathy; Cole, Peter; Wiffen, Philip J.; Knaggs, Roger; Aldington, Dominic; Moore, R. Andrew (2016-10-11). "Fentanyl for neuropathic pain in adults". The Cochrane Database of Systematic Reviews. 10: CD011605. doi:10.1002/14651858.CD011605.pub2. PMC 6457928. PMID 27727431.
- Volkow, Nora D.; McLellan, A. Thomas (2016-03-30). "Opioid Abuse in Chronic Pain — Misconceptions and Mitigation Strategies". New England Journal of Medicine. New England Journal of Medicine 374:1253-1263. 374 (13): 1253–1263. doi:10.1056/nejmra1507771. PMID 27028915.
- Nicholas, Michael K.; Asghari, Ali; Sharpe, Louise; Beeston, Lee; Brooker, Charles; Glare, Paul; Martin, Rebecca; Molloy, Allan; Wrigley, Paul J. (March 2020). "Reducing the use of opioids by patients with chronic pain: an effectiveness study with long-term follow-up". PAIN. 161 (3): 509–519. doi:10.1097/j.pain.0000000000001763. ISSN 0304-3959. PMID 31764391.
- McPherson, Sterling; Lederhos Smith, Crystal; Dobscha, Steven K.; Morasco, Benjamin J.; Demidenko, Michael I.; Meath, Thomas H. A.; Lovejoy, Travis I. (October 2018). "Changes in pain intensity after discontinuation of long-term opioid therapy for chronic noncancer pain". PAIN. 159 (10): 2097–2104. doi:10.1097/j.pain.0000000000001315. ISSN 0304-3959. PMC 6993952. PMID 29905648.
- Colloca, Luana; Ludman, Taylor; Bouhassira, Didier; Baron, Ralf; Dickenson, Anthony H.; Yarnitsky, David; Freeman, Roy; Truini, Andrea; Attal, Nadine; Finnerup, Nanna B.; Eccleston, Christopher (2017-02-16). "Neuropathic pain". Nature Reviews. Disease Primers. 3: 17002. doi:10.1038/nrdp.2017.2. ISSN 2056-676X. PMC 5371025. PMID 28205574.
- Mittal, SO; Safarpour, D; Jabbari, B (February 2016). "Botulinum Toxin Treatment of Neuropathic Pain". Seminars in Neurology. 36 (1): 73–83. doi:10.1055/s-0036-1571953. PMID 26866499.
- Mason O1, Morgan CJ, Dhiman SK, Patel A, Parti N, Patel A, Curran HV (19 November 2008). "Acute cannabis use causes increased psychotomimetic experiences in individuals prone to psychosis". Psychological Medicine (journal). 39 (6): 951–956. doi:10.1017/S0033291708004741. PMID 19017430.CS1 maint: uses authors parameter (link)
- Mücke M, Phillips T, Radbruch L, Petzke F, Häuser W (March 2018). "Cannabis-based medicines for chronic neuropathic pain in adults". Cochrane Database Syst Rev (Systematic review). 3: CD012182. doi:10.1002/14651858.CD012182.pub2. PMC 6494210. PMID 29513392.
- Krames ES (2006). "Neuromodulatory devices are part of our "Tools of the Trade"". Pain Med. 7: S3–5. doi:10.1111/j.1526-4637.2006.00116.x.
- Bittar RG, Kar-Purkayastha I, Owen SL, et al. (2005). "Deep brain stimulation for pain relief: a meta-analysis". J Clin Neurosci. 12 (5): 515–9. doi:10.1016/j.jocn.2004.10.005. PMID 15993077. S2CID 24246117.
- Oh MY, Abosch A, Kim SH, Lang AE, Lozano AM (2002). "Long-term hardware-related complications of deep brain stimulation". Neurosurgery. 50 (6): 1268–74, discussion 1274–6. doi:10.1097/00006123-200206000-00017. PMID 12015845. S2CID 36904630.
- Brown JA, Pilitsis JG. Motor Cortex Stimulation Pain Medicine 2006; 7:S140.
- Osenbach, R. Neurostimulation for the Treatment of Intractable Facial Pain Pain Medicine 2006; 7:S126
- Turner JA, Loeser JD, Deyo RA, Sanders SB (2004). "Spinal cord stimulation for patients with failed back surgery syndrome or complex regional pain syndrome: a systematic review of effectiveness and complications". Pain. 108 (1–2): 137–47. doi:10.1016/j.pain.2003.12.016. PMID 15109517. S2CID 16722691.
- Nelson KA, Park KM, Robinovitz E, Tsigos C, Max MB (1997). "High-dose oral dextromethorphan versus placebo in painful diabetic neuropathy and postherpetic neuralgia". Neurology. 48 (5): 1212–8. doi:10.1212/WNL.48.5.1212. PMID 9153445. S2CID 25663595.
- Lynch SS, Cheng CM, Yee JL (2006). "Intrathecal ziconotide for refractory chronic pain". Ann Pharmacother. 40 (7–8): 1293–300. doi:10.1345/aph.1G584. PMID 16849624. S2CID 1159955.
- Nolano M, Simone DA, Wendelschafer-Crabb G, Johnson T, Hazen E, Kennedy WR (1999). "Topical capsaicin in humans: parallel loss of epidermal nerve fibers and pain sensation". Pain. 81 (1–2): 135–45. doi:10.1016/S0304-3959(99)00007-X. PMID 10353501. S2CID 8952555.
- Derry, Sheena; Moore, R. Andrew (2012). "Topical capsaicin (low concentration) for chronic neuropathic pain in adults". Cochrane Database of Systematic Reviews (9): CD010111. doi:10.1002/14651858.CD010111. ISSN 1465-1858. PMC 6540838. PMID 22972149.
- Boyd A, Bleakley C, Hurley DA, Gill C, Hannon-Fletcher M, Bell P, McDonough S (April 2019). "Herbal medicinal products or preparations for neuropathic pain". Cochrane Database Syst Rev (Systematic review). 4: CD010528. doi:10.1002/14651858.CD010528.pub4. PMC 6445324. PMID 30938843.
- Foster TS (2007). "Efficacy and safety of alpha-lipoic acid supplementation in the treatment of symptomatic diabetic neuropathy". Diabetes Educ. 33 (1): 111–7. doi:10.1177/0145721706297450. PMID 17272797. S2CID 22801230.
ALA appears to improve neuropathic symptoms and deficits when administered via parenteral supplementation over a 3-week period. Oral treatment with ALA appears to have more conflicting data whether it improves sensory symptoms or just neuropathic deficits alone.
- Ziegler D, Ametov A, Barinov A, et al. (2006). "Oral treatment with alpha-lipoic acid improves symptomatic diabetic polyneuropathy: the SYDNEY 2 trial". Diabetes Care. 29 (11): 2365–70. doi:10.2337/dc06-1216. PMID 17065669.
- Patton LL, Siegel MA, Benoliel R, De Laat A (2007). "Management of burning mouth syndrome: systematic review and management recommendations". Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 103 Suppl: S39.e1–13. doi:10.1016/j.tripleo.2006.11.009. PMID 17379153.
- Stracke H, Lindemann A, Federlin K (1996). "A benfotiamine-vitamin B combination in treatment of diabetic polyneuropathy". Exp. Clin. Endocrinol. Diabetes. 104 (4): 311–6. doi:10.1055/s-0029-1211460. PMID 8886748.
- Thornalley PJ (2005). "The potential role of thiamine (vitamin B(1)) in diabetic complications". Curr Diabetes Rev. 1 (3): 287–98. doi:10.2174/157339905774574383. PMID 18220605.
- Freemon, Frank R. (1994). "Galen's ideas on neurological function". J Hist Neurosci. 3 (4): 263–71. doi:10.1080/09647049409525619. PMID 11618827.
- Heydari, M.; Shams, M.; Hashempur, M. H.; Zargaran, A.; Dalfardi, B.; Borhani-Haghighi, A. (2015). "The origin of the concept of neuropathic pain in Early Medieval Persia (9th - 12th century CE)". Acta Med Hist Adriat. 13 Suppl 1: 9–22. PMID 26966748.
- Scadding, JW (March 2004). "Treatment of neuropathic pain: historical aspects". Pain Medicine. 5 Suppl 1: S3-8. doi:10.1111/j.1526-4637.2004.04018.x. PMID 14996226.
- Gritsch, Simon; Bali, Kiran Kumar; Kuner, Rohini; Vardeh, Daniel (2016-03-08). "Functional characterization of a mouse model for central post-stroke pain". Molecular Pain. 12. doi:10.1177/1744806916629049. ISSN 1744-8069. PMC 4956143. PMID 27030713.
- Costigan, Michael; Scholz, Joachim; Woolf, Clifford J. (2009). "Neuropathic Pain". Annual Review of Neuroscience. 32: 1–32. doi:10.1146/annurev.neuro.051508.135531. ISSN 0147-006X. PMC 2768555. PMID 19400724.
- Manion, John; Waller, Matthew A.; Clark, Teleri; Massingham, Joshua N.; Neely, G. Gregory (2019). "Developing Modern Pain Therapies". Frontiers in Neuroscience. 13: 1370. doi:10.3389/fnins.2019.01370. ISSN 1662-453X. PMC 6933609. PMID 31920521.
- Henderson, Luke A.; Peck, Chris C.; Petersen, Esben T.; Rae, Caroline D.; Youssef, Andrew M.; Reeves, Jenna M.; Wilcox, Sophie L.; Akhter, Rahena; Murray, Greg M.; Gustin, Sylvia M. (2013-04-24). "Chronic Pain: Lost Inhibition?". Journal of Neuroscience. 33 (17): 7574–7582. doi:10.1523/JNEUROSCI.0174-13.2013. ISSN 0270-6474. PMC 6619566. PMID 23616562.
- Ji, Ru-Rong; Donnelly, Christopher R.; Nedergaard, Maiken (November 2019). "Astrocytes in chronic pain and itch". Nature Reviews. Neuroscience. 20 (11): 667–685. doi:10.1038/s41583-019-0218-1. ISSN 1471-003X. PMC 6874831. PMID 31537912.
- Rr, Ji; A, Chamessian; Yq, Zhang (2016-11-04). "Pain Regulation by Non-Neuronal Cells and Inflammation". Science. 354 (6312): 572–577. Bibcode:2016Sci...354..572J. doi:10.1126/science.aaf8924. PMC 5488328. PMID 27811267.
- Siniscalco, Dario; Giordano, Catia; Rossi, Francesco; Maione, Sabatino; de Novellis, Vito (December 2011). "Role of Neurotrophins in Neuropathic Pain". Current Neuropharmacology. 9 (4): 523–529. doi:10.2174/157015911798376208. ISSN 1570-159X. PMC 3263449. PMID 22654713.
- Latremoliere, Alban; Woolf, Clifford J. (September 2009). "Central Sensitization: A Generator of Pain Hypersensitivity by Central Neural Plasticity". The Journal of Pain. 10 (9): 895–926. doi:10.1016/j.jpain.2009.06.012. ISSN 1526-5900. PMC 2750819. PMID 19712899.
- Rr, Ji; T, Kohno; Ka, Moore; Cj, Woolf (December 2003). "Central Sensitization and LTP: Do Pain and Memory Share Similar Mechanisms?". Trends in Neurosciences. 26 (12): 696–705. doi:10.1016/j.tins.2003.09.017. PMID 14624855. S2CID 14214986.
- Truini A, Cruccu G (May 2006). "Pathophysiological mechanisms of neuropathic pain". Neurol. Sci. 27 Suppl 2: S179–82. doi:10.1007/s10072-006-0597-8. PMID 16688626. S2CID 28736907.
External links
| Classification |
|---|
- Centre for Clinical Practice at NICE (UK). The Pharmacological Management of Neuropathic Pain in Adults in Non-Specialist Settings. NICE Clinical Guidelines. March 2010.